Region:Middle East
Author(s):Geetanshi
Product Code:KRAB1170
Pages:86
Published On:January 2026
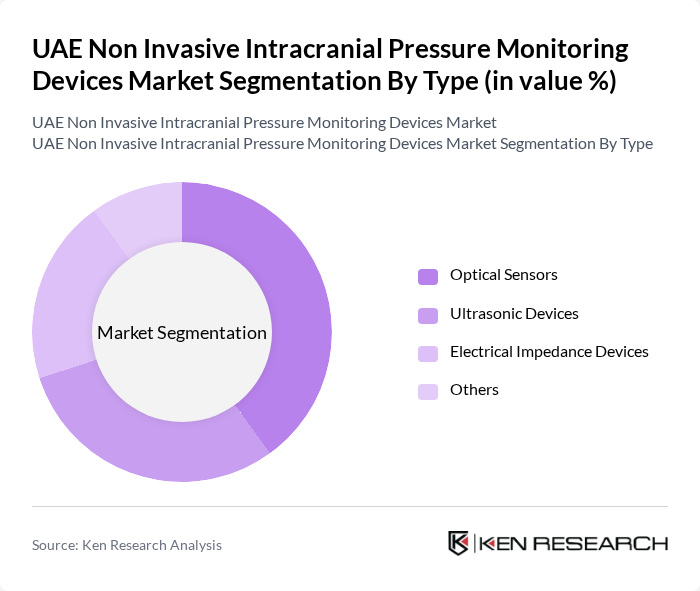
By Type:The market is segmented into various types of non-invasive intracranial pressure monitoring devices, including Optical Sensors, Ultrasonic Devices, Electrical Impedance Devices, and Others. Among these, Optical Sensors are gaining traction due to their accuracy and reliability in measuring intracranial pressure without the need for invasive procedures. Ultrasonic Devices are also popular, particularly in emergency care settings, due to their non-invasive nature and ease of use.
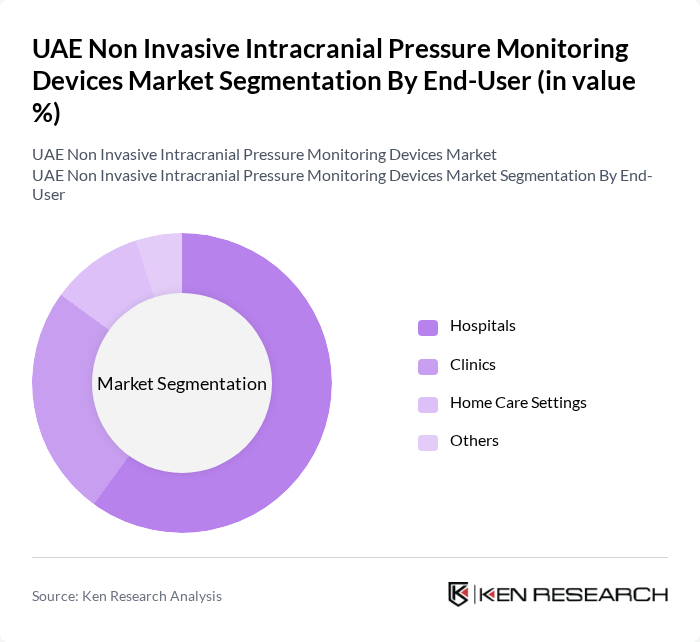
By End-User:The end-user segmentation includes Hospitals, Clinics, Home Care Settings, and Others. Hospitals are the primary end-users of non-invasive ICP monitoring devices, driven by the need for advanced monitoring solutions in critical care units. Clinics are also increasingly adopting these devices for outpatient monitoring, while home care settings are emerging as a growing segment due to the rising trend of remote patient monitoring.
The UAE Non Invasive Intracranial Pressure Monitoring Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Natus Medical Incorporated, Raumedic AG, Codman Neuro, Integra LifeSciences, NeuroWave Systems, Ornim Medical, BrainScope Company, Inc., Nonin Medical, Inc., Cereset, Inc., HeadSense Medical, Eko Devices, Advanced Brain Monitoring, Neurotech Solutions, Soterix Medical contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE non-invasive intracranial pressure monitoring devices market appears promising, driven by digital transformation in healthcare and advancements in artificial intelligence. The integration of national e-health systems and telehealth initiatives will facilitate the adoption of non-invasive monitoring solutions. Additionally, the development of AI infrastructure will enhance data processing capabilities, making it easier to implement sophisticated monitoring technologies that improve patient outcomes and operational efficiencies in healthcare settings.
| Segment | Sub-Segments |
|---|---|
| By Type | Optical Sensors Ultrasonic Devices Electrical Impedance Devices Others |
| By End-User | Hospitals Clinics Home Care Settings Others |
| By Application | Emergency Care Neurological Monitoring Post-Surgical Monitoring Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Region | Abu Dhabi Dubai Sharjah Others |
| By Technology | Non-invasive Pressure Sensors Wearable Monitoring Devices Telemonitoring Systems Others |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurosurgery Departments | 100 | Neurosurgeons, Clinical Directors |
| Critical Care Units | 80 | Intensivists, Critical Care Nurses |
| Medical Device Procurement | 70 | Procurement Managers, Supply Chain Officers |
| Healthcare Technology Assessment | 60 | Health Economists, Policy Makers |
| Medical Device Distributors | 50 | Sales Managers, Product Specialists |
The UAE Non Invasive Intracranial Pressure Monitoring Devices Market is valued at approximately USD 10 million, reflecting a growing interest in advanced medical technologies due to rising incidences of traumatic brain injuries and strokes.