Region:Middle East
Author(s):Shubham
Product Code:KRAD1962
Pages:91
Published On:December 2025
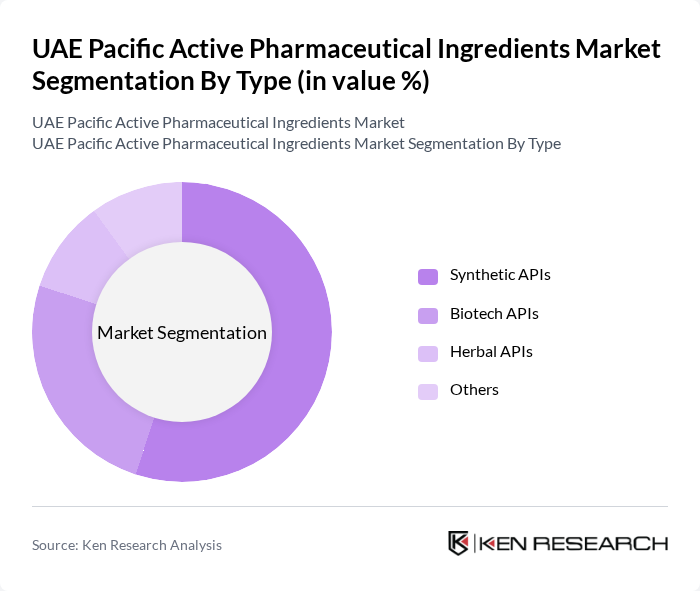
By Type:The market is segmented into various types of active pharmaceutical ingredients, including Synthetic APIs, Biotech APIs, Herbal APIs, and Others. Among these, Synthetic APIs dominate the market due to their widespread use in the production of generic drugs and their cost-effectiveness. The increasing demand for affordable medications has led to a significant rise in the production and consumption of synthetic APIs, making them a crucial component of the pharmaceutical industry.
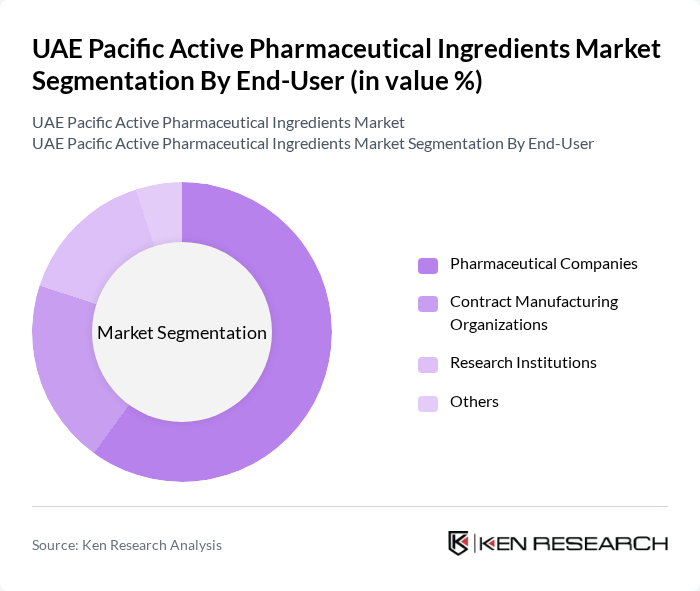
By End-User:The end-user segment includes Pharmaceutical Companies, Contract Manufacturing Organizations (CMOs), Research Institutions, and Others. Pharmaceutical companies hold the largest share in this segment, driven by the increasing demand for innovative and generic drugs. The growth of the pharmaceutical sector in the UAE, supported by government initiatives and investments, has further solidified the position of pharmaceutical companies as the primary consumers of active pharmaceutical ingredients.
The UAE Pacific Active Pharmaceutical Ingredients Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abu Dhabi Pharmaceutical Manufacturing Company, Julphar, Neopharma, Gulf Pharmaceutical Industries (Julphar), Hikma Pharmaceuticals, Aster DM Healthcare, Al Ain Pharmaceutical Manufacturing, Pharma International, United Pharmaceuticals, Emirates Pharmaceutical Industries, Globalpharma, Tabuk Pharmaceuticals, Al Haramain Pharmaceuticals, Dar Al Dawa Development & Investment, Medpharma contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE Pacific Active Pharmaceutical Ingredients market appears promising, driven by advancements in digital transformation and technology integration. The Ministry of Health and Prevention (MoHAP) plans to launch a unified digital healthcare licensing platform by future, enhancing operational efficiency. Additionally, the growing adoption of AI and machine learning in pharmaceutical processes is expected to revolutionize API production, ensuring better quality control and innovation. These trends will likely position the UAE as a leader in the regional pharmaceutical landscape.
| Segment | Sub-Segments |
|---|---|
| By Type | Synthetic APIs Biotech APIs Herbal APIs Others |
| By End-User | Pharmaceutical Companies Contract Manufacturing Organizations Research Institutions Others |
| By Application | Cardiovascular Oncology Neurology Others |
| By Source | Domestic Production Imports Exports Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Formulation | Solid Formulations Liquid Formulations Semi-Solid Formulations Others |
| By Therapeutic Area | Infectious Diseases Metabolic Disorders Autoimmune Diseases Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 100 | Production Managers, Quality Control Officers |
| Healthcare Providers | 80 | Pharmacy Managers, Hospital Administrators |
| Regulatory Bodies | 50 | Regulatory Affairs Specialists, Compliance Officers |
| Research Institutions | 60 | Research Scientists, Academic Professors |
| Distributors and Wholesalers | 70 | Supply Chain Managers, Sales Directors |
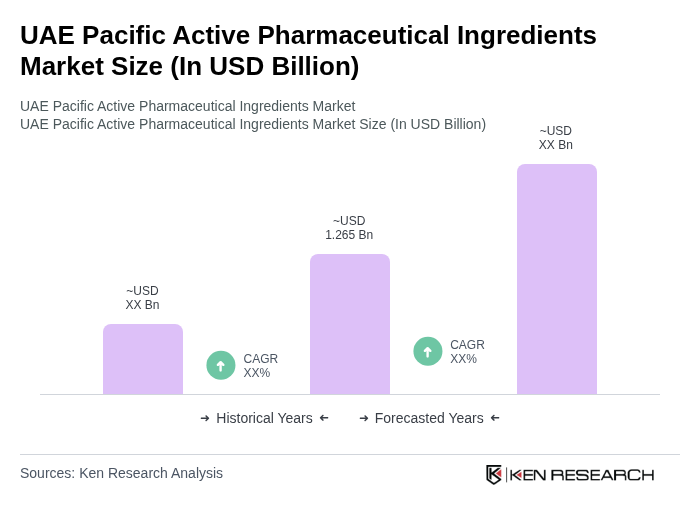
The UAE Pacific Active Pharmaceutical Ingredients Market is valued at approximately USD 1.265 billion, driven by the rising prevalence of chronic diseases and the strong adoption of generic and biosimilar drugs, alongside advancements in biopharmaceutical manufacturing.