About the Report
Base Year 2024APAC Biomarker Based Immunoassays Market Overview
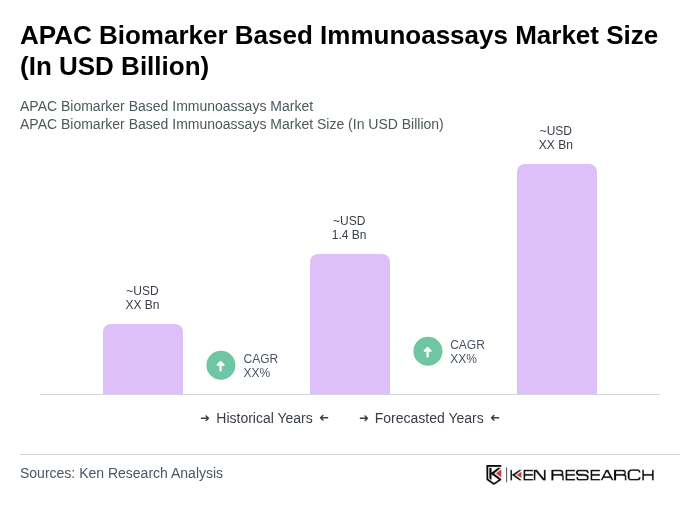
- The APAC Biomarker Based Immunoassays Market is valued at USD 1.4 billion, based on a five-year historical analysis. This growth is primarily driven by the rising prevalence of chronic and infectious diseases, rapid healthcare modernization, and the adoption of advanced diagnostic technologies. The market is further supported by increased investments in healthcare infrastructure, expansion of diagnostic laboratories, and a surge in research collaborations across the region .
- Key players in this market include China, Japan, and India, which dominate due to their large populations, significant healthcare expenditures, and robust pharmaceutical and biotechnology sectors. These countries have established strong research and development capabilities, leading to innovations in biomarker-based diagnostics and immunoassays, and further strengthening their market positions .
- The National Health Policy, 2017 issued by the Ministry of Health and Family Welfare, Government of India, emphasizes early disease detection and management through advanced diagnostic tools, including biomarker-based immunoassays. The policy mandates the strengthening of laboratory infrastructure, integration of new diagnostic technologies, and improved accessibility to advanced diagnostics at primary and secondary healthcare levels, thereby supporting market growth and improved healthcare outcomes in the region .
APAC Biomarker Based Immunoassays Market Segmentation
By Type:The market is segmented into various types of immunoassays, including Enzyme-Linked Immunosorbent Assay (ELISA), Lateral Flow Assays, Radioimmunoassays, Chemiluminescent Immunoassays, Multiplex Immunoassays, and Others. Each of these sub-segments plays a crucial role in the overall market dynamics.
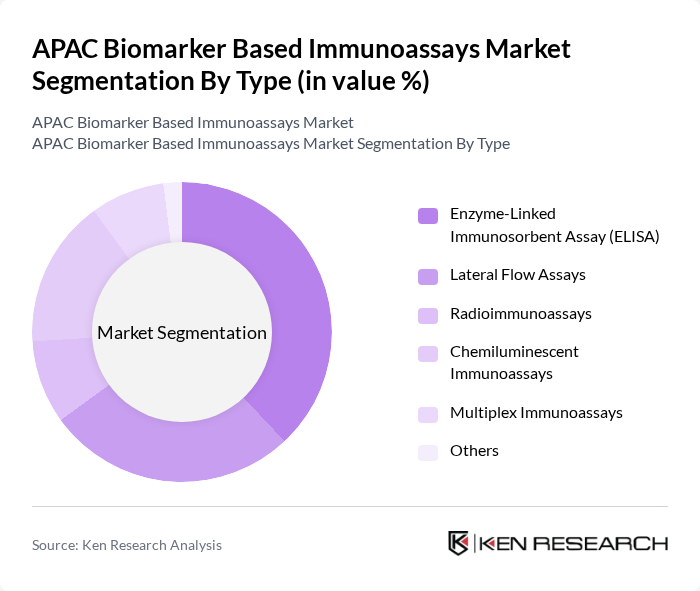
The Enzyme-Linked Immunosorbent Assay (ELISA) sub-segment remains the dominant player in the market due to its broad application in infectious disease diagnostics, oncology, and hormone level assessments. Its high sensitivity, specificity, and cost-effectiveness make it a preferred choice among healthcare professionals. The increasing adoption of ELISA in both clinical and research laboratories, along with growing demand for rapid and accurate diagnostic solutions, continues to drive the growth of this sub-segment .
