About the Report
Base Year 2024US Immunomodulators Market Overview
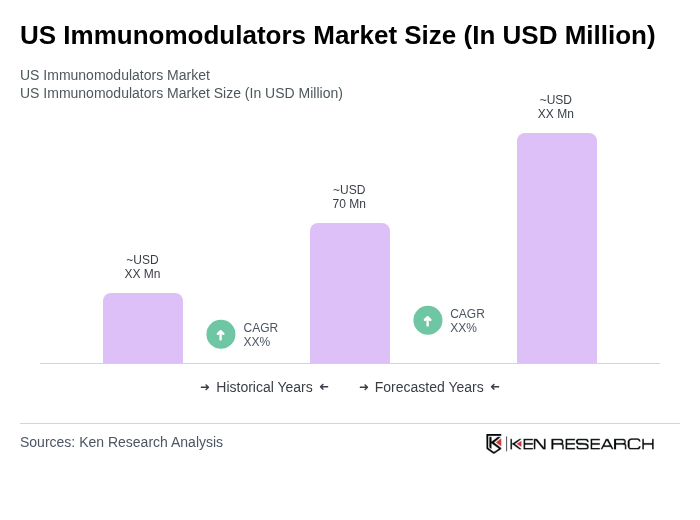
- The US Immunomodulators Market is valued at USD 70 million, based on recent analysis. Growth is primarily driven by a rising prevalence of autoimmune disorders and increasing demand for targeted immunotherapies, as immunosuppressants remain essential for managing transplantation and chronic immune-related conditions. Expansion is further supported by innovations in biologics, monoclonal antibodies, and cytokine therapies, which are progressively adopted in treating complex diseases.
- Key players in this market include F. Hoffmann-La Roche Ltd., Amgen Inc., AbbVie Inc., Novartis AG, Bristol-Myers Squibb Company, Merck & Co., Inc., Pfizer Inc., Johnson & Johnson, Sanofi S.A., Biogen Inc., and Gilead Sciences Inc. These companies play a significant role in driving growth and maintaining high standards in immunomodulatory therapies.
- In 2024, the FDA proposed to simplify the Investigational New Drug (IND) process, including reduced data submission burdens and streamlined clinical hold criteria, leading to estimated annual savings of USD 3.3 million for sponsors and facilitating faster access to innovative therapies.
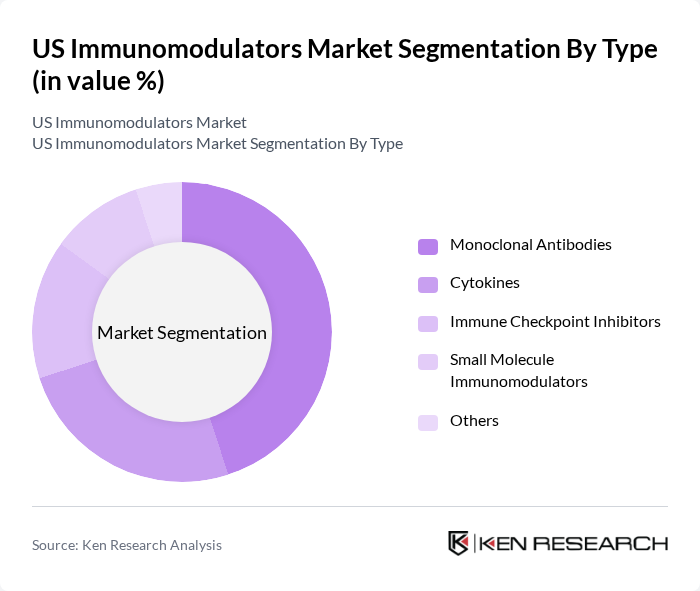
US Immunomodulators Market Segmentation
By Type:The immunomodulators market is segmented into various types, including Monoclonal Antibodies, Cytokines, Immune Checkpoint Inhibitors, Small Molecule Immunomodulators, and Others. Among these, Monoclonal Antibodies dominate the market due to their targeted action and effectiveness in treating a range of autoimmune diseases. The increasing adoption of these therapies in clinical settings is driven by their ability to provide specific treatment options with fewer side effects compared to traditional therapies.
