Region:Middle East
Author(s):Geetanshi
Product Code:KRAC9485
Pages:98
Published On:November 2025
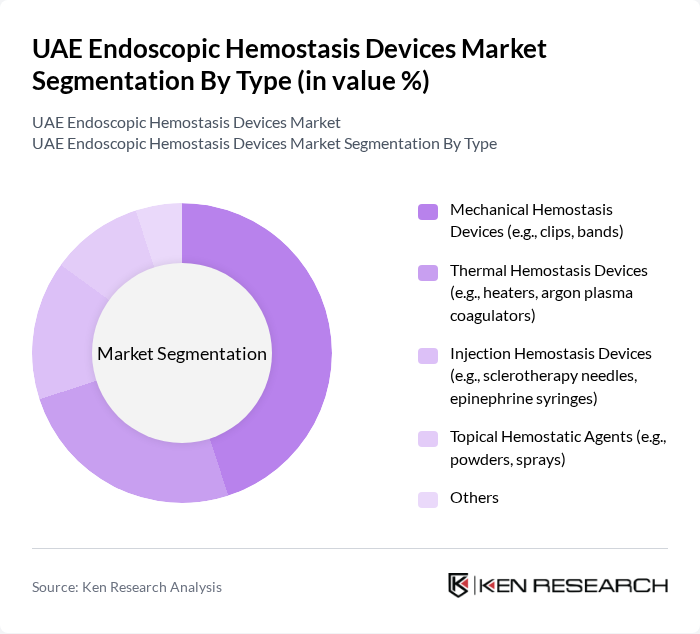
By Type:The market is segmented into various types of hemostatic devices, each catering to specific surgical needs. The primary subsegments include Mechanical Hemostasis Devices, Thermal Hemostasis Devices, Injection Hemostasis Devices, Topical Hemostatic Agents, and Others. Among these, Mechanical Hemostasis Devices—such as clips and bands—lead the market due to their reliability, widespread clinical adoption, and ease of use in diverse surgical procedures. The demand for these devices is driven by their proven effectiveness in controlling gastrointestinal bleeding during endoscopic surgeries.
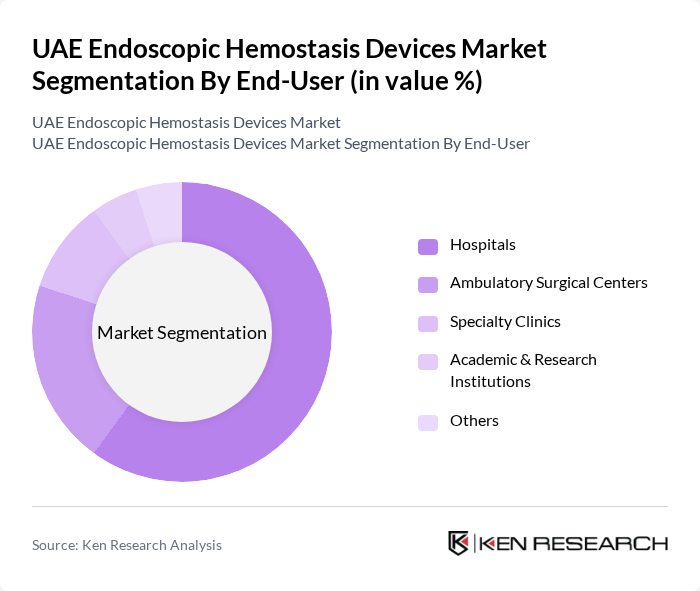
By End-User:The end-user segmentation includes Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Academic & Research Institutions, and Others. Hospitals are the dominant end-user segment, accounting for the largest portion of the market. This is attributed to the high volume of surgical procedures performed in hospitals and the increasing adoption of advanced hemostatic devices to enhance patient safety and surgical outcomes.
The UAE Endoscopic Hemostasis Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Boston Scientific Corporation, Olympus Corporation, Cook Medical LLC, Ethicon (Johnson & Johnson), Stryker Corporation, CONMED Corporation, B. Braun Melsungen AG, Hologic, Inc., Merit Medical Systems, Inc., Medline Industries, LP, EndoChoice, Inc., Intuitive Surgical, Inc., Ambu A/S, Aesculap, Inc. (a division of B. Braun), FUJIFILM Holdings Corporation, KARL STORZ SE & Co. KG, Smith+Nephew plc, STERIS plc, MICRO-TECH (Nanjing) Co., Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE endoscopic hemostasis devices market appears promising, driven by technological advancements and an increasing focus on patient-centric care. As healthcare providers continue to adopt innovative solutions, the integration of artificial intelligence and robotics in endoscopic procedures is expected to enhance efficiency and accuracy. Additionally, the expansion of healthcare infrastructure and the rise of medical tourism will further stimulate demand for these devices, positioning the UAE as a leader in advanced medical technologies.
| Segment | Sub-Segments |
|---|---|
| By Type | Mechanical Hemostasis Devices (e.g., clips, bands) Thermal Hemostasis Devices (e.g., heaters, argon plasma coagulators) Injection Hemostasis Devices (e.g., sclerotherapy needles, epinephrine syringes) Topical Hemostatic Agents (e.g., powders, sprays) Others |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Academic & Research Institutions Others |
| By Application | Gastroenterology (e.g., GI bleeding, peptic ulcers) Pulmonology (e.g., airway bleeding) Urology (e.g., hematuria management) Colorectal Surgery Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Tender/Procurement Agencies Others |
| By Region | Abu Dhabi Dubai Sharjah Northern Emirates Others |
| By Device Material | Stainless Steel Polymer/Plastic Composite Materials Others |
| By Technology | Conventional Techniques Advanced Techniques (e.g., robotic-assisted, AI-enabled) Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gastroenterology Clinics | 60 | Gastroenterologists, Clinic Managers |
| Hospitals with Endoscopy Units | 90 | Surgeons, Hospital Administrators |
| Medical Device Distributors | 40 | Sales Managers, Product Specialists |
| Healthcare Regulatory Bodies | 40 | Regulatory Affairs Officers, Compliance Managers |
| Research Institutions | 50 | Clinical Researchers, Medical Device Analysts |
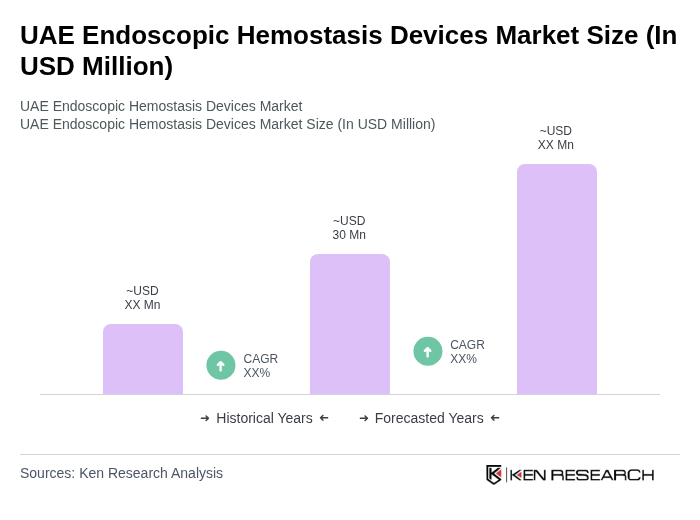
The UAE Endoscopic Hemostasis Devices Market is valued at approximately USD 30 million, reflecting a robust growth driven by the increasing prevalence of gastrointestinal disorders and advancements in endoscopic technologies.