By End-User:The market is segmented based on end-users, including Hospitals, Diagnostic Laboratories, Research Institutions, Pharmaceutical & Biotechnology Companies, Academic & Medical Centers, and Others. Each end-user category has distinct requirements and contributes differently to the market.
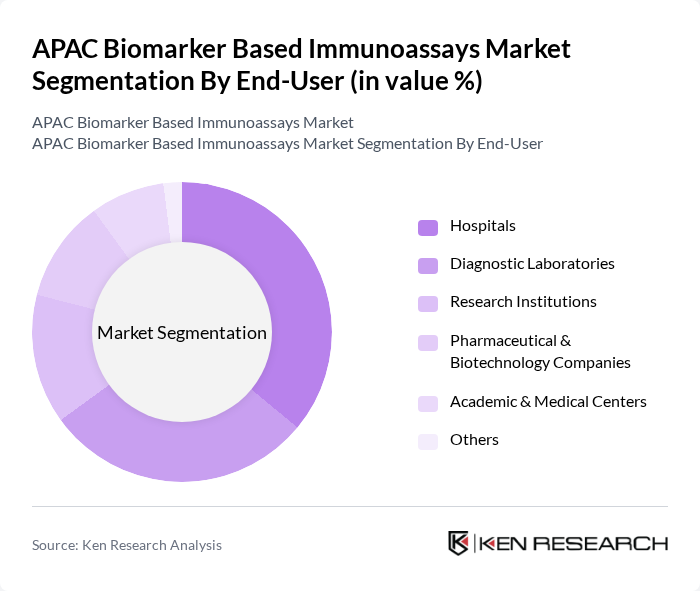
Hospitals are the leading end-user segment, primarily due to the high volume of diagnostic tests performed for a wide range of diseases. The focus on early diagnosis, patient-centric care, and the integration of advanced diagnostic technologies in hospitals drives the demand for biomarker-based immunoassays. Hospitals are also equipped with skilled professionals and modern laboratory infrastructure, enabling effective utilization of these assays. The trend towards personalized medicine further enhances the role of hospitals in this market .
APAC Biomarker Based Immunoassays Market Competitive Landscape
The APAC Biomarker Based Immunoassays Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Thermo Fisher Scientific, Bio-Rad Laboratories, Ortho Clinical Diagnostics, PerkinElmer, QIAGEN, Agilent Technologies, Merck KGaA, Becton, Dickinson and Company, Sysmex Corporation, Fujirebio, Mindray Bio-Medical Electronics Co., Ltd., Tosoh Corporation, Shenzhen New Industries Biomedical Engineering Co., Ltd. (Snibe), BioMérieux, Wondfo Biotech Co., Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
APAC Biomarker Based Immunoassays Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The APAC region is witnessing a significant rise in chronic diseases, with the World Health Organization reporting that non-communicable diseases accounted for approximately 60% of all deaths in the region. This trend is expected to continue, with a substantial and growing patient population driving the demand for biomarker-based immunoassays, as they are essential for early diagnosis and effective management of these diseases.
