Region:Global
Author(s):Dev
Product Code:KRAA6111
Pages:100
Published On:January 2026
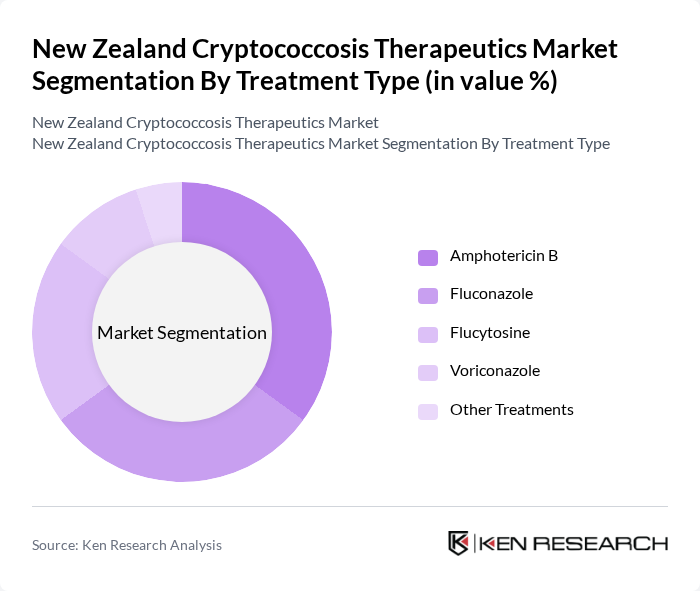
By Treatment Type:The treatment type segmentation includes various antifungal medications used to manage cryptococcosis. The subsegments are Amphotericin B, Fluconazole, Flucytosine, Voriconazole, and Other Treatments. Among these, **Amphotericin B** is the leading subsegment due to its established efficacy in treating severe fungal infections, particularly cryptococcal meningitis, with liposomal formulations enhancing its safety profile and increasing adoption.
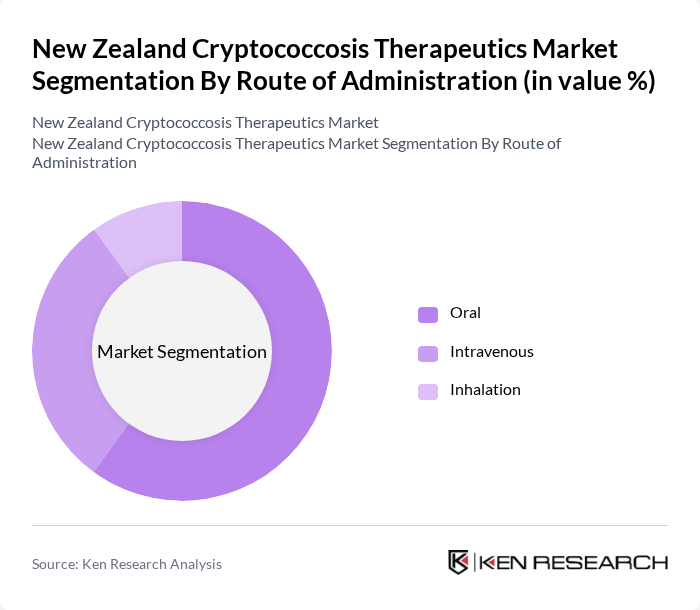
By Route of Administration:The route of administration segmentation includes Oral, Intravenous, and Inhalation methods. Oral administration is the most dominant route due to its convenience and ease of use, particularly for outpatient treatment. This method allows for better patient compliance and is often preferred for long-term management of cryptococcosis.
The New Zealand Cryptococcosis Therapeutics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pfizer Inc., Novartis AG, Johnson & Johnson Services, Inc., Asahi Kasei Pharma, Sigmapharm Laboratories LLC, Gilead Sciences, Inc., Merck & Co., Inc., Astellas Pharma Inc., GlaxoSmithKline plc, Sanofi S.A., Amgen Inc., AbbVie Inc., Teva Pharmaceutical Industries Ltd., Eli Lilly and Company, Bayer AG contribute to innovation, geographic expansion, and service delivery in this space.
The future of the New Zealand cryptococcosis therapeutics market appears promising, driven by ongoing advancements in medical technology and increased healthcare investments. As the healthcare infrastructure expands, particularly in underserved regions, access to diagnostics and treatments will improve. Additionally, the trend towards personalized medicine is expected to enhance therapeutic efficacy, leading to better patient outcomes. Collaborative efforts between pharmaceutical companies and research institutions will further accelerate the development of innovative treatment options, ensuring a robust market landscape.
| Segment | Sub-Segments |
|---|---|
| By Treatment Type | Amphotericin B Fluconazole Flucytosine Voriconazole Other Treatments |
| By Route of Administration | Oral Intravenous Inhalation |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Others |
| By Drug Type | Branded Drugs Generic Drugs |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 100 | Infectious Disease Specialists, General Practitioners |
| Pharmaceutical Distributors | 50 | Sales Representatives, Distribution Managers |
| Patient Advocacy Groups | 40 | Patient Advocates, Support Group Leaders |
| Pharmacy Managers | 45 | Pharmacy Directors, Clinical Pharmacists |
| Health Policy Makers | 40 | Public Health Officials, Policy Analysts |
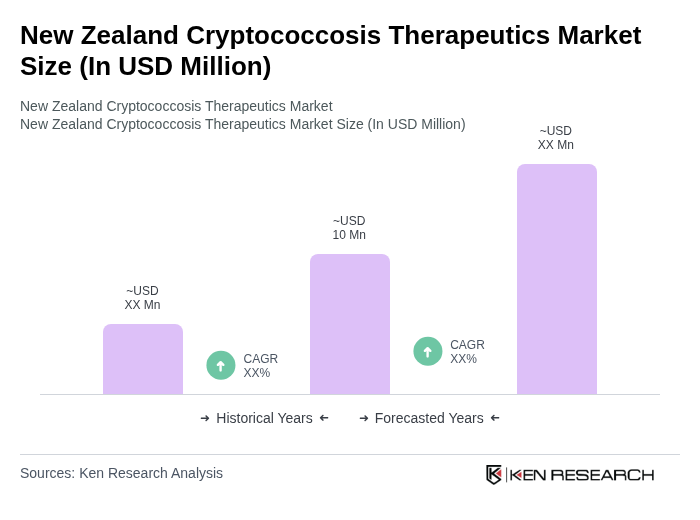
The New Zealand Cryptococcosis Therapeutics Market is valued at approximately USD 10 million, reflecting a five-year historical analysis. This valuation is influenced by the rising prevalence of cryptococcal infections and advancements in treatment options.