About the Report
Base Year 2024APAC Antinuclear Antibody Ana Test Market Overview
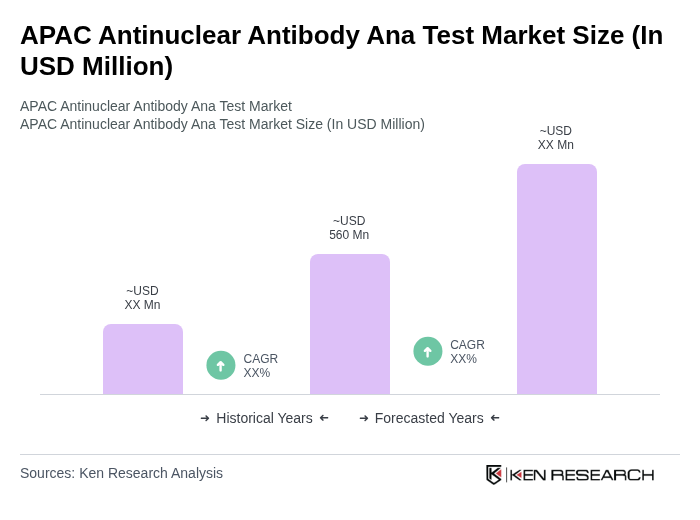
- The APAC Antinuclear Antibody Ana Test Market is valued at USD 560 million, based on a five-year historical analysis. This growth is primarily driven by the rising prevalence of autoimmune diseases such as systemic lupus erythematosus and rheumatoid arthritis, increasing awareness about early diagnosis, and rapid advancements in diagnostic technologies. The demand for ANA tests is further fueled by the expanding geriatric population, greater healthcare access, and the adoption of automated and AI-driven diagnostic platforms in the region .
- Key players in this market include China, Japan, and India, which dominate due to their large populations and increasing healthcare expenditure. China leads with its significant investments in healthcare infrastructure and the highest regional market share, while Japan benefits from advanced diagnostic technologies and a strong focus on early disease detection. India is witnessing rapid growth in demand for diagnostic tests, driven by rising healthcare awareness, increased healthcare spending, and a growing burden of autoimmune disorders .
- In 2023, the Indian government reinforced the National Health Mission (NHM), issued by the Ministry of Health and Family Welfare, Government of India, to enhance healthcare access and improve diagnostic services nationwide. The NHM includes operational guidelines for expanding laboratory infrastructure and increasing the availability of autoimmune disease testing, thereby supporting the routine use of antinuclear antibody tests in public healthcare facilities .
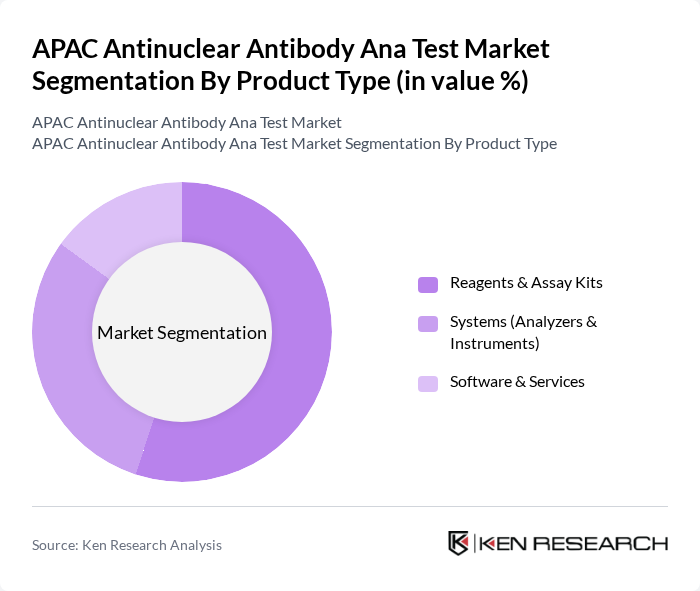
APAC Antinuclear Antibody Ana Test Market Segmentation
By Product Type:The product type segmentation includes Reagents & Assay Kits, Systems (Analyzers & Instruments), and Software & Services. Among these, Reagents & Assay Kits hold the largest market share due to their essential role in ANA testing workflows and their widespread use in both centralized and decentralized laboratory settings. The demand for accurate and reliable testing solutions continues to drive growth in this segment, while the Systems segment benefits from ongoing technological innovation, including automation and AI-enabled analyzers .
