Region:North America
Author(s):Geetanshi
Product Code:KRAC4562
Pages:88
Published On:October 2025
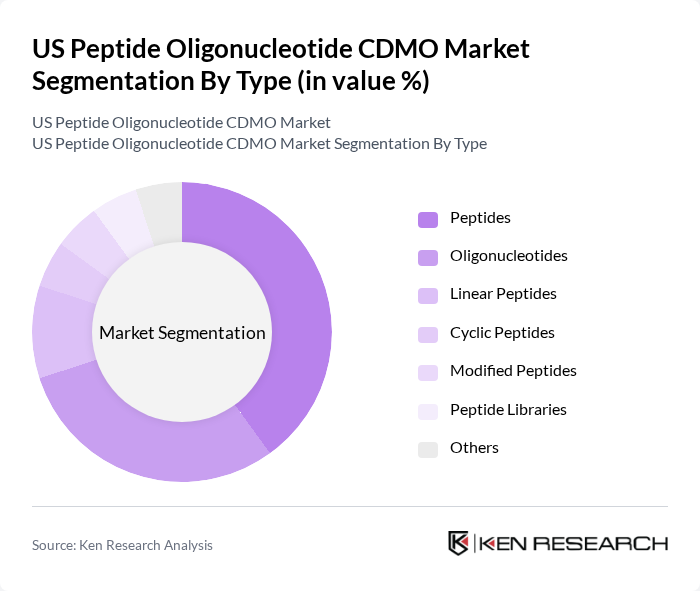
By Type:The market is segmented into various types, including peptides, oligonucleotides, linear peptides, cyclic peptides, modified peptides, peptide libraries, and others. Among these, peptides and oligonucleotides are the most significant contributors to market growth. Peptides are widely used in therapeutics due to their specificity and efficacy, while oligonucleotides are gaining traction in gene therapy and personalized medicine. The demand for modified peptides and peptide libraries is also increasing, driven by advancements in drug discovery and development .
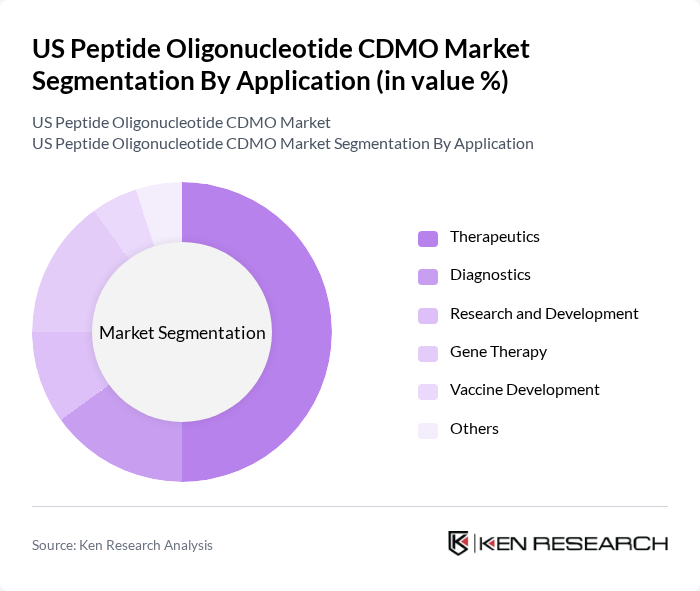
By Application:The applications of peptide and oligonucleotide products are diverse, including therapeutics, diagnostics, research and development, gene therapy, vaccine development, and others. The therapeutics segment is the largest, driven by the increasing prevalence of diseases such as cancer and diabetes, which require innovative treatment options. Gene therapy is also emerging as a significant application, with growing investments in research and clinical trials aimed at developing novel therapies .
The US Peptide Oligonucleotide CDMO Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lonza Group AG, WuXi AppTec, Bachem Holding AG, PeptiDream Inc., CordenPharma, Ferring Pharmaceuticals, Evonik Industries AG, AAPPTec LLC, GenScript Biotech Corporation, PolyPeptide Group AG, Thermo Fisher Scientific Inc., Curia Global, Inc., Creative Peptides, Merck KGaA, Euroapi SA contribute to innovation, geographic expansion, and service delivery in this space .
The US peptide oligonucleotide CDMO market is poised for significant growth, driven by technological advancements and increasing demand for personalized therapies. As biopharmaceutical companies continue to prioritize innovative drug development, the outsourcing of manufacturing processes is expected to rise. Additionally, the integration of artificial intelligence in drug discovery and production processes will enhance efficiency and reduce time-to-market, positioning CDMOs as critical partners in the evolving healthcare landscape.
| Segment | Sub-Segments |
|---|---|
| By Type | Peptides Oligonucleotides Linear Peptides Cyclic Peptides Modified Peptides Peptide Libraries Others |
| By Application | Therapeutics Diagnostics Research and Development Gene Therapy Vaccine Development Others |
| By End-User | Pharmaceutical Companies Biopharmaceutical Companies Biotechnology Firms Academic & Research Institutions Contract Research Organizations (CROs) Others |
| By Delivery Method | Injectable Oral Transdermal Inhalation Others |
| By Region | Northeast Midwest South West |
| By Manufacturing Process | Solid-Phase Peptide Synthesis (SPPS) Liquid-Phase Peptide Synthesis (LPPS) Hybrid Synthesis Recombinant Technology Others |
| By Pricing Model | Cost-Plus Pricing Value-Based Pricing Competitive Pricing Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical R&D Departments | 100 | R&D Directors, Lead Scientists |
| CDMO Operations | 60 | Operations Managers, Production Supervisors |
| Regulatory Affairs | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Market Access and Pricing | 50 | Market Access Managers, Pricing Analysts |
| Quality Control in Manufacturing | 40 | Quality Control Managers, Lab Technicians |
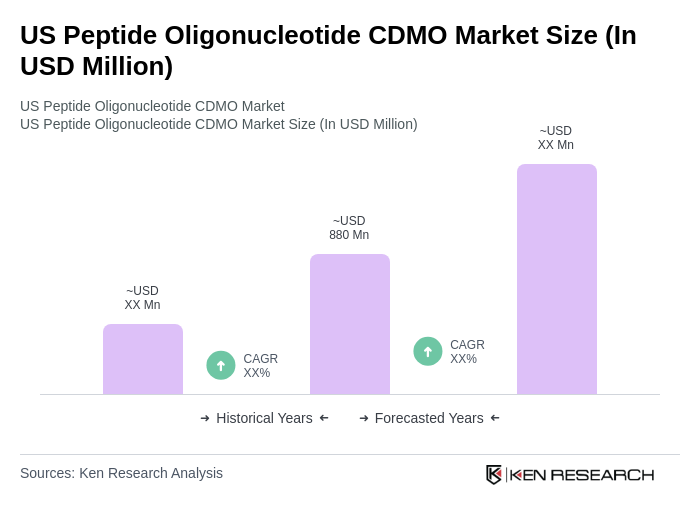
The US Peptide Oligonucleotide CDMO Market is valued at approximately USD 880 million, reflecting a robust growth trajectory driven by increasing demand for peptide and oligonucleotide-based therapeutics and advancements in drug development technologies.