Region:North America
Author(s):Dev
Product Code:KRAC4733
Pages:97
Published On:October 2025
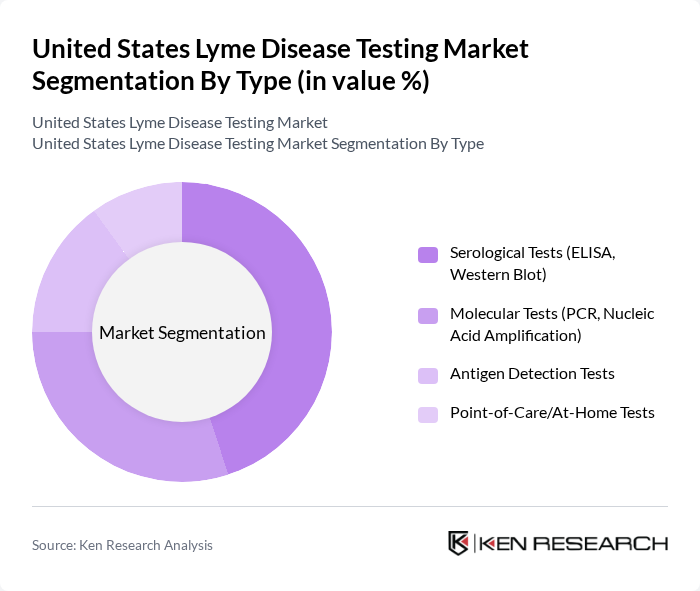
By Type:The market is segmented into various types of testing methods, including Serological Tests (ELISA, Western Blot), Molecular Tests (PCR, Nucleic Acid Amplification), Antigen Detection Tests, and Point-of-Care/At-Home Tests. Among these, Serological Tests are the most widely used due to their established reliability and accuracy in detecting Lyme disease antibodies. Molecular Tests are gaining traction due to their rapid results and high sensitivity, particularly in early-stage infections. The increasing adoption of point-of-care and at-home tests is also notable, driven by patient demand for convenience and faster diagnosis .
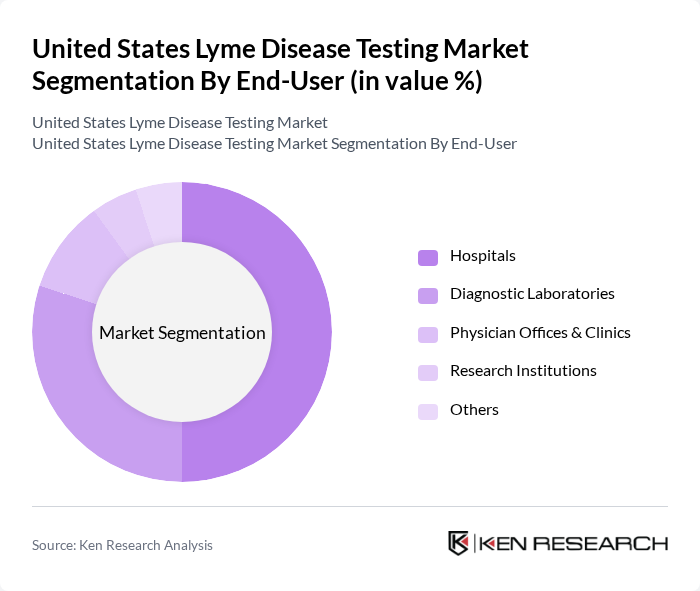
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Physician Offices & Clinics, Research Institutions, and Others. Hospitals are the leading end-users due to their capacity to handle complex cases and provide comprehensive care. Diagnostic Laboratories follow closely, driven by the demand for specialized testing services and the need for accurate diagnostics in clinical settings. Physician offices and clinics are increasingly utilizing rapid and point-of-care tests to facilitate early diagnosis, while research institutions contribute to the development of novel diagnostic technologies .
The United States Lyme Disease Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Quest Diagnostics, Bio-Rad Laboratories, Roche Diagnostics, Siemens Healthineers, Cepheid, Hologic, Inc., Laboratory Corporation of America Holdings (LabCorp), Thermo Fisher Scientific, Becton, Dickinson and Company, Meridian Bioscience, InBios International, Inc., IGeneX Inc., bioMérieux, DiaSorin S.p.A., Trinity Biotech plc, Zeus Scientific, Inc., Gold Standard Diagnostics, Oxford Immunotec, T2 Biosystems, Inc. contribute to innovation, geographic expansion, and service delivery in this space .
The future of the Lyme disease testing market in the United States appears promising, driven by technological advancements and increased public awareness. As diagnostic methods continue to evolve, the integration of artificial intelligence in testing processes is expected to enhance accuracy and efficiency. Additionally, the expansion of telemedicine services will facilitate remote consultations and testing, making it easier for patients in rural areas to access necessary care. These trends indicate a robust growth trajectory for the market in future.
| Segment | Sub-Segments |
|---|---|
| By Type | Serological Tests (ELISA, Western Blot) Molecular Tests (PCR, Nucleic Acid Amplification) Antigen Detection Tests Point-of-Care/At-Home Tests |
| By End-User | Hospitals Diagnostic Laboratories Physician Offices & Clinics Research Institutions Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Pharmacies Others |
| By Test Method | Enzyme Immunoassays (EIA/ELISA) Western Blot Nucleic Acid Amplification Tests (NAAT/PCR) Rapid Diagnostic Tests Others |
| By Sample Type | Blood Samples Cerebrospinal Fluid (CSF) Urine Samples Tissue Samples Others |
| By Age Group | Pediatric Adult Geriatric |
| By Geographic Region | Northeast Midwest South West |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers (General Practitioners) | 100 | Family Physicians, Internists |
| Diagnostic Laboratories | 60 | Laboratory Managers, Technicians |
| Infectious Disease Specialists | 40 | Infectious Disease Physicians, Epidemiologists |
| Patient Advocacy Groups | 40 | Advocacy Leaders, Lyme Disease Survivors |
| Public Health Officials | 50 | Public Health Directors, Epidemiologists |
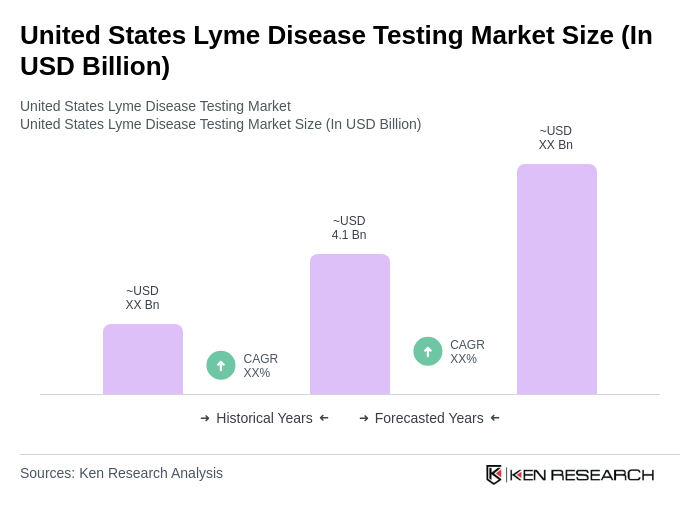
The United States Lyme Disease Testing Market is valued at approximately USD 4.1 billion, reflecting a significant increase driven by the rising incidence of Lyme disease and advancements in diagnostic technologies.