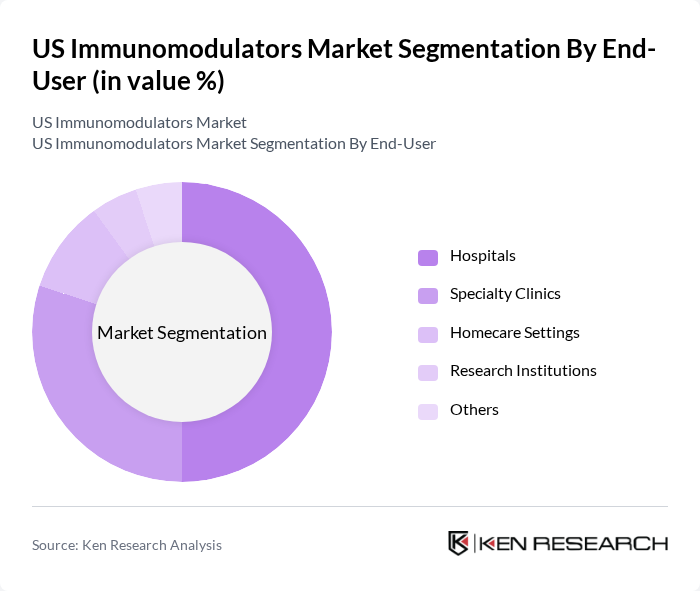
By End-User:The end-user segmentation includes Hospitals, Specialty Clinics, Homecare Settings, Research Institutions, and Others. Hospitals are the leading end-users of immunomodulators, primarily due to their capacity to provide comprehensive care and access to advanced treatment options. The increasing number of patients with chronic diseases requiring immunomodulatory therapies in hospital settings drives this segment's growth.
US Immunomodulators Market Competitive Landscape
The US Immunomodulators Market is characterized by a dynamic mix of regional and international players. Leading participants such as AbbVie Inc., Amgen Inc., Johnson & Johnson, Merck & Co., Inc., Pfizer Inc., Bristol-Myers Squibb Company, Gilead Sciences, Inc., Sanofi S.A., Novartis AG, Eli Lilly and Company, Regeneron Pharmaceuticals, Inc., Takeda Pharmaceutical Company Limited, Biogen Inc., Astellas Pharma Inc., and UCB S.A. contribute to innovation, geographic expansion, and service delivery in this space.
US Immunomodulators Market Industry Analysis
Growth Drivers
- High U.S. Market Revenue in Future:The U.S. market for immunomodulators is projected to reach approximately USD 68.3 million in future. This substantial revenue reflects a strong domestic demand for immunomodulatory therapies, driven by an increasing prevalence of autoimmune diseases and a growing focus on personalized medicine. The investment in innovative treatment options is expected to further bolster market growth, as healthcare providers seek effective solutions for complex health conditions.
- North America Leadership – U.S. Share:In future, North America is anticipated to generate USD 89.5 billion in immunomodulators revenue, with the U.S. accounting for approximately 78% of this total. This dominance is attributed to the advanced healthcare infrastructure, significant research and development capabilities, and a robust regulatory framework that supports the introduction of new therapies. The U.S. market's leadership is crucial for driving innovation and improving patient outcomes in immunomodulation.
- Substantial Organ Transplant Volume Future:The U.S. is expected to perform around 48,149 organ transplants in future, significantly driving the demand for immunosuppressant therapies, a key sub-segment of immunomodulators. This high volume of transplants necessitates effective immunomodulatory treatments to prevent organ rejection, thereby creating a consistent market for these therapies. The increasing number of transplant procedures highlights the critical role of immunomodulators in enhancing patient survival and quality of life.
Market Challenges
- High Cost of Immunomodulatory Therapies:The cost of immunomodulatory therapies poses a significant barrier to access, with some checkpoint inhibitor regimens exceeding USD 100,000 annually per patient. This high financial burden affects patients, payers, and healthcare systems, limiting the widespread adoption of these therapies. The economic strain associated with such costs can hinder treatment accessibility, particularly for underinsured populations, thereby impacting overall market growth.
- Regulatory Complexity and Approval Burden:The regulatory landscape for immunomodulators is complex, with the FDA having approved 60 biosimilars as of future. This intricate approval process can slow market entry for new therapies, particularly biosimilars that could reduce costs and increase competition. The lengthy timelines and stringent requirements for clinical trials and safety assessments can deter investment and innovation in the immunomodulator sector, posing a challenge to market expansion.