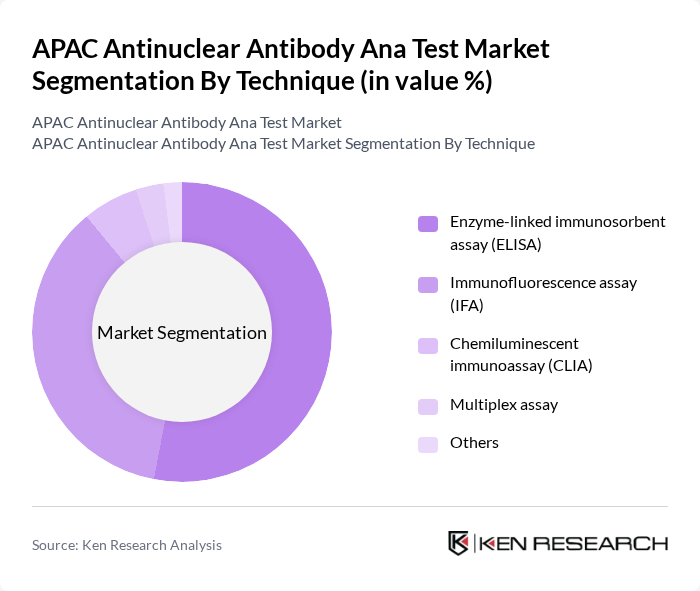
By Technique:The technique segmentation includes Enzyme-linked immunosorbent assay (ELISA), Immunofluorescence assay (IFA), Chemiluminescent immunoassay (CLIA), Multiplex assay, and Others. The ELISA technique is the most widely used in the APAC region, accounting for over half of the market share, due to its high sensitivity, cost-effectiveness, and suitability for large-scale screening. The IFA technique also holds a significant share, especially in specialized clinical laboratories, for its ability to detect a broad spectrum of autoantibodies .
APAC Antinuclear Antibody Ana Test Market Competitive Landscape
The APAC Antinuclear Antibody Ana Test Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Thermo Fisher Scientific, Bio-Rad Laboratories, Ortho Clinical Diagnostics, Fujirebio, EUROIMMUN AG, HYCOR Biomedical, Medac GmbH, Trinity Biotech plc, Inova Diagnostics, ERBA Diagnostics, Inc., Immuno Concepts, and Zeus Scientific contribute to innovation, geographic expansion, and service delivery in this space .
APAC Antinuclear Antibody Ana Test Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Autoimmune Diseases:The APAC region has witnessed a significant rise in autoimmune diseases, with an estimated 50 million people affected in future. This increase is attributed to factors such as environmental changes and genetic predispositions. The World Health Organization reported that autoimmune diseases are among the top ten leading causes of death in women aged 15-49, driving demand for antinuclear antibody tests as essential diagnostic tools in clinical settings.
- Rising Awareness and Diagnosis Rates:Public awareness campaigns and educational initiatives have led to a notable increase in the diagnosis of autoimmune diseases in APAC. In future, diagnostic rates are projected to rise by 30%, with healthcare providers emphasizing early detection. This heightened awareness is supported by government health initiatives, which allocated approximately $1.2 billion to improve diagnostic services, thereby boosting the demand for antinuclear antibody tests across the region.
- Technological Advancements in Testing Methods:The introduction of advanced testing technologies, such as multiplex assays and automated systems, has revolutionized the antinuclear antibody testing landscape. In future, the market is expected to see a 25% increase in the adoption of these technologies, driven by their ability to provide faster and more accurate results. This shift is supported by a $500 million investment in research and development by leading diagnostic companies in the APAC region.