- Advancements in Biomarker Discovery Technologies:The APAC region is experiencing rapid advancements in biomarker discovery technologies, with investments in research and development estimated to be in the multi-billion USD range.These innovations enhance the sensitivity and specificity of immunoassays, enabling the identification of novel biomarkers. As a result, healthcare providers are increasingly adopting these technologies, leading to improved diagnostic capabilities and better patient outcomes, which further fuels market growth.
- Rising Demand for Personalized Medicine:The shift towards personalized medicine is gaining momentum in the APAC region, with the market for personalized healthcare recognized as a major growth area.This trend is driven by the increasing recognition of the need for tailored treatment approaches based on individual patient profiles. Biomarker-based immunoassays play a crucial role in this paradigm shift, as they facilitate the identification of specific biomarkers that guide personalized treatment decisions, thereby enhancing therapeutic efficacy.
Market Challenges
- High Costs Associated with Biomarker Development:The development of biomarkers is often a costly endeavor, with estimates suggesting that the average cost of bringing a new biomarker to market can be substantial, potentially reaching several hundred million USD depending on the complexity and regulatory requirements.This financial burden poses a significant challenge for companies operating in the APAC biomarker-based immunoassays market, as it limits the number of players who can afford to invest in research and development, potentially stifling innovation and market growth.
- Regulatory Hurdles in Product Approval:The regulatory landscape for biomarker-based immunoassays in the APAC region is complex and varies significantly across countries. For instance, the approval process can take several years in some jurisdictions, leading to delays in market entry.These regulatory hurdles can deter investment and slow down the introduction of innovative products, ultimately impacting the growth potential of the biomarker-based immunoassays market in the region.
APAC Biomarker Based Immunoassays Market Future Outlook
The future of the APAC biomarker-based immunoassays market appears promising, driven by technological advancements and increasing healthcare investments. As the region continues to prioritize early disease detection and personalized medicine, the integration of artificial intelligence in biomarker analysis is expected to enhance diagnostic accuracy. Furthermore, the expansion of healthcare infrastructure will facilitate greater access to immunoassays, ultimately improving patient outcomes and driving market growth in the coming years.
Market Opportunities
- Expansion of Healthcare Infrastructure in APAC:The ongoing expansion of healthcare infrastructure in APAC countries is supported by significant investments, with major economies in the region allocating substantial resources to healthcare development.Improved facilities and access to advanced diagnostic tools will enhance the adoption of these technologies, ultimately benefiting patient care and driving market growth.
- Collaborations with Research Institutions:Collaborations between biotechnology firms and research institutions are on the rise, facilitating knowledge sharing and resource pooling, which accelerates the development of innovative biomarker-based immunoassays.Such partnerships are crucial for advancing research and enhancing the market's competitive landscape, creating new opportunities for growth.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Enzyme-Linked Immunosorbent Assay (ELISA) Lateral Flow Assays Radioimmunoassays Chemiluminescent Immunoassays Multiplex Immunoassays Others |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Pharmaceutical & Biotechnology Companies Academic & Medical Centers Others |
| By Application | Cancer Diagnostics Infectious Disease Testing Autoimmune Disease Testing Cardiovascular Disease Testing Neurological Disease Testing Others |
| By Technology | Microarray Technology Mass Spectrometry Next-Generation Sequencing (NGS) Polymerase Chain Reaction (PCR) Others |
| By Sample Type | Blood Samples Urine Samples Tissue Samples Saliva Samples Cerebrospinal Fluid Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | China Japan India South Korea Australia & New Zealand Southeast Asia Rest of APAC |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Therapeutic Goods Administration, Ministry of Health, National Medical Products Administration)
Biotechnology and Pharmaceutical Companies
Clinical Laboratories and Diagnostic Centers
Healthcare Providers and Hospitals
Medical Device Manufacturers
Biomarker Research Organizations
Health Insurance Companies
Players Mentioned in the Report:
Roche Diagnostics
Abbott Laboratories
Siemens Healthineers
Thermo Fisher Scientific
Bio-Rad Laboratories
Ortho Clinical Diagnostics
PerkinElmer
QIAGEN
Agilent Technologies
Merck KGaA
Becton, Dickinson and Company
Sysmex Corporation
Fujirebio
Mindray Bio-Medical Electronics Co., Ltd.