US Immunomodulators Market Future Outlook
The future of the U.S. immunomodulators market appears promising, driven by advancements in artificial intelligence and machine learning that are transforming drug development processes. The FDA's consideration of AI-based models for preclinical testing could significantly reduce time-to-market for new therapies. Additionally, the growing pipeline of immunostimulants, projected to reach USD 65,036.95 million by future, indicates a shift towards enhancing immune responses, which may open new therapeutic avenues in oncology and inflammatory diseases.
Market Opportunities
- Organ Transplantation Segment:The U.S. immunosuppressant market is projected to reach USD 23,000 million by future. This substantial market size reflects the ongoing demand for specialized immunosuppressive regimens, driven by the increasing number of organ transplants. Companies can capitalize on this opportunity by developing targeted therapies that address the unique needs of transplant patients, enhancing treatment efficacy and patient outcomes.
- Immunostimulant Expansion:The U.S. immunostimulants market is expected to reach USD 18,900 million by future. This growth presents opportunities for innovation in immune-enhancing treatments, particularly in oncology and infectious diseases. As research progresses, companies can explore new applications for immunostimulants, potentially leading to breakthroughs in treatment paradigms and expanding their market presence.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Monoclonal Antibodies Cytokines Immune Checkpoint Inhibitors Small Molecule Immunomodulators Others |
| By End-User | Hospitals Specialty Clinics Homecare Settings Research Institutions Others |
| By Disease Type | Rheumatoid Arthritis Multiple Sclerosis Psoriasis Inflammatory Bowel Disease Others |
| By Route of Administration | Injectable Oral Intravenous Others |
| By Distribution Channel | Retail Pharmacies Online Pharmacies Hospital Pharmacies Others |
| By Region | Northeast Midwest South West |
| By Patient Demographics | Age Group (Pediatric, Adult, Geriatric) Gender Socioeconomic Status Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration, Centers for Disease Control and Prevention)
Pharmaceutical Manufacturers
Biotechnology Companies
Healthcare Providers and Institutions
Health Insurance Companies
Clinical Research Organizations
Pharmaceutical Distributors
Players Mentioned in the Report:
AbbVie Inc.
Amgen Inc.
Johnson & Johnson
Merck & Co., Inc.
Pfizer Inc.
Bristol-Myers Squibb Company
Gilead Sciences, Inc.
Sanofi S.A.
Novartis AG
Eli Lilly and Company
Regeneron Pharmaceuticals, Inc.
Takeda Pharmaceutical Company Limited
Biogen Inc.
Astellas Pharma Inc.
UCB S.A.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. US Immunomodulators Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 US Immunomodulators Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. US Immunomodulators Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of autoimmune diseases
3.1.2 Rising demand for personalized medicine
3.1.3 Advancements in biotechnology and drug development
3.1.4 Growing investment in research and development
3.2 Market Challenges
3.2.1 High cost of immunomodulator therapies
3.2.2 Stringent regulatory requirements
3.2.3 Competition from alternative therapies
3.2.4 Limited patient access in rural areas
3.3 Market Opportunities
3.3.1 Expansion into emerging markets
3.3.2 Development of combination therapies
3.3.3 Increasing collaborations between biotech firms
3.3.4 Growth in telemedicine and digital health solutions
3.4 Market Trends
3.4.1 Shift towards biologics and biosimilars
3.4.2 Focus on patient-centric treatment approaches
3.4.3 Integration of artificial intelligence in drug discovery
3.4.4 Rise of home-based healthcare solutions
3.5 Government Regulation
3.5.1 FDA approval processes for new immunomodulators
3.5.2 Policies promoting biosimilar market entry
3.5.3 Regulations on drug pricing and reimbursement
3.5.4 Guidelines for clinical trials and patient safety
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. US Immunomodulators Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. US Immunomodulators Market Segmentation