Market Challenges
- High Cost of Advanced Testing Equipment:The initial investment required for advanced testing equipment poses a significant barrier to market growth. In future, the average cost of high-end diagnostic machines is projected to be around $150,000, limiting access for smaller laboratories and healthcare facilities. This financial constraint can hinder the widespread adoption of antinuclear antibody tests, particularly in developing regions within APAC, where budgets are tighter.
- Limited Access to Healthcare in Rural Areas:Access to healthcare services remains a critical challenge in rural areas of the APAC region. Approximately 60% of the population in these areas lacks adequate healthcare facilities, which directly impacts the availability of antinuclear antibody testing. In future, it is estimated that only 40% of rural patients will have access to necessary diagnostic services, creating disparities in disease detection and treatment outcomes.
APAC Antinuclear Antibody Ana Test Market Future Outlook
The future of the APAC antinuclear antibody test market appears promising, driven by ongoing advancements in diagnostic technologies and increasing healthcare investments. As healthcare infrastructure expands, particularly in underserved regions, access to testing will improve significantly. Additionally, the integration of artificial intelligence in diagnostics is expected to enhance accuracy and efficiency, further propelling market growth. The focus on preventive healthcare will also encourage early diagnosis, leading to better patient outcomes and increased demand for testing services.
Market Opportunities
- Expansion of Healthcare Infrastructure:The ongoing expansion of healthcare infrastructure in APAC presents a significant opportunity for the antinuclear antibody test market. With governments investing over $10 billion in healthcare facilities in future, access to diagnostic services will improve, facilitating increased testing and early disease detection.
- Development of Cost-Effective Testing Solutions:There is a growing demand for cost-effective testing solutions in the APAC region. By future, the market for affordable diagnostic tests is expected to grow by 20%, driven by innovations in testing technologies and materials. This trend will enable broader access to antinuclear antibody tests, particularly in low-income populations.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Reagents & Assay Kits Systems (Analyzers & Instruments) Software & Services |
| By Technique | Enzyme-linked immunosorbent assay (ELISA) Immunofluorescence assay (IFA) Chemiluminescent immunoassay (CLIA) Multiplex assay Others |
| By Antibody Type | Extractable Nuclear Antigens (ENA) dsDNA Histones Others |
| By Application | Systemic Lupus Erythematosus (SLE) Rheumatoid Arthritis Sjögren’s Syndrome Scleroderma Others |
| By End-User | Hospitals Clinical/Diagnostic Laboratories Research Institutions Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Country/Region | China Japan India South Korea Southeast Asia (Indonesia, Malaysia, Philippines, etc.) Australia & New Zealand Rest of APAC |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Therapeutic Goods Administration, Ministry of Health, Food and Drug Administration)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Laboratories
Biotechnology and Pharmaceutical Companies
Industry Associations (e.g., Asia Pacific Association of Allergy, Asthma and Clinical Immunology)
Financial Institutions
Players Mentioned in the Report:
Roche Diagnostics
Abbott Laboratories
Siemens Healthineers
Thermo Fisher Scientific
Bio-Rad Laboratories
Ortho Clinical Diagnostics
Fujirebio
EUROIMMUN AG
HYCOR Biomedical
Medac GmbH
Trinity Biotech plc
Inova Diagnostics
ERBA Diagnostics, Inc.
