Region:Middle East
Author(s):Dev
Product Code:KRAA3920
Pages:88
Published On:January 2026
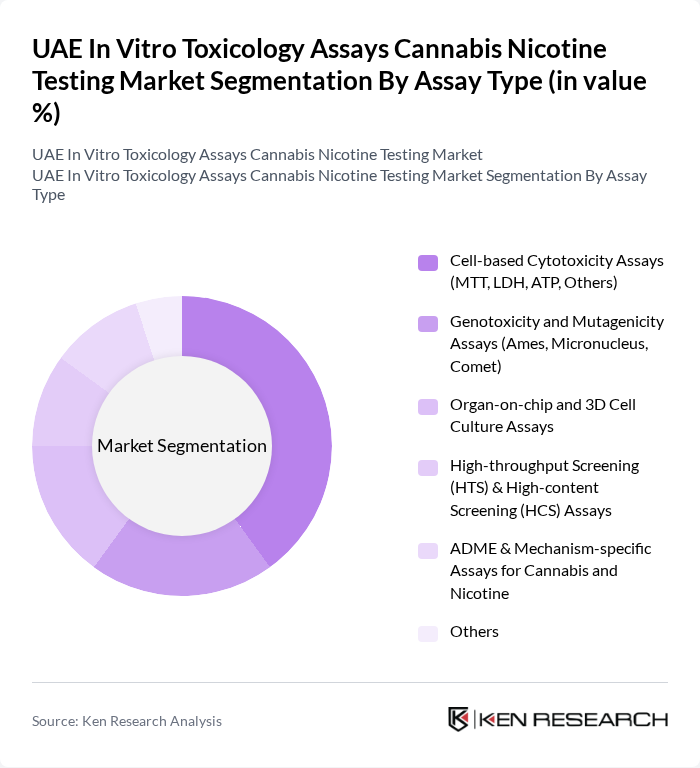
By Assay Type:The assay type segmentation includes various methodologies used to assess the toxicity of cannabis and nicotine products. The dominant sub-segment in this category is Cell-based Cytotoxicity Assays, which are widely utilized due to their effectiveness in evaluating the toxic effects of substances on living cells. This method is favored for its ability to provide rapid and reliable results, is compatible with high-throughput screening formats, and supports mechanistic evaluation of endpoints such as apoptosis, oxidative stress, and mitochondrial function, making it essential for regulatory compliance and product safety assessments for inhalation and oral exposure products.
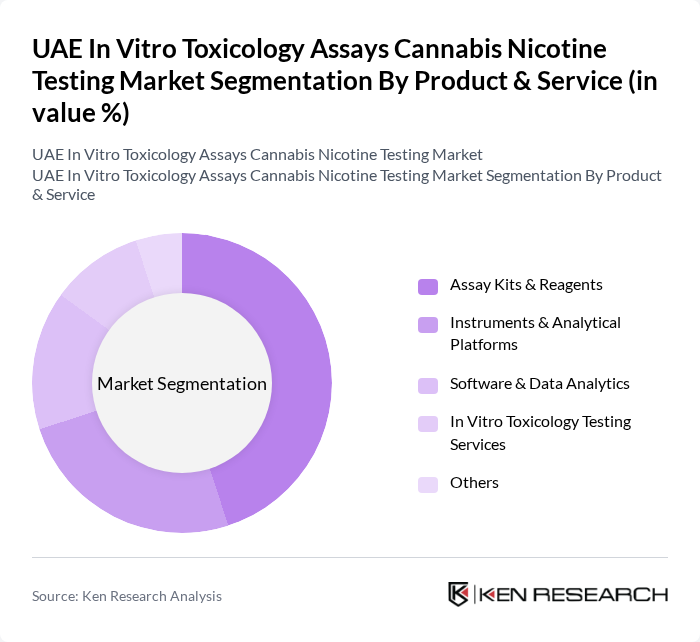
By Product & Service:This segmentation encompasses the various products and services offered in the market, including assay kits, instruments, and testing services. The leading sub-segment is Assay Kits & Reagents, which are essential for conducting toxicology tests and are increasingly tailored for endpoints relevant to inhalation toxicology, e?liquid evaluation, and cannabinoid profiling. The demand for these kits is driven by the increasing number of laboratories and research institutions focusing on cannabis and nicotine testing, the adoption of high-throughput and high-content platforms, and the need for standardized, validated testing protocols that align with international guidelines such as OECD test methods and good laboratory practice requirements.
The UAE In Vitro Toxicology Assays Cannabis Nicotine Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as LabCorp, Quest Diagnostics, Eurofins Scientific, Charles River Laboratories, Thermo Fisher Scientific, MedTox Scientific, Bio-Rad Laboratories, PerkinElmer, Agilent Technologies, Merck KGaA, SGS SA, Intertek Group, Mérieux NutriSciences, Aegis Sciences Corporation, Medpace Holdings contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE in vitro toxicology assays market for cannabis and nicotine testing appears promising, driven by technological advancements and evolving regulatory landscapes. As the government continues to support research initiatives, the market is likely to witness an influx of innovative testing methodologies. Additionally, the increasing integration of artificial intelligence in testing processes is expected to enhance efficiency and accuracy, positioning the UAE as a leader in toxicology testing in the region.
| Segment | Sub-Segments |
|---|---|
| By Assay Type | Cell-based Cytotoxicity Assays (MTT, LDH, ATP, Others) Genotoxicity and Mutagenicity Assays (Ames, Micronucleus, Comet) Organ-on-chip and 3D Cell Culture Assays High-throughput Screening (HTS) & High-content Screening (HCS) Assays ADME & Mechanism-specific Assays for Cannabis and Nicotine Others |
| By Product & Service | Assay Kits & Reagents Instruments & Analytical Platforms Software & Data Analytics In Vitro Toxicology Testing Services Others |
| By Toxicity Endpoint | Acute and Chronic Toxicity Genotoxicity / Mutagenicity Carcinogenicity Reproductive & Developmental Toxicity Organ-specific Toxicity (Cardio, Neuro, Pulmonary, Hepatic, Others) Others |
| By Application | Cannabis Product Safety & Potency Assessment Nicotine & E-cigarette / Vaping Product Evaluation Pharmaceutical R&D and Preclinical Screening Regulatory & Compliance Testing Public Health & Toxicovigilance Studies Others |
| By End-User | Pharmaceutical & Biotechnology Companies Independent & Contract Research Laboratories (CROs) Government & Regulatory Agencies Academic & Research Institutions Hospitals and Clinical Toxicology Centers Others |
| By Region | Abu Dhabi Dubai Sharjah Ajman Others |
| By Regulatory Compliance & Quality Standard | UAE Federal Drug Control Regulations Good Laboratory Practice (GLP) Good Manufacturing Practice (GMP) ISO/IEC 17025 and Other Accreditation Standards Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cannabis Testing Laboratories | 100 | Laboratory Managers, Quality Assurance Officers |
| Nicotine Testing Facilities | 90 | Research Scientists, Compliance Managers |
| Regulatory Bodies | 50 | Policy Makers, Regulatory Affairs Specialists |
| Healthcare Providers | 70 | Medical Directors, Toxicologists |
| Academic Institutions | 60 | Research Professors, Graduate Students in Toxicology |
The UAE In Vitro Toxicology Assays Cannabis Nicotine Testing Market is valued at approximately USD 75 million, reflecting a significant growth driven by the increasing demand for accurate testing methods and regulatory compliance in the healthcare sector.