About the Report
Base Year 2024APAC Pharmaceutical Fine Chemicals Market Overview
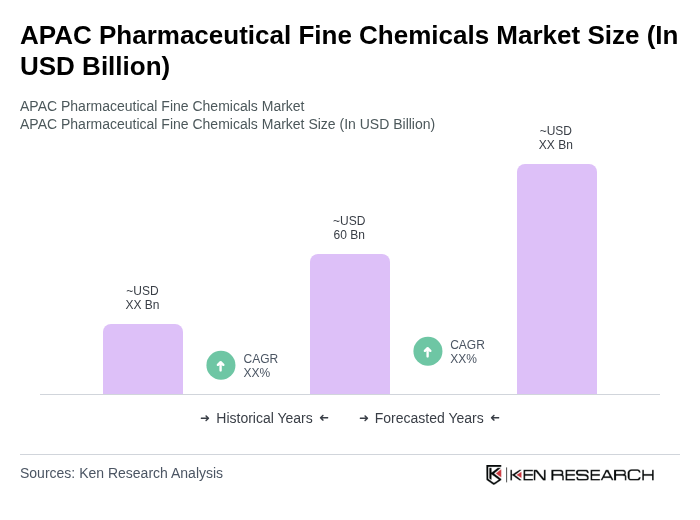
- The APAC Pharmaceutical Fine Chemicals Market is valued at approximately USD 60 billion, reflecting the region's dominant position in global pharmaceutical fine chemical production. This growth is primarily driven by the increasing demand for high-quality pharmaceuticals, advancements in drug development technologies, and the rising prevalence of chronic diseases across the region. The market is also supported by the growing trend of outsourcing pharmaceutical manufacturing to specialized companies, enhancing efficiency and reducing costs. Asia-Pacific accounts for more than 47% of global fine chemicals production, with over 44,000 metric tons produced, underscoring the region's critical role in the global pharmaceutical supply chain.
- China and India are the dominant countries in the APAC Pharmaceutical Fine Chemicals Market due to their robust manufacturing capabilities, large pool of skilled labor, and favorable government policies promoting pharmaceutical production. China leads the region with approximately 24,300 metric tons of production, focusing on both pharmaceutical APIs and intermediates, while India follows with around 13,200 metric tons, supported by strong exports and API production for over 120 countries. Additionally, Japan and South Korea are significant players, driven by their advanced technological infrastructure and strong research and development initiatives in the pharmaceutical sector. Japan contributed approximately 4,100 metric tons of fine chemicals production, mainly in electronic-grade fine chemicals and specialty materials.
- The Indian government's Production-Linked Incentive (PLI) scheme continues to boost domestic manufacturing of pharmaceutical ingredients. This initiative includes financial incentives for companies that increase their production capacity, thereby enhancing the country's self-sufficiency in pharmaceutical fine chemicals and reducing dependency on imports. The region witnessed significant capacity expansion, with over 300 new reactors installed across 65 sites, reflecting strong investment momentum in pharmaceutical fine chemical infrastructure.
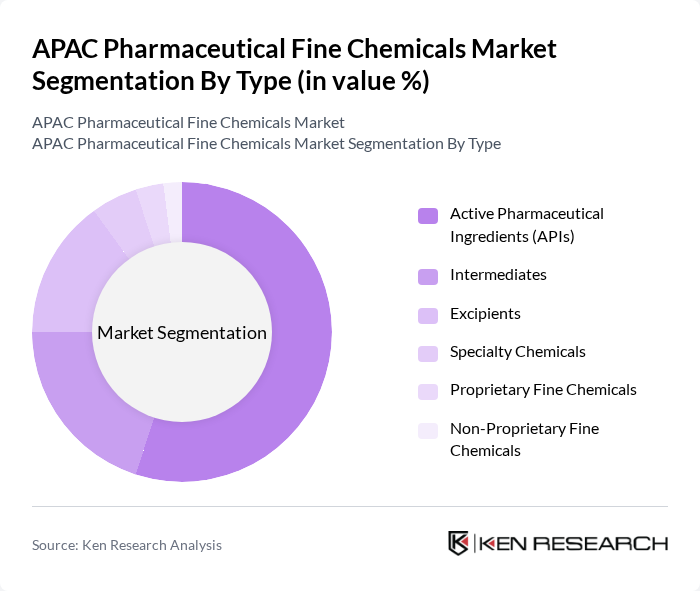
APAC Pharmaceutical Fine Chemicals Market Segmentation
By Type:The market is segmented into various types, including Active Pharmaceutical Ingredients (APIs), Intermediates, Excipients, Specialty Chemicals, Proprietary Fine Chemicals, and Non-Proprietary Fine Chemicals. Among these, Active Pharmaceutical Ingredients (APIs) dominate the market, accounting for approximately 55% of the pharmaceutical fine chemicals segment, due to their critical role in drug formulation and the increasing demand for generic and branded medications. The rise in chronic diseases and the need for innovative therapies further drive the demand for APIs, making them a key focus for manufacturers. Pharmaceutical-grade fine chemicals accounted for over 58,000 metric tons globally, driven by stringent purity requirements exceeding 99.9%.
