Region:Middle East
Author(s):Geetanshi
Product Code:KRAA9184
Pages:89
Published On:November 2025
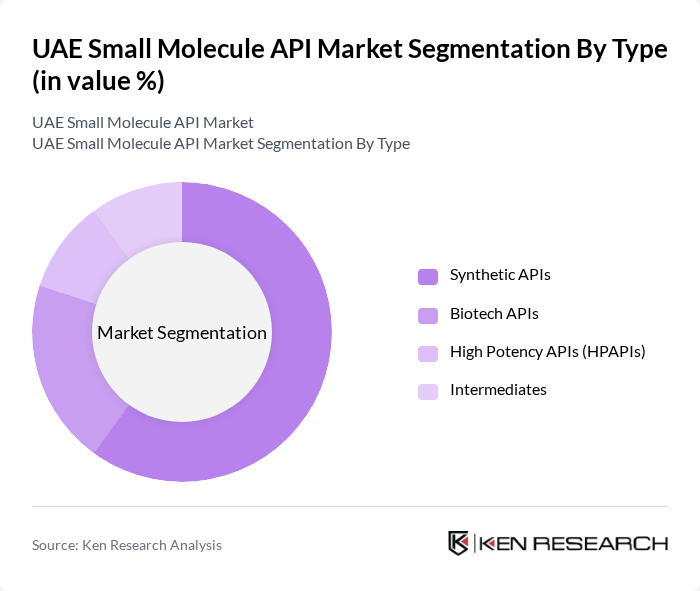
By Type:The market is segmented into Synthetic APIs, Biotech APIs, High Potency APIs (HPAPIs), and Intermediates. Among these, Synthetic APIs dominate the market due to their widespread application in various therapeutic areas and the established manufacturing processes that allow for cost-effective production. The increasing demand for generic drugs further propels the growth of this segment, as synthetic APIs are often the primary components in these formulations .
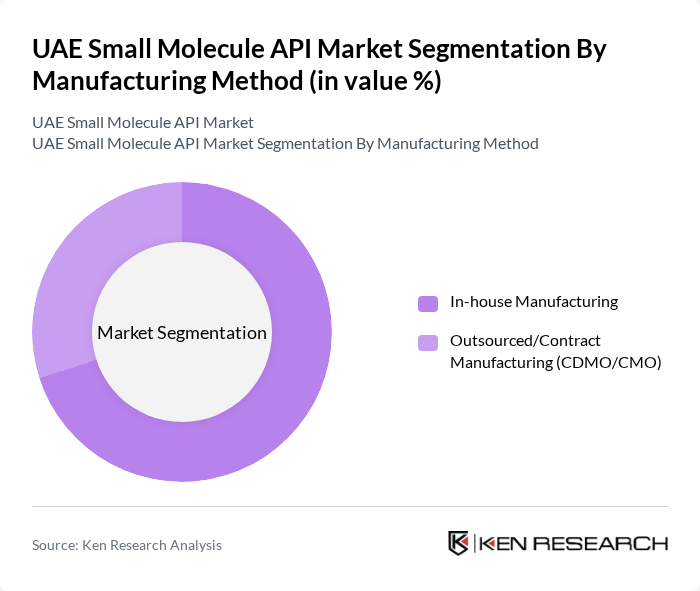
By Manufacturing Method:The market is divided into In-house Manufacturing and Outsourced/Contract Manufacturing (CDMO/CMO). In-house manufacturing is the leading method due to the control it offers over production processes and quality assurance. Many pharmaceutical companies prefer this method to maintain proprietary technologies and ensure compliance with regulatory standards, which is crucial in the highly regulated API market. This trend is consistent with the broader global market, where in-house manufacturing remains dominant .
The UAE Small Molecule API Market is characterized by a dynamic mix of regional and international players. Leading participants such as Julphar (Gulf Pharmaceutical Industries), Neopharma, Globalpharma (a Sanofi company), Hikma Pharmaceuticals, Pharma International, Aster DM Healthcare, Al Ain Pharmaceutical Manufacturing Co., United Pharmaceuticals, Tabuk Pharmaceuticals, Bayer Middle East, Sanofi UAE, Pfizer Gulf FZ LLC, Merck Sharp & Dohme (MSD GCC), GlaxoSmithKline (GSK Gulf), AbbVie Biopharmaceuticals GmbH contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE small molecule API market appears promising, driven by increasing healthcare investments and a growing focus on local production. As the government continues to implement policies that support domestic manufacturing, the market is likely to see enhanced capabilities and reduced dependency on imports. Additionally, the rise of personalized medicine and digital technologies in manufacturing processes will further transform the landscape, fostering innovation and efficiency in API production.
| Segment | Sub-Segments |
|---|---|
| By Type | Synthetic APIs Biotech APIs High Potency APIs (HPAPIs) Intermediates |
| By Manufacturing Method | In-house Manufacturing Outsourced/Contract Manufacturing (CDMO/CMO) |
| By End-User | Pharmaceutical Companies Biotechnology Companies Contract Development and Manufacturing Organizations (CDMOs/CMOs) Research Institutions |
| By Therapeutic Area | Oncology Cardiovascular Central Nervous System (CNS) & Neurology Endocrinology & Metabolic Disorders Infectious Diseases Others |
| By Formulation Type | Oral Injectable Topical Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Geography | Abu Dhabi Dubai Sharjah Others |
| By Policy Support | Subsidies Tax Incentives Research Grants Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical API Manufacturers | 80 | Production Managers, Quality Control Heads |
| Regulatory Affairs in Pharmaceuticals | 60 | Regulatory Managers, Compliance Officers |
| Research & Development in Biotech | 50 | R&D Directors, Lead Scientists |
| Procurement in Pharmaceutical Companies | 70 | Procurement Directors, Supply Chain Managers |
| Healthcare Policy Makers | 40 | Health Economists, Policy Analysts |
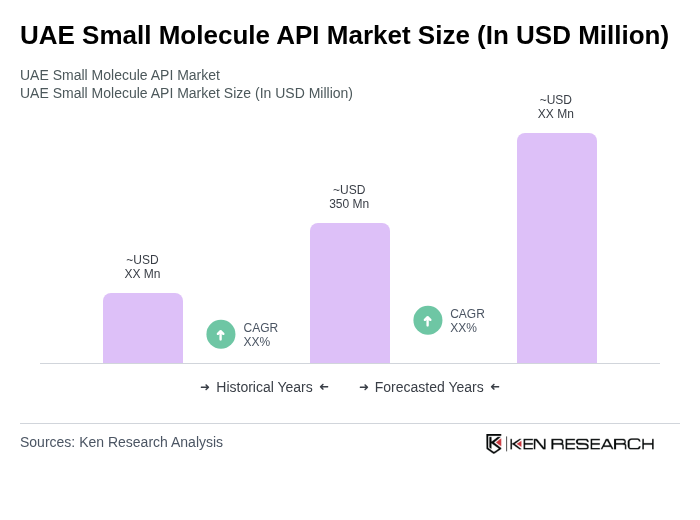
The UAE Small Molecule API Market is valued at approximately USD 350 million, driven by the increasing demand for generic drugs, advancements in pharmaceutical research, and the rising prevalence of chronic diseases.