Region:Middle East
Author(s):Dev
Product Code:KRAC8871
Pages:87
Published On:November 2025
By Type:The segmentation by type includes various methods of dissolution testing, which are crucial for assessing the release characteristics of pharmaceutical formulations. The subsegments are Manual Dissolution Testing, Semi-Automated Dissolution Testing, Fully Automated Dissolution Testing, In-vitro Dissolution Testing, In-vivo Dissolution Testing, and Others. Among these, Fully Automated Dissolution Testing is gaining traction due to its efficiency and accuracy, which are essential for meeting regulatory standards and expediting the drug development process. In-vitro Dissolution Testing remains the most widely adopted method globally, favored for its non-invasive nature and alignment with regulatory guidelines, while the Middle East market is rapidly adopting automation to improve throughput and compliance .

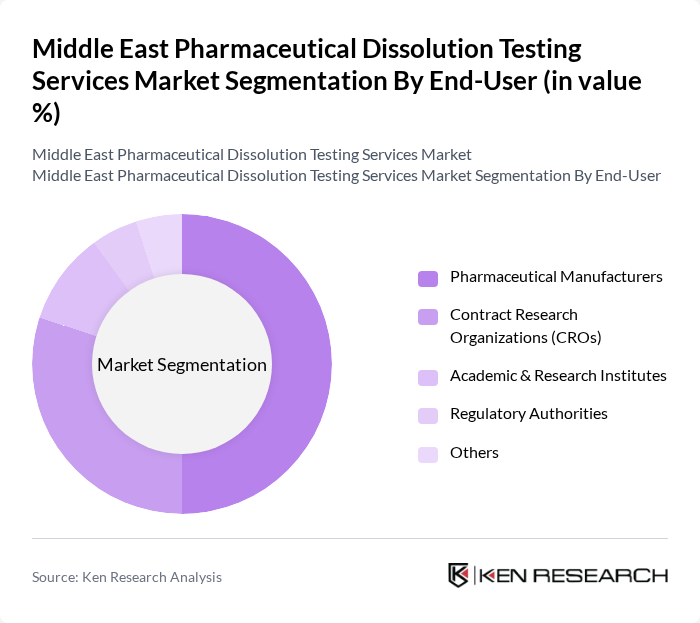
By End-User:The end-user segmentation includes Pharmaceutical Manufacturers, Contract Research Organizations (CROs), Academic & Research Institutes, Regulatory Authorities, and Others. Pharmaceutical Manufacturers are the leading end-users, driven by the need for compliance with stringent regulatory requirements and the increasing focus on drug quality and efficacy. CROs are also significant players, as they provide essential testing services to pharmaceutical companies, enhancing the overall market growth. The trend toward outsourcing analytical testing to CROs is accelerating, particularly among regional manufacturers seeking to streamline operations and meet evolving regulatory expectations .
The Middle East Pharmaceutical Dissolution Testing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as SGS SA, Eurofins Scientific, Intertek Group plc, Charles River Laboratories, Labcorp Drug Development, Q2 Solutions, BioAnalytical Research Corporation (BARC), Al Borg Medical Laboratories, Life Diagnostics, Pharmazone Analytical Services, Dubai Science Park Laboratory Services, Julphar (Gulf Pharmaceutical Industries), Tabuk Pharmaceuticals, Tamer Group, Synergy Pharma contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Middle East pharmaceutical dissolution testing services market appears promising, driven by technological advancements and increasing regulatory scrutiny. As the industry embraces automation and artificial intelligence, testing processes will become more efficient and accurate. Additionally, the ongoing expansion of pharmaceutical manufacturing facilities will create a greater need for reliable testing services. Companies that adapt to these trends will likely enhance their competitive edge and ensure compliance with evolving standards, positioning themselves for sustained growth in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Manual Dissolution Testing Semi-Automated Dissolution Testing Fully Automated Dissolution Testing In-vitro Dissolution Testing In-vivo Dissolution Testing Others |
| By End-User | Pharmaceutical Manufacturers Contract Research Organizations (CROs) Academic & Research Institutes Regulatory Authorities Others |
| By Dosage Form | Tablets Capsules Powders & Granules Liquids & Suspensions Others |
| By Testing Method | USP Method EP Method BP Method JP Method Others |
| By Country/Region | GCC Countries (Saudi Arabia, UAE, Qatar, Kuwait, Oman, Bahrain) Levant (Jordan, Lebanon, etc.) North Africa (Egypt, Morocco, etc.) Rest of Middle East |
| By Service Type | Analytical Testing Services Method Development & Validation Stability Testing Services Training & Consulting Services Others |
| By Customer Type | Large Enterprises Small & Medium Enterprises (SMEs) Startups Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 120 | Quality Control Managers, R&D Directors |
| Regulatory Bodies | 45 | Regulatory Affairs Officers, Compliance Managers |
| Contract Testing Laboratories | 60 | Laboratory Directors, Technical Managers |
| Healthcare Providers | 55 | Pharmacists, Clinical Researchers |
| Industry Associations | 40 | Policy Analysts, Industry Advocates |
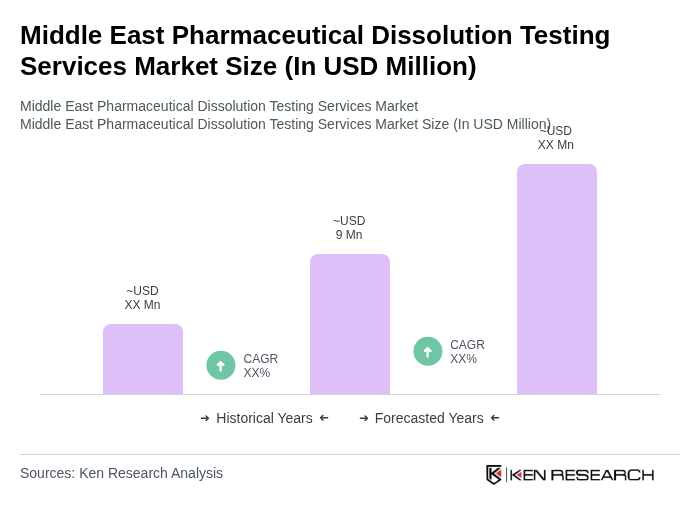
The Middle East Pharmaceutical Dissolution Testing Services Market is valued at approximately USD 9 million, based on a five-year historical analysis. This market is experiencing growth due to increasing demand for quality assurance and regulatory compliance in pharmaceutical products.