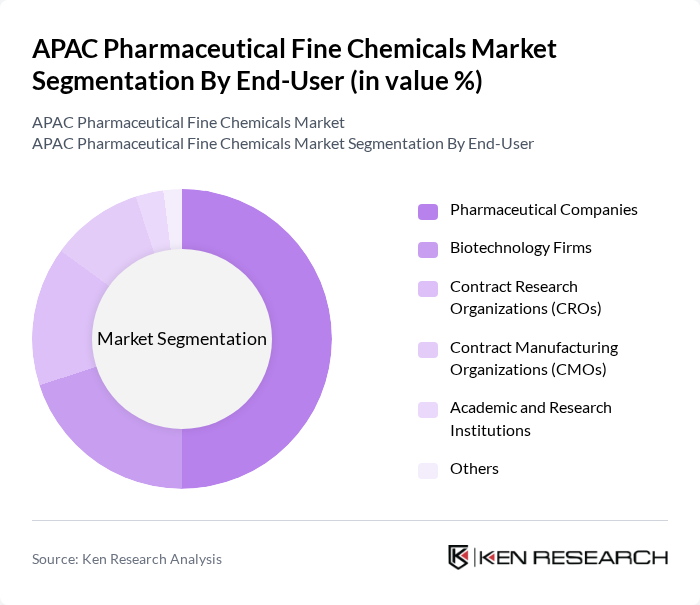
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Contract Research Organizations (CROs), Contract Manufacturing Organizations (CMOs), Academic and Research Institutions, and Others. Pharmaceutical Companies are the leading end-users, driven by the continuous need for new drug development and the increasing focus on personalized medicine. The pharmaceutical sector consumes over 65% of all fine chemical output globally, with over 23,000 drug development projects in pipeline stages. The growing collaboration between pharmaceutical companies and CROs for research and development activities further enhances the demand for fine chemicals. Biotechnology firms have increased demand for niche chemicals by 27% year-on-year due to rising biologics development, with 430 biotech firms in Asia-Pacific alone entering the fine chemical space for individualized drug formulation.
APAC Pharmaceutical Fine Chemicals Market Competitive Landscape
The APAC Pharmaceutical Fine Chemicals Market is characterized by a dynamic mix of regional and international players. Leading participants such as BASF SE, Merck KGaA, Pfizer Inc., Novartis AG, Teva Pharmaceutical Industries Ltd., Lonza Group AG, Aurobindo Pharma Ltd., Dr. Reddy's Laboratories Ltd., Sun Pharmaceutical Industries Ltd., Cipla Ltd., Sandoz (a Novartis division), AbbVie Inc., GlaxoSmithKline plc (GSK), Hikma Pharmaceuticals PLC, Amgen Inc., Zhejiang Huahai Pharmaceutical Co., Ltd., WuXi AppTec Co., Ltd., Shandong Xinhua Pharmaceutical Co., Ltd., Asahi Kasei Corporation, Sumitomo Chemical Co., Ltd., Samsung Biologics Co., Ltd., Kyowa Hakko Bio Co., Ltd., Suven Pharmaceuticals Ltd., Piramal Pharma Solutions, Jubilant Ingrevia Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
APAC Pharmaceutical Fine Chemicals Market Industry Analysis
Growth Drivers
- Increasing Demand for Generic Drugs:The APAC region is witnessing a significant rise in the demand for generic drugs, driven by the increasing prevalence of chronic diseases. In future, the generic drug market in APAC is projected to reach approximately $60 billion, reflecting a compound growth rate of 8% annually. This surge is attributed to cost-effective healthcare solutions and the expiration of patents for several blockbuster drugs, enabling broader access to essential medications across diverse populations.