8.1 By Type
8.1.1 Monoclonal Antibodies
8.1.2 Cytokines
8.1.3 Immune Checkpoint Inhibitors
8.1.4 Small Molecule Immunomodulators
8.1.5 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Specialty Clinics
8.2.3 Homecare Settings
8.2.4 Research Institutions
8.2.5 Others
8.3 By Disease Type
8.3.1 Rheumatoid Arthritis
8.3.2 Multiple Sclerosis
8.3.3 Psoriasis
8.3.4 Inflammatory Bowel Disease
8.3.5 Others
8.4 By Route of Administration
8.4.1 Injectable
8.4.2 Oral
8.4.3 Intravenous
8.4.4 Others
8.5 By Distribution Channel
8.5.1 Retail Pharmacies
8.5.2 Online Pharmacies
8.5.3 Hospital Pharmacies
8.5.4 Others
8.6 By Region
8.6.1 Northeast
8.6.2 Midwest
8.6.3 South
8.6.4 West
8.7 By Patient Demographics
8.7.1 Age Group (Pediatric, Adult, Geriatric)
8.7.2 Gender
8.7.3 Socioeconomic Status
8.7.4 Others
9. US Immunomodulators Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Development Cycle Time
9.2.8 Sales Conversion Rate
9.2.9 Distribution Efficiency
9.2.10 Brand Equity Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 AbbVie Inc.
9.5.2 Amgen Inc.
9.5.3 Johnson & Johnson
9.5.4 Merck & Co., Inc.
9.5.5 Pfizer Inc.
9.5.6 Bristol-Myers Squibb Company
9.5.7 Gilead Sciences, Inc.
9.5.8 Sanofi S.A.
9.5.9 Novartis AG
9.5.10 Eli Lilly and Company
9.5.11 Regeneron Pharmaceuticals, Inc.
9.5.12 Takeda Pharmaceutical Company Limited
9.5.13 Biogen Inc.
9.5.14 Astellas Pharma Inc.
9.5.15 UCB S.A.
10. US Immunomodulators Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Compliance Requirements
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends
10.2.2 Funding Sources
10.2.3 Strategic Partnerships
10.2.4 Cost Management Strategies
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to Treatment
10.3.2 Affordability Issues
10.3.3 Quality of Care
10.3.4 Availability of Information
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training Needs
10.4.3 Technology Acceptance
10.4.4 Support Systems
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 User Feedback Mechanisms
10.5.3 Scalability Potential
10.5.4 Long-term Value Assessment
11. US Immunomodulators Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Competitive Advantage Assessment
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Market Identification
2.4 Communication Strategy
2.5 Digital Marketing Approaches
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 E-commerce Integration
3.4 Direct Sales Channels
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Comparison
5. Unmet Demand & Latent Needs
5.1 Category Gaps Identification
5.2 Consumer Segments Analysis
5.3 Product Development Opportunities
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Feedback Mechanisms
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Customer-Centric Approaches
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Innovations
9.2 Export Entry Strategy
9.2.1 Target Countries Selection
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements Analysis
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
12.2 Risk Mitigation Strategies
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Assessment
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Identification
15.2.2 Activity Scheduling
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from industry associations such as the American Society of Clinical Oncology
- Review of published articles in peer-reviewed journals focusing on immunomodulators
- Examination of regulatory documents from the FDA regarding approved immunomodulator therapies
Primary Research
- Interviews with oncologists and rheumatologists to understand prescribing patterns
- Surveys with pharmacists to gauge market availability and patient access
- Field interviews with healthcare administrators to assess budget allocations for immunomodulators
Validation & Triangulation
- Cross-validation of data through multiple sources including clinical trial results and market surveys
- Triangulation of findings from primary interviews with secondary data insights
- Sanity checks through expert panels comprising healthcare professionals and market analysts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total market size based on national healthcare expenditure on immunomodulators
- Segmentation by therapeutic area, including oncology, autoimmune diseases, and transplant medicine
- Incorporation of demographic trends and disease prevalence rates in the US population
Bottom-up Modeling
- Collection of sales data from leading pharmaceutical companies specializing in immunomodulators
- Estimation of patient populations receiving treatment based on clinical guidelines
- Volume x average treatment cost calculations to derive revenue projections
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as healthcare policy changes and drug pricing trends
- Scenario modeling based on potential market disruptions, including new entrants and patent expirations
- Baseline, optimistic, and pessimistic forecasts through 2030 based on historical growth rates
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Immunomodulator Prescriptions | 150 | Oncologists, Clinical Pharmacists |
| Rheumatology Treatment Patterns | 100 | Rheumatologists, Nurse Practitioners |
| Patient Access and Affordability | 80 | Healthcare Administrators, Insurance Analysts |
| Clinical Trial Insights | 70 | Clinical Researchers, Trial Coordinators |
| Market Trends in Autoimmune Therapies | 90 | Immunologists, Health Economists |
Frequently Asked Questions
What is the current value of the US Immunomodulators Market?
The US Immunomodulators Market is currently valued at approximately USD 70 million. This valuation reflects the increasing demand for targeted immunotherapies and the rising prevalence of autoimmune disorders, alongside the essential role of immunosuppressants in managing chronic immune-related conditions.