Region:Asia
Author(s):Shubham
Product Code:KRAC2192
Pages:98
Published On:October 2025
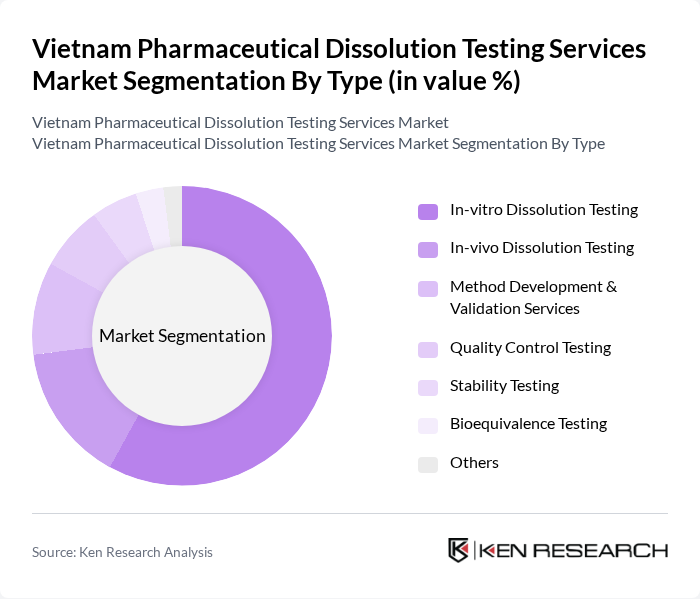
By Type:The market is segmented into various types of dissolution testing services, including In-vitro Dissolution Testing, In-vivo Dissolution Testing, Method Development & Validation Services, Quality Control Testing, Stability Testing, Bioequivalence Testing, and Others. Among these, In-vitro Dissolution Testing is the most prominent due to its critical role in drug formulation and development, allowing for the assessment of drug release profiles in a controlled environment. The dominance of in-vitro testing is reinforced by regulatory requirements and the need for rapid, ethical, and cost-effective analysis in both generic and innovative drug development .
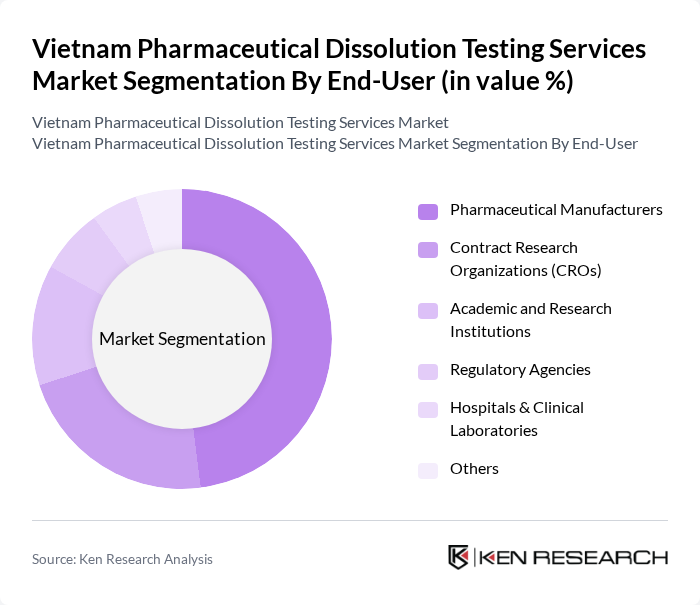
By End-User:The end-user segmentation includes Pharmaceutical Manufacturers, Contract Research Organizations (CROs), Academic and Research Institutions, Regulatory Agencies, Hospitals & Clinical Laboratories, and Others. Pharmaceutical Manufacturers are the leading end-users, as they require extensive dissolution testing to ensure product quality and compliance with regulatory standards throughout the drug development process. CROs are also significant contributors, driven by the outsourcing trend in pharmaceutical R&D and the need for specialized analytical services .
The Vietnam Pharmaceutical Dissolution Testing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as Eurofins Scientific, SGS Vietnam Ltd., Intertek Vietnam Ltd., Charles River Laboratories, Labcorp Drug Development, WuXi AppTec, Q2 Solutions, Medlab Vietnam, ALS Vietnam, Pharmedic Pharmaceutical Testing Center, Vina Control, Syngene International, ICON plc, PRA Health Sciences, Toxikon Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Vietnam pharmaceutical dissolution testing services market appears promising, driven by ongoing advancements in technology and regulatory frameworks. As local pharmaceutical companies expand their operations, the demand for reliable testing services will likely increase. Furthermore, the integration of artificial intelligence and machine learning into testing processes is expected to enhance efficiency and accuracy, positioning Vietnam as a competitive player in the global pharmaceutical landscape. Continuous investment in research and development will further bolster this growth trajectory.
| Segment | Sub-Segments |
|---|---|
| By Type | In-vitro Dissolution Testing In-vivo Dissolution Testing Method Development & Validation Services Quality Control Testing Stability Testing Bioequivalence Testing Others |
| By End-User | Pharmaceutical Manufacturers Contract Research Organizations (CROs) Academic and Research Institutions Regulatory Agencies Hospitals & Clinical Laboratories Others |
| By Application | Drug Development Quality Assurance & Quality Control Regulatory Compliance Research and Development Bioequivalence Studies Others |
| By Service Type | Routine Dissolution Testing Services Customized/Method Development Services Consultation & Regulatory Support Services Training & Education Services Others |
| By Region | Northern Vietnam Central Vietnam Southern Vietnam Others |
| By Pricing Model | Fixed Pricing Variable Pricing Subscription-Based Pricing Project-Based Pricing Others |
| By Customer Type | Large Enterprises Medium Enterprises Small Enterprises Startups Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 100 | Quality Control Managers, R&D Directors |
| Contract Testing Laboratories | 70 | Laboratory Directors, Testing Specialists |
| Regulatory Bodies | 50 | Regulatory Affairs Officers, Compliance Managers |
| Academic Institutions | 60 | Pharmaceutical Sciences Professors, Research Fellows |
| Industry Associations | 40 | Policy Analysts, Industry Advocates |
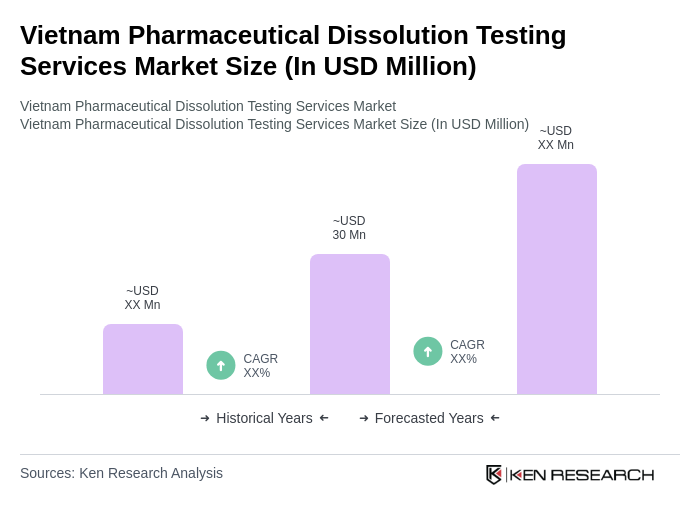
The Vietnam Pharmaceutical Dissolution Testing Services Market is valued at approximately USD 30 million, reflecting a significant growth driven by increased demand for quality assurance, regulatory compliance, and the expansion of drug development projects in the country.