Region:Asia
Author(s):Dev
Product Code:KRAB1865
Pages:96
Published On:January 2026
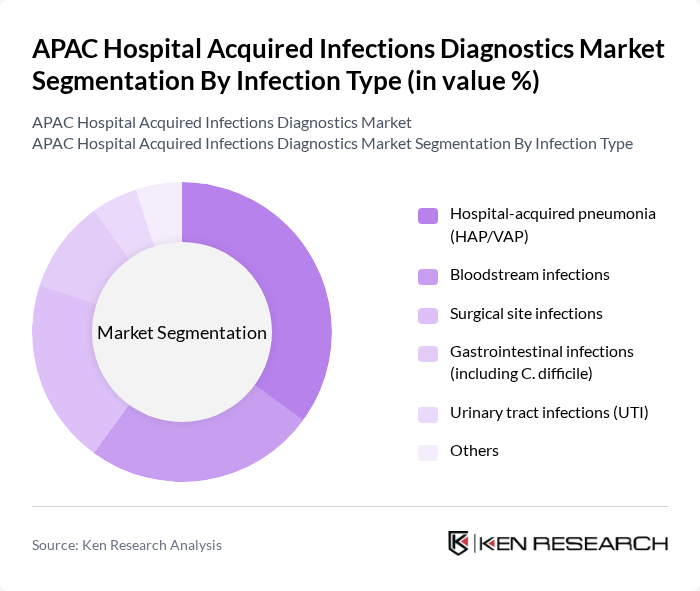
By Infection Type:The infection type segmentation includes various categories of hospital-acquired infections, which are critical for understanding the specific diagnostic needs within healthcare settings. The subsegments include Hospital-acquired pneumonia (HAP/VAP), Bloodstream infections, Surgical site infections, Gastrointestinal infections (including C. difficile), Urinary tract infections (UTI), and Others. Among these, Hospital-acquired pneumonia (HAP/VAP) is the leading subsegment due to its high incidence rates in intensive care units and the complexity of diagnosis and treatment. The increasing number of ventilated patients in intensive care units (ICUs), coupled with longer lengths of stay and high use of invasive devices, contributes significantly to the prevalence of HAP/VAP, driving demand for effective culture-based and molecular respiratory diagnostic solutions.
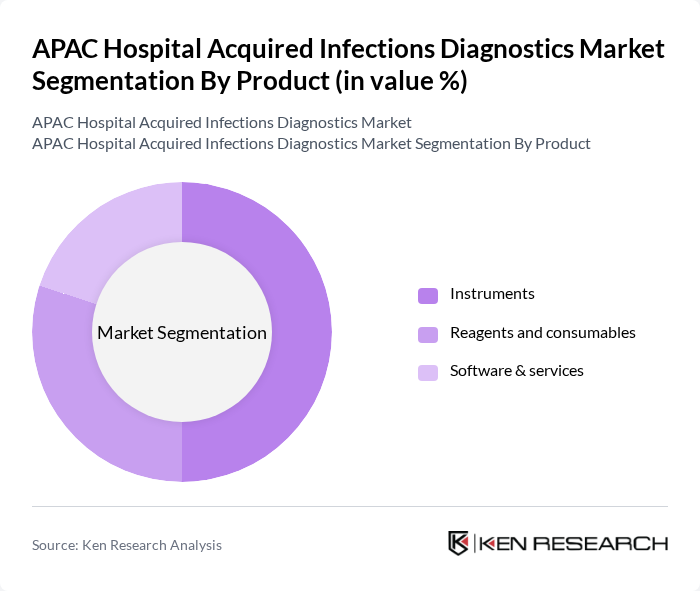
By Product:The product segmentation encompasses various diagnostic tools and solutions used in the detection of hospital-acquired infections. This includes Instruments, Reagents and consumables, and Software & services. The Reagents and consumables subsegment typically accounts for the largest share of global HAI diagnostics revenue due to the recurring nature of test kits, culture media, and assay cartridges used across high testing volumes, while Instruments and analyzers form a substantial installed base in hospitals and reference laboratories. In the APAC market, ongoing investments in automated microbiology systems, molecular platforms, and point-of-care analyzers, combined with growing integration of software and connectivity solutions, are strengthening the role of instruments and software & services in enabling faster, more reliable infection detection and reporting.
