Region:Asia
Author(s):Dev
Product Code:KRAA6109
Pages:89
Published On:January 2026
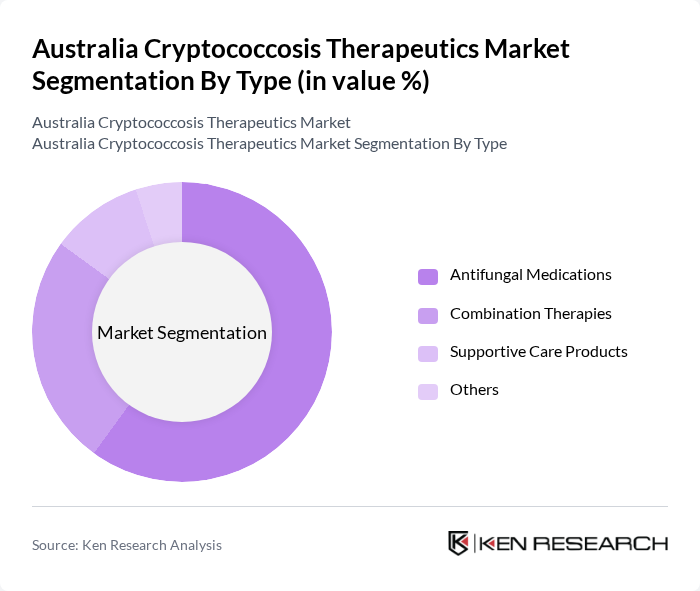
By Type:The market is segmented into various types of therapeutics, including antifungal medications, combination therapies, supportive care products, and others. Antifungal medications such as Amphotericin B, Flucytosine, and Fluconazole are the most widely used due to their direct effectiveness against cryptococcal infections. Combination therapies are gaining traction as they enhance treatment efficacy and reduce resistance. Supportive care products play a crucial role in managing symptoms and improving patient outcomes.
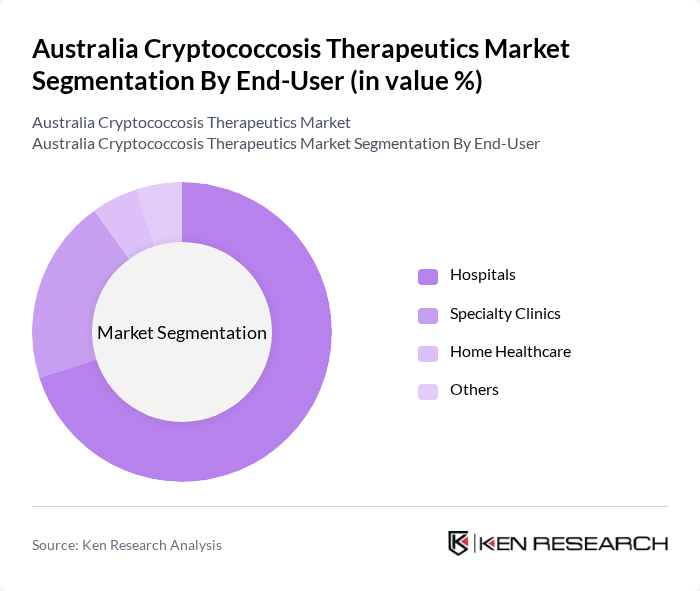
By End-User:The end-user segmentation includes hospitals, specialty clinics, home healthcare, and others. Hospitals are the primary end-users due to their capacity to provide comprehensive care for cryptococcosis patients. Specialty clinics are also significant as they focus on infectious diseases, while home healthcare is emerging as a viable option for ongoing treatment and monitoring.
The Australia Cryptococcosis Therapeutics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Gilead Sciences, Inc., Pfizer Inc., Merck & Co., Inc., Astellas Pharma Inc., Novartis AG, Bristol-Myers Squibb Company, Amgen Inc., Johnson & Johnson, Sanofi S.A., AbbVie Inc., Teva Pharmaceutical Industries Ltd., Eli Lilly and Company, AstraZeneca PLC, Bayer AG, Takeda Pharmaceutical Company Limited contribute to innovation, geographic expansion, and service delivery in this space.
The future of the cryptococcosis therapeutics market in Australia appears promising, driven by ongoing advancements in drug development and increased healthcare investments. As the prevalence of immunocompromised patients continues to rise, the demand for effective treatments will likely grow. Additionally, the integration of digital health technologies and telemedicine is expected to enhance patient management, improving access to care and treatment adherence, ultimately fostering a more robust market environment in future.
| Segment | Sub-Segments |
|---|---|
| By Type | Antifungal Medications Combination Therapies Supportive Care Products Others |
| By End-User | Hospitals Specialty Clinics Home Healthcare Others |
| By Patient Demographics | Adults Pediatric Patients Geriatric Patients Others |
| By Distribution Channel | Retail Pharmacies Online Pharmacies Hospital Pharmacies Others |
| By Geography | New South Wales Victoria Queensland Others |
| By Treatment Duration | Short-term Treatment Long-term Treatment Others |
| By Research and Development Focus | Clinical Trials Post-Marketing Surveillance Drug Repurposing Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 120 | Infectious Disease Specialists, General Practitioners |
| Pharmaceutical Companies | 40 | Product Managers, R&D Directors |
| Patient Advocacy Groups | 60 | Patient Representatives, Healthcare Advocates |
| Clinical Researchers | 50 | Clinical Trial Coordinators, Research Scientists |
| Health Policy Makers | 40 | Government Health Officials, Policy Analysts |
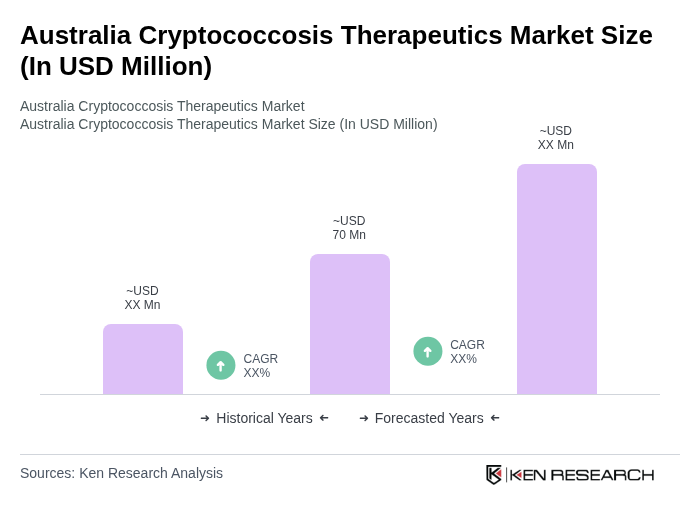
The Australia Cryptococcosis Therapeutics Market is valued at approximately USD 70 million, reflecting a five-year historical analysis. This valuation is influenced by the rising prevalence of cryptococcal infections and advancements in antifungal therapies.