Immuno Concepts
Zeus Scientific
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. APAC Antinuclear Antibody Ana Test Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 APAC Antinuclear Antibody Ana Test Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. APAC Antinuclear Antibody Ana Test Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of autoimmune diseases
3.1.2 Rising awareness and diagnosis rates
3.1.3 Technological advancements in testing methods
3.1.4 Growing healthcare expenditure in APAC
3.2 Market Challenges
3.2.1 High cost of advanced testing equipment
3.2.2 Limited access to healthcare in rural areas
3.2.3 Regulatory hurdles in product approvals
3.2.4 Competition from alternative diagnostic methods
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Increasing demand for personalized medicine
3.3.3 Collaborations with research institutions
3.3.4 Development of cost-effective testing solutions
3.4 Market Trends
3.4.1 Shift towards point-of-care testing
3.4.2 Integration of AI in diagnostic processes
3.4.3 Focus on preventive healthcare
3.4.4 Rise in telemedicine and remote diagnostics
3.5 Government Regulation
3.5.1 Stricter quality control measures
3.5.2 Enhanced approval processes for diagnostics
3.5.3 Incentives for local manufacturing
3.5.4 Guidelines for clinical testing standards
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. APAC Antinuclear Antibody Ana Test Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. APAC Antinuclear Antibody Ana Test Market Segmentation
8.1 By Product Type
8.1.1 Reagents & Assay Kits
8.1.2 Systems (Analyzers & Instruments)
8.1.3 Software & Services
8.2 By Technique
8.2.1 Enzyme-linked immunosorbent assay (ELISA)
8.2.2 Immunofluorescence assay (IFA)
8.2.3 Chemiluminescent immunoassay (CLIA)
8.2.4 Multiplex assay
8.2.5 Others
8.3 By Antibody Type
8.3.1 Extractable Nuclear Antigens (ENA)
8.3.2 dsDNA
8.3.3 Histones
8.3.4 Others
8.4 By Application
8.4.1 Systemic Lupus Erythematosus (SLE)
8.4.2 Rheumatoid Arthritis
8.4.3 Sjögren’s Syndrome
8.4.4 Scleroderma
8.4.5 Others
8.5 By End-User
8.5.1 Hospitals
8.5.2 Clinical/Diagnostic Laboratories
8.5.3 Research Institutions
8.5.4 Others
8.6 By Distribution Channel
8.6.1 Direct Sales
8.6.2 Distributors
8.6.3 Online Sales
8.6.4 Others
8.7 By Country/Region
8.7.1 China
8.7.2 Japan
8.7.3 India
8.7.4 South Korea
8.7.5 Southeast Asia (Indonesia, Malaysia, Philippines, etc.)
8.7.6 Australia & New Zealand
8.7.7 Rest of APAC
9. APAC Antinuclear Antibody Ana Test Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Revenue (USD Million)
9.2.2 Market Share in APAC ANA Test Segment (%)
9.2.3 CAGR (2019-2024, 2025-2032)
9.2.4 Number of ANA Test Products/Assays Approved in APAC
9.2.5 Penetration in Key APAC Countries (China, Japan, India, etc.)
9.2.6 R&D Investment as % of Revenue
9.2.7 Product Portfolio Breadth (Number of ANA-related SKUs)
9.2.8 Distribution Network Coverage (Number of APAC countries served)
9.2.9 Regulatory Approval Track Record (e.g., NMPA, PMDA, CDSCO)
9.2.10 Strategic Partnerships/Collaborations in APAC
9.2.11 Customer Satisfaction/Net Promoter Score (NPS)
9.2.12 Local Manufacturing/Localization Initiatives
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Roche Diagnostics
9.5.2 Abbott Laboratories
9.5.3 Siemens Healthineers
9.5.4 Thermo Fisher Scientific
9.5.5 Bio-Rad Laboratories
9.5.6 Ortho Clinical Diagnostics
9.5.7 Fujirebio
9.5.8 EUROIMMUN AG
9.5.9 HYCOR Biomedical
9.5.10 Medac GmbH
9.5.11 Trinity Biotech plc
9.5.12 Inova Diagnostics
9.5.13 ERBA Diagnostics, Inc.
9.5.14 Immuno Concepts
9.5.15 Zeus Scientific
10. APAC Antinuclear Antibody Ana Test Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget allocation trends
10.1.2 Decision-making processes
10.1.3 Preferred suppliers
10.1.4 Compliance requirements
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment trends in healthcare
10.2.2 Funding sources
10.2.3 Infrastructure development plans
10.2.4 Partnerships with private sector
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to quality diagnostics
10.3.2 Cost of testing
10.3.3 Timeliness of results
10.3.4 Availability of trained personnel
10.4 User Readiness for Adoption
10.4.1 Training and support needs
10.4.2 Technology acceptance levels
10.4.3 Infrastructure readiness
10.4.4 Financial readiness
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of success metrics
10.5.2 Case studies of successful implementations
10.5.3 Opportunities for scaling
10.5.4 Feedback mechanisms for improvement
11. APAC Antinuclear Antibody Ana Test Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation analysis
1.5 Competitive landscape overview
1.6 Key partnerships identification
1.7 Risk assessment
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience definition
2.4 Communication strategies
2.5 Digital marketing tactics
2.6 Event marketing plans
2.7 Performance metrics
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 Online distribution channels
3.4 Direct sales approaches
3.5 Partnership with healthcare providers
3.6 Logistics and supply chain management
3.7 Performance evaluation
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing strategies
4.4 Customer willingness to pay
4.5 Value-based pricing models
4.6 Discount strategies
4.7 Pricing optimization techniques
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
5.3 Product development opportunities
5.4 Market entry strategies
5.5 Customer feedback incorporation
5.6 Future trends forecasting
5.7 Competitive advantage development
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer engagement strategies
6.4 Feedback collection methods
6.5 Relationship management tools
6.6 Retention strategies
6.7 Performance tracking
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Customer-centric approaches
7.4 Innovation in product offerings
7.5 Cost-effectiveness
7.6 Quality assurance
7.7 Market differentiation
8. Key Activities
8.1 Regulatory compliance
8.2 Branding efforts
8.3 Distribution setup
8.4 Training and development
8.5 Marketing campaigns
8.6 Performance monitoring
8.7 Stakeholder engagement
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band analysis
9.1.3 Packaging strategies
9.2 Export Entry Strategy
9.2.1 Target countries selection
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
10.5 Risk assessment
10.6 Market entry feasibility
10.7 Strategic partnerships
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines for market entry
11.3 Budget allocation
11.4 Financial projections
11.5 Funding sources
11.6 Cost management strategies
11.7 Performance metrics
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnerships evaluation
12.3 Risk management strategies
12.4 Control mechanisms
12.5 Long-term sustainability
12.6 Exit strategies
12.7 Performance tracking
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
13.3 Revenue projections
13.4 Cost structure analysis
13.5 Profit margin assessment
13.6 Financial health indicators
13.7 Market share growth
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition targets
14.4 Strategic alliances
14.5 Research collaborations
14.6 Supplier partnerships
14.7 Industry associations
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Timeline for key activities</
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published market reports and white papers on antinuclear antibody tests
- Review of scientific literature and clinical studies related to autoimmune diseases
- Examination of regulatory frameworks and guidelines from health authorities in APAC countries
Primary Research
- Interviews with laboratory directors and pathologists specializing in autoimmune diagnostics
- Surveys with healthcare professionals, including rheumatologists and immunologists
- Focus groups with patients diagnosed with autoimmune disorders to understand testing experiences
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and secondary data sources
- Triangulation of market data from healthcare providers, laboratories, and industry reports
- Sanity checks through peer reviews and expert panel discussions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on healthcare expenditure in APAC
- Segmentation of the market by country, type of test, and end-user demographics
- Incorporation of trends in autoimmune disease prevalence and diagnostic advancements
Bottom-up Modeling
- Collection of sales data from leading diagnostic laboratories and manufacturers
- Estimation of test volumes based on patient demographics and disease incidence rates
- Cost analysis of antinuclear antibody tests to determine pricing strategies
Forecasting & Scenario Analysis
- Multi-variable forecasting using historical growth rates and market drivers
- Scenario analysis based on potential changes in healthcare policies and technology adoption
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Diagnostic Laboratories | 80 | Laboratory Managers, Clinical Pathologists |
| Healthcare Providers | 60 | Rheumatologists, General Practitioners |
| Patient Advocacy Groups | 40 | Patient Representatives, Community Health Workers |
| Medical Device Manufacturers | 40 | Product Managers, Regulatory Affairs Specialists |
| Health Insurance Companies | 40 | Policy Analysts, Claims Managers |
Frequently Asked Questions
What is the current value of the APAC Antinuclear Antibody Ana Test Market?
The APAC Antinuclear Antibody Ana Test Market is valued at approximately USD 560 million, reflecting a significant growth driven by the rising prevalence of autoimmune diseases and advancements in diagnostic technologies.