Region:Middle East
Author(s):Rebecca
Product Code:KRAD2904
Pages:85
Published On:November 2025
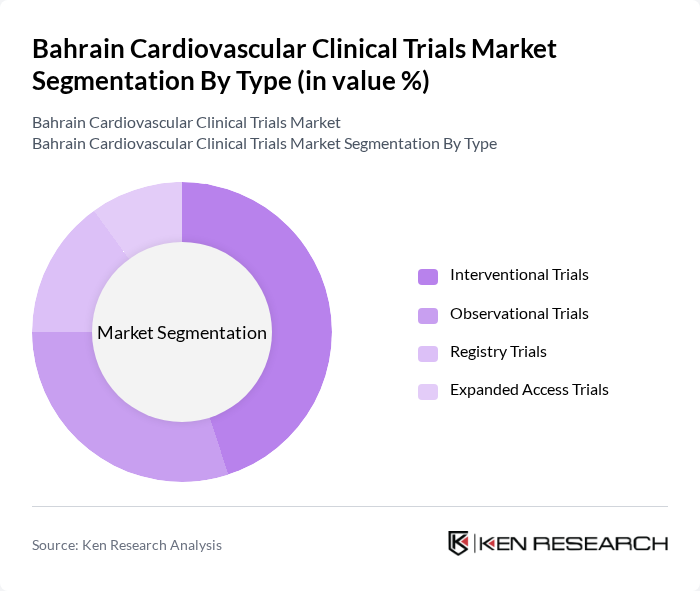
By Type:The market is segmented into various types of clinical trials, including interventional trials, observational trials, registry trials, and expanded access trials. Interventional trials typically lead in market share due to their