Tosoh Corporation
Shenzhen New Industries Biomedical Engineering Co., Ltd. (Snibe)
BioMerieux
Wondfo Biotech Co., Ltd.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. APAC Biomarker Based Immunoassays Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 APAC Biomarker Based Immunoassays Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. APAC Biomarker Based Immunoassays Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic diseases
3.1.2 Advancements in biomarker discovery technologies
3.1.3 Rising demand for personalized medicine
3.1.4 Growing investments in R&D for immunoassays
3.2 Market Challenges
3.2.1 High costs associated with biomarker development
3.2.2 Regulatory hurdles in product approval
3.2.3 Limited awareness among healthcare professionals
3.2.4 Competition from alternative diagnostic methods
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure in APAC
3.3.2 Collaborations with research institutions
3.3.3 Increasing focus on early disease detection
3.3.4 Development of cost-effective immunoassays
3.4 Market Trends
3.4.1 Integration of AI in biomarker analysis
3.4.2 Shift towards point-of-care testing
3.4.3 Growing emphasis on companion diagnostics
3.4.4 Rise in consumer awareness regarding health monitoring
3.5 Government Regulation
3.5.1 Stricter guidelines for clinical validation
3.5.2 Incentives for R&D in biomarker technologies
3.5.3 Policies promoting personalized medicine
3.5.4 Regulations on data privacy in health diagnostics
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. APAC Biomarker Based Immunoassays Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. APAC Biomarker Based Immunoassays Market Segmentation
8.1 By Type
8.1.1 Enzyme-Linked Immunosorbent Assay (ELISA)
8.1.2 Lateral Flow Assays
8.1.3 Radioimmunoassays
8.1.4 Chemiluminescent Immunoassays
8.1.5 Multiplex Immunoassays
8.1.6 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Diagnostic Laboratories
8.2.3 Research Institutions
8.2.4 Pharmaceutical & Biotechnology Companies
8.2.5 Academic & Medical Centers
8.2.6 Others
8.3 By Application
8.3.1 Cancer Diagnostics
8.3.2 Infectious Disease Testing
8.3.3 Autoimmune Disease Testing
8.3.4 Cardiovascular Disease Testing
8.3.5 Neurological Disease Testing
8.3.6 Others
8.4 By Technology
8.4.1 Microarray Technology
8.4.2 Mass Spectrometry
8.4.3 Next-Generation Sequencing (NGS)
8.4.4 Polymerase Chain Reaction (PCR)
8.4.5 Others
8.5 By Sample Type
8.5.1 Blood Samples
8.5.2 Urine Samples
8.5.3 Tissue Samples
8.5.4 Saliva Samples
8.5.5 Cerebrospinal Fluid
8.5.6 Others
8.6 By Distribution Channel
8.6.1 Direct Sales
8.6.2 Distributors
8.6.3 Online Sales
8.6.4 Others
8.7 By Region
8.7.1 China
8.7.2 Japan
8.7.3 India
8.7.4 South Korea
8.7.5 Australia & New Zealand
8.7.6 Southeast Asia
8.7.7 Rest of APAC
9. APAC Biomarker Based Immunoassays Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Regional Revenue (APAC)
9.2.4 Revenue Growth Rate (APAC)
9.2.5 Market Share (APAC Biomarker Immunoassays)
9.2.6 R&D Expenditure (% of Revenue)
9.2.7 Product Portfolio Breadth (Number of Biomarker Immunoassay Products)
9.2.8 Regulatory Approvals (Number of APAC-specific Approvals)
9.2.9 Distribution Network Strength (Number of APAC Countries Covered)
9.2.10 Strategic Collaborations/Partnerships in APAC
9.2.11 Customer Base (Number of Hospitals/Labs Served in APAC)
9.2.12 Manufacturing Footprint in APAC
9.2.13 Sustainability/ESG Initiatives (Relevant to APAC Operations)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Roche Diagnostics