The APAC Hospital Acquired Infections Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Becton, Dickinson and Company, bioMérieux, Thermo Fisher Scientific, Cepheid, Hologic, Inc., QIAGEN N.V., QuidelOrtho Corporation, Danaher Corporation (Beckman Coulter, Cepheid), Grifols, S.A., Eiken Chemical Co., Ltd., Sysmex Corporation, and local/regional APAC players (e.g., Shenzhen Mindray Bio-Medical Electronics) contribute to innovation, geographic expansion, and service delivery in this space.
The APAC hospital-acquired infections diagnostics market is poised for significant transformation, driven by technological advancements and increased healthcare investments. The integration of artificial intelligence and machine learning into diagnostic processes is expected to enhance accuracy and efficiency. Additionally, the growing emphasis on preventive healthcare measures will likely lead to increased demand for rapid diagnostic tests, enabling timely interventions. As healthcare systems evolve, the focus on personalized medicine will further shape the landscape, fostering innovation and improved patient outcomes.
| Segment | Sub-Segments |
|---|---|
| By Infection Type | Hospital-acquired pneumonia (HAP/VAP) Bloodstream infections Surgical site infections Gastrointestinal infections (including C. difficile) Urinary tract infections (UTI) Others |
| By Product | Instruments Reagents and consumables Software & services |
| By Test Type / Technology | Molecular diagnostics (PCR and others) Immunoassays (including ELISA) Urinalysis Microbiology culture and susceptibility testing Other technologies |
| By Application | Disease testing Drug resistance testing (AMR) Surveillance & screening |
| By End User | Hospitals (including ICUs) Clinics & ambulatory surgery centers (ASC) Diagnostic laboratories Others |
| By Country (APAC) | China Japan India South Korea Australia & New Zealand Southeast Asia Rest of APAC |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Infection Control Departments | 120 | Infection Control Officers, Hospital Administrators |
| Diagnostic Equipment Manufacturers | 90 | Product Managers, Sales Directors |
| Clinical Laboratories | 80 | Laboratory Managers, Microbiologists |
| Healthcare Policy Analysts | 60 | Health Economists, Policy Advisors |
| Public Health Officials | 70 | Epidemiologists, Public Health Directors |
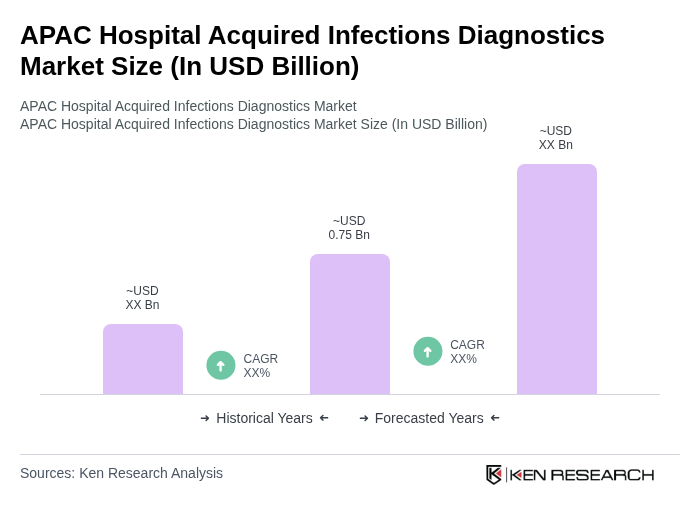
The APAC Hospital Acquired Infections Diagnostics Market is valued at approximately USD 0.75 billion, reflecting the region's significant share in the global market for hospital-acquired infections diagnostics.