- Rising Investment in R&D Activities:Investment in research and development (R&D) within the pharmaceutical fine chemicals sector is expected to exceed $15 billion in future across APAC. This growth is fueled by government initiatives and private sector funding aimed at fostering innovation. Countries like China and India are leading this trend, with R&D spending increasing by 10% annually, enhancing the development of new drugs and improving existing formulations, thereby driving market expansion.
- Expanding Healthcare Infrastructure:The healthcare infrastructure in APAC is rapidly expanding, with government spending projected to reach $1 trillion in future. This investment is focused on improving healthcare access and quality, particularly in rural areas. Enhanced infrastructure facilitates better distribution of pharmaceutical fine chemicals, ensuring that essential drugs are available to a larger segment of the population, thus driving market growth and improving health outcomes.
Market Challenges
- Stringent Regulatory Compliance:The pharmaceutical fine chemicals market in APAC faces significant challenges due to stringent regulatory compliance requirements. In future, the cost of compliance is expected to rise to approximately $5 billion, as companies must adhere to complex regulations set by agencies like the FDA and EMA. This can lead to delays in product approvals and increased operational costs, hindering market growth and innovation.
- High Production Costs:High production costs remain a critical challenge for the APAC pharmaceutical fine chemicals market. In future, the average production cost per kilogram of fine chemicals is projected to be around $300, influenced by raw material prices and labor costs. These elevated costs can limit the competitiveness of local manufacturers, particularly against low-cost producers from regions with less stringent regulations, impacting overall market dynamics.
APAC Pharmaceutical Fine Chemicals Market Future Outlook
The future of the APAC pharmaceutical fine chemicals market appears promising, driven by technological advancements and increasing healthcare demands. The integration of artificial intelligence in drug development is expected to streamline processes, reducing time-to-market for new drugs. Additionally, the shift towards personalized medicine will create tailored therapeutic solutions, enhancing patient outcomes. As the region continues to invest in healthcare infrastructure, the market is poised for sustainable growth, addressing both local and global health challenges effectively.
Market Opportunities
- Expansion into Emerging Markets:The APAC region presents significant opportunities for pharmaceutical fine chemicals through expansion into emerging markets. Countries like Vietnam and Indonesia are experiencing rapid economic growth, with healthcare spending projected to increase by 12% annually. This growth opens avenues for local manufacturers to introduce innovative products, catering to the rising demand for affordable healthcare solutions.
- Development of Sustainable Chemicals:There is a growing emphasis on sustainability within the pharmaceutical sector, with investments in the development of sustainable chemicals expected to reach $2 billion in future. Companies focusing on green chemistry practices can capitalize on this trend, appealing to environmentally conscious consumers and regulatory bodies, thereby enhancing their market position and contributing to long-term sustainability goals.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Active Pharmaceutical Ingredients (APIs) Intermediates Excipients Specialty Chemicals Proprietary Fine Chemicals Non-Proprietary Fine Chemicals |
| By End-User | Pharmaceutical Companies Biotechnology Firms Contract Research Organizations (CROs) Contract Manufacturing Organizations (CMOs) Academic and Research Institutions Others |
| By Application | Drug Formulation Drug Development Quality Control and Testing Biopharmaceutical Manufacturing Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | China India Japan South Korea Australia Southeast Asia (including Indonesia, Malaysia, Vietnam, Thailand, Singapore, Philippines) Rest of APAC |
| By Manufacturing Process | Batch Production Continuous Production Green Chemistry Processes Others |
| By Regulatory Compliance | Good Manufacturing Practices (GMP) ISO Standards Local Regulatory Approvals (e.g., CFDA, CDSCO, PMDA) Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Food and Drug Administration)
Pharmaceutical Manufacturers and Producers
Contract Research Organizations (CROs)
Active Pharmaceutical Ingredient (API) Suppliers
Biotechnology Firms
Pharmaceutical Distributors
Quality Control and Assurance Agencies
Players Mentioned in the Report:
BASF SE
Merck KGaA
Pfizer Inc.
Novartis AG
Teva Pharmaceutical Industries Ltd.
Lonza Group AG
Aurobindo Pharma Ltd.
Dr. Reddy's Laboratories Ltd.
Sun Pharmaceutical Industries Ltd.
Cipla Ltd.
Sandoz (a Novartis division)
AbbVie Inc.
GlaxoSmithKline plc (GSK)
Hikma Pharmaceuticals PLC
Amgen Inc.
Zhejiang Huahai Pharmaceutical Co., Ltd.
WuXi AppTec Co., Ltd.
Shandong Xinhua Pharmaceutical Co., Ltd.
Asahi Kasei Corporation
Sumitomo Chemical Co., Ltd.