9.5.2 Abbott Laboratories
9.5.3 Siemens Healthineers
9.5.4 Thermo Fisher Scientific
9.5.5 Bio-Rad Laboratories
9.5.6 Ortho Clinical Diagnostics
9.5.7 PerkinElmer
9.5.8 QIAGEN
9.5.9 Agilent Technologies
9.5.10 Merck KGaA
9.5.11 Becton, Dickinson and Company
9.5.12 Sysmex Corporation
9.5.13 Fujirebio
9.5.14 Mindray Bio-Medical Electronics Co., Ltd.
9.5.15 Tosoh Corporation
9.5.16 Shenzhen New Industries Biomedical Engineering Co., Ltd. (Snibe)
9.5.17 BioMérieux
9.5.18 Wondfo Biotech Co., Ltd.
10. APAC Biomarker Based Immunoassays Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Compliance Requirements
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends in Healthcare
10.2.2 Funding Sources
10.2.3 Infrastructure Development Plans
10.2.4 Energy Efficiency Initiatives
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges in Diagnostic Accuracy
10.3.2 Supply Chain Disruptions
10.3.3 Cost Management Issues
10.3.4 Technology Integration Difficulties
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Technology Acceptance Levels
10.4.3 Infrastructure Readiness
10.4.4 User Feedback Mechanisms
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Success Metrics
10.5.2 Case Studies of Successful Implementations
10.5.3 Future Use Case Opportunities
10.5.4 Long-term Value Assessment
11. APAC Biomarker Based Immunoassays Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from industry associations such as the Asia Pacific Biotech Association
- Review of scientific literature and publications on biomarker-based immunoassays from journals like Nature and Clinical Chemistry
- Examination of regulatory frameworks and guidelines from health authorities across APAC countries
Primary Research
- Interviews with key opinion leaders in the field of immunology and biomarker research
- Surveys targeting laboratory managers and clinical researchers in hospitals and diagnostic centers
- Focus group discussions with product development teams from leading biotech firms
Validation & Triangulation
- Cross-validation of findings through multiple data sources including market reports and expert interviews
- Triangulation of quantitative data with qualitative insights from industry experts
- Sanity checks through peer reviews and feedback from advisory panels
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on overall healthcare expenditure in APAC countries
- Segmentation of the market by application areas such as oncology, infectious diseases, and autoimmune disorders
- Incorporation of growth rates from related sectors such as diagnostics and personalized medicine
Bottom-up Modeling
- Collection of sales data from leading manufacturers of immunoassay kits and reagents
- Estimation of market share based on product types and geographical distribution
- Volume and pricing analysis based on historical sales trends and projected growth
Forecasting & Scenario Analysis
- Development of forecasting models using historical data and market trends
- Scenario analysis based on potential regulatory changes and technological advancements
- Creation of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Biomarker Testing | 120 | Clinical Oncologists, Pathologists |
| Infectious Disease Immunoassays | 95 | Infectious Disease Specialists, Laboratory Technicians |
| Autoimmune Disorder Diagnostics | 75 | Rheumatologists, Immunologists |
| Research and Development in Biotech | 65 | Biotech Researchers, Product Managers |
| Regulatory Affairs in Diagnostics | 55 | Regulatory Affairs Specialists, Compliance Officers |
Frequently Asked Questions
What is the current value of the APAC Biomarker Based Immunoassays Market?
The APAC Biomarker Based Immunoassays Market is valued at approximately USD 1.4 billion, driven by the increasing prevalence of chronic diseases, advancements in diagnostic technologies, and significant investments in healthcare infrastructure across the region.