Region:Middle East
Author(s):Dev
Product Code:KRAA3942
Pages:94
Published On:January 2026
By Service Type:The service type segmentation includes various categories such as Analytical Testing (QC, release, and bioanalytical testing), Stability Testing & Method Development/Validation, Regulatory & CMC Consulting Services, Manufacturing Support & Technology Transfer Services, and Others. This structure is consistent with how global CMC outsourcing is categorized, where analytical and quality control services, method development/validation, regulatory CMC support, and tech transfer/manufacturing support form the core service clusters offered by specialized CROs and CDMOs. Among these, Analytical Testing is the leading sub-segment due to the increasing emphasis on quality control, regulatory-compliant release testing, and specialized bioanalytical capabilities required for complex small molecules and biologics.

By Product Type:The product type segmentation encompasses API (Small Molecule), API (Large Molecule/Biologics), Finished Drug Formulation, Medical Device & Combination Products, and Others. At the UAE level, CMC outsourcing is typically reported across API, finished drug formulation, and medical device categories, reflecting demand from both originator and generic manufacturers as well as device and combination?product companies. Finished Drug Formulation currently accounts for the largest share of CMC outsourcing revenue in the UAE, followed by API services, driven by strong demand for formulation development, scale?up, and commercial batch release for oral solid, sterile, and specialty dosage forms supplied to regional markets.

The UAE Chemistry Manufacturing Control Services Outsourcing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Eurofins Scientific, SGS SA, Intertek Group plc, Charles River Laboratories International, Inc., Labcorp (Laboratory Corporation of America Holdings), WuXi AppTec Co., Ltd., Pace Analytical Services, LLC, Medpace Holdings, Inc., Q2 Solutions, BioReliance (Merck KGaA), Covance (Labcorp Drug Development), PPD, Inc. (Thermo Fisher Scientific), Charles Lawrence International Ltd., AMETEK, Inc., Merck KGaA contribute to innovation, geographic expansion, and service delivery in this space. Global CMC outsourcing providers are increasingly leveraging UAE?based facilities, regional distribution hubs, and partnerships with local manufacturers and healthcare stakeholders to offer integrated analytical, regulatory, and manufacturing?support solutions across the Middle East and Africa.
The future of the UAE chemistry manufacturing control services outsourcing market appears promising, driven by technological advancements and a growing emphasis on sustainability. As companies increasingly adopt digital solutions and automation, the demand for integrated service offerings is expected to rise. Furthermore, the expansion of contract research organizations (CROs) will create new avenues for collaboration, enhancing service delivery and efficiency in the sector, ultimately positioning the UAE as a leader in the regional market.
| Segment | Sub-Segments |
|---|---|
| By Service Type | Analytical Testing (QC, release, and bioanalytical testing) Stability Testing & Method Development/Validation Regulatory & CMC Consulting Services Manufacturing Support & Technology Transfer Services Others |
| By Product Type | API (Small Molecule) API (Large Molecule/Biologics) Finished Drug Formulation Medical Device & Combination Products Others |
| By End-Use | Pharmaceutical Companies Biotechnology Companies Medical Device Companies Contract Research/Development & Manufacturing Organizations (CROs/CDMOs/CMOs) Others |
| By Application | Preclinical Development & IND-Enabling Studies Clinical Trial Material & CMC Support Commercial Manufacturing Support Lifecycle Management & Post-Approval Changes Others |
| By Company Size (Client Type) | Large Multinational Enterprises Regional & Mid-sized Companies Small Enterprises Startups & Emerging Biotech Others |
| By UAE Region | Abu Dhabi Dubai Sharjah Ajman Others |
| By Ownership & Partnership Model | Global Multinational Providers Regional/UAE-Based Providers Joint Ventures & Strategic Alliances Academic / Government-Linked Entities Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Chemical Manufacturing Quality Control | 100 | Quality Assurance Managers, Laboratory Directors |
| Regulatory Compliance Services | 80 | Compliance Officers, Regulatory Affairs Managers |
| Testing and Analysis Services | 70 | Laboratory Technicians, R&D Managers |
| Outsourcing Service Providers | 60 | Business Development Managers, Operations Directors |
| Market Trends and Insights | 90 | Industry Analysts, Market Research Specialists |
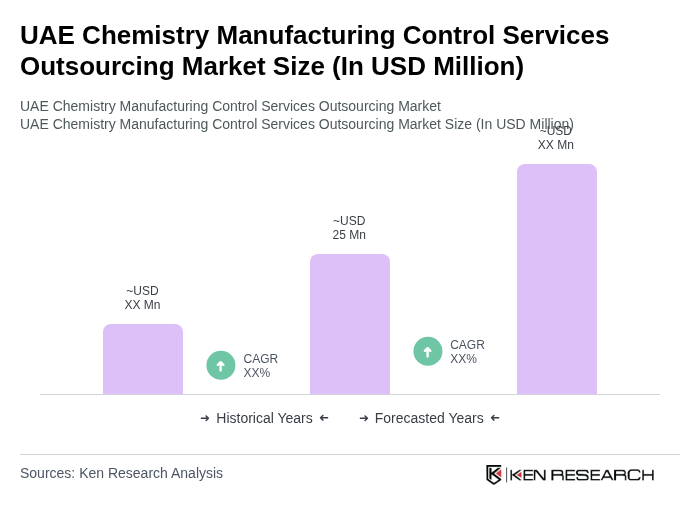
The UAE Chemistry Manufacturing Control Services Outsourcing Market is valued at approximately USD 25 million, reflecting a five-year historical analysis driven by the rising demand for pharmaceutical products and stringent regulatory requirements.