Samsung Biologics Co., Ltd.
Kyowa Hakko Bio Co., Ltd.
Suven Pharmaceuticals Ltd.
Piramal Pharma Solutions
Jubilant Ingrevia Ltd.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. APAC Pharmaceutical Fine Chemicals Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 APAC Pharmaceutical Fine Chemicals Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. APAC Pharmaceutical Fine Chemicals Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Generic Drugs
3.1.2 Rising Investment in R&D Activities
3.1.3 Expanding Healthcare Infrastructure
3.1.4 Growing Focus on Biopharmaceuticals
3.2 Market Challenges
3.2.1 Stringent Regulatory Compliance
3.2.2 High Production Costs
3.2.3 Supply Chain Disruptions
3.2.4 Competition from Low-Cost Manufacturers
3.3 Market Opportunities
3.3.1 Expansion into Emerging Markets
3.3.2 Development of Sustainable Chemicals
3.3.3 Collaborations with Biotech Firms
3.3.4 Adoption of Advanced Manufacturing Technologies
3.4 Market Trends
3.4.1 Shift Towards Personalized Medicine
3.4.2 Increasing Use of AI in Drug Development
3.4.3 Growth of Contract Manufacturing Organizations (CMOs)
3.4.4 Emphasis on Green Chemistry Practices
3.5 Government Regulation
3.5.1 Regulatory Framework for Drug Approval
3.5.2 Environmental Regulations on Chemical Manufacturing
3.5.3 Intellectual Property Rights Enforcement
3.5.4 Pricing and Reimbursement Policies
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. APAC Pharmaceutical Fine Chemicals Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. APAC Pharmaceutical Fine Chemicals Market Segmentation
8.1 By Type
8.1.1 Active Pharmaceutical Ingredients (APIs)
8.1.2 Intermediates
8.1.3 Excipients
8.1.4 Specialty Chemicals
8.1.5 Proprietary Fine Chemicals
8.1.6 Non-Proprietary Fine Chemicals
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Biotechnology Firms
8.2.3 Contract Research Organizations (CROs)
8.2.4 Contract Manufacturing Organizations (CMOs)
8.2.5 Academic and Research Institutions
8.2.6 Others
8.3 By Application
8.3.1 Drug Formulation
8.3.2 Drug Development
8.3.3 Quality Control and Testing
8.3.4 Biopharmaceutical Manufacturing
8.3.5 Others
8.4 By Distribution Channel
8.4.1 Direct Sales
8.4.2 Distributors
8.4.3 Online Sales
8.4.4 Others
8.5 By Region
8.5.1 China
8.5.2 India
8.5.3 Japan
8.5.4 South Korea
8.5.5 Australia
8.5.6 Southeast Asia (including Indonesia, Malaysia, Vietnam, Thailand, Singapore, Philippines)
8.5.7 Rest of APAC
8.6 By Manufacturing Process
8.6.1 Batch Production
8.6.2 Continuous Production
8.6.3 Green Chemistry Processes
8.6.4 Others
8.7 By Regulatory Compliance
8.7.1 Good Manufacturing Practices (GMP)
8.7.2 ISO Standards
8.7.3 Local Regulatory Approvals (e.g., CFDA, CDSCO, PMDA)
8.7.4 Others
9. APAC Pharmaceutical Fine Chemicals Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue (USD Million/Billion, APAC or Global)
9.2.4 Revenue Growth Rate (CAGR %)
9.2.5 Market Share in APAC (%)
9.2.6 Market Penetration Rate (by product or region)
9.2.7 R&D Investment as % of Revenue
9.2.8 Product Portfolio Breadth (APIs, Intermediates, Excipients, etc.)
9.2.9 Regulatory Compliance Track Record (GMP, local approvals)
9.2.10 Supply Chain Efficiency (lead time, on-time delivery %)
9.2.11 Capacity Utilization Rate (%)
9.2.12 Export Ratio (APAC exports as % of total sales)
9.2.13 Customer Retention Rate (%)
9.2.14 Sustainability/ESG Score
9.2.15 Brand Equity Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 BASF SE
9.5.2 Merck KGaA
9.5.3 Pfizer Inc.
9.5.4 Novartis AG
9.5.5 Teva Pharmaceutical Industries Ltd.
9.5.6 Lonza Group AG
9.5.7 Aurobindo Pharma Ltd.
9.5.8 Dr. Reddy's Laboratories Ltd.
9.5.9 Sun Pharmaceutical Industries Ltd.
9.5.10 Cipla Ltd.
9.5.11 Sandoz (a Novartis division)
9.5.12 AbbVie Inc.
9.5.13 GlaxoSmithKline plc (GSK)
9.5.14 Hikma Pharmaceuticals PLC
9.5.15 Amgen Inc.
9.5.16 Zhejiang Huahai Pharmaceutical Co., Ltd.
9.5.17 WuXi AppTec Co., Ltd.
9.5.18 Shandong Xinhua Pharmaceutical Co., Ltd.
9.5.19 Asahi Kasei Corporation
9.5.20 Sumitomo Chemical Co., Ltd.
9.5.21 Samsung Biologics Co., Ltd.
9.5.22 Kyowa Hakko Bio Co., Ltd.
9.5.23 Suven Pharmaceuticals Ltd.
9.5.24 Piramal Pharma Solutions
9.5.25 Jubilant Ingrevia Ltd.
10. APAC Pharmaceutical Fine Chemicals Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Government Procurement Policies
10.1.2 Budget Allocations for Pharmaceuticals
10.1.3 Supplier Selection Criteria
10.1.4 Contracting Processes
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends in Pharmaceutical Infrastructure
10.2.2 Energy Consumption Patterns
10.2.3 Cost Management Strategies
10.2.4 Sustainability Initiatives
10.3 Pain Point Analysis by End-User Category
10.3.1 Quality Assurance Challenges
10.3.2 Supply Chain Disruptions
10.3.3 Regulatory Compliance Issues
10.3.4 Cost Pressures
10.4 User Readiness for Adoption
10.4.1 Awareness of New Technologies
10.4.2 Training and Skill Development Needs
10.4.3 Financial Readiness
10.4.4 Infrastructure Readiness
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of ROI
10.5.2 Case Studies of Successful Implementations
10.5.3 Future Use Case Opportunities
10.5.4 Feedback Mechanisms for Improvement
11. APAC Pharmaceutical Fine Chemicals Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Identification
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Market Identification
2.4 Communication Strategies
2.5 Digital Marketing Approaches
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 E-commerce Integration
3.4 Logistics and Supply Chain Management
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
5. Unmet Demand & Latent Needs
5.1 Category Gaps Identification
5.2 Consumer Segments Analysis
5.3 Future Demand Projections
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service Strategies
6.3 Customer Feedback Mechanisms
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Competitive Advantages
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategies
9.1.3 Packaging Innovations
9.2 Export Entry Strategy
9.2.1 Target Countries Analysis
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements Analysis
11.2 Timelines for Market Entry
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
12.2 Risk Mitigation Strategies
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Identification
15.2.2 Activity Scheduling
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from pharmaceutical associations across APAC
- Review of market publications and white papers on fine chemicals
- Examination of regulatory frameworks and compliance guidelines from health authorities
Primary Research
- Interviews with R&D Directors at leading pharmaceutical companies
- Surveys with procurement managers in fine chemical manufacturing
- Field interviews with quality assurance professionals in production facilities
Validation & Triangulation
- Cross-validation of data from multiple industry sources and expert opinions
- Triangulation of market size estimates using sales data and production statistics
- Sanity checks through feedback from industry panels and focus groups
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national pharmaceutical expenditure trends
- Segmentation by therapeutic areas and chemical types
- Incorporation of growth rates from emerging markets within APAC
Bottom-up Modeling
- Volume estimates derived from production capacities of key manufacturers
- Cost analysis based on raw material prices and operational expenses
- Calculation of market size using unit sales and average selling prices
Forecasting & Scenario Analysis
- Multi-variable forecasting models incorporating economic indicators and healthcare trends
- Scenario planning based on potential regulatory changes and market disruptions
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturing Insights | 60 | Production Managers, Quality Control Analysts |
| Regulatory Compliance in Fine Chemicals | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Market Trends in Active Pharmaceutical Ingredients (APIs) | 50 | Market Analysts, Product Managers |
| Supply Chain Dynamics in Fine Chemicals | 45 | Supply Chain Managers, Logistics Coordinators |
| Innovation in Pharmaceutical Fine Chemicals | 40 | R&D Directors, Innovation Managers |
Frequently Asked Questions
What is the current value of the APAC Pharmaceutical Fine Chemicals Market?
The APAC Pharmaceutical Fine Chemicals Market is valued at approximately USD 60 billion, highlighting the region's significant role in global pharmaceutical fine chemical production, driven by high-quality pharmaceutical demand and advancements in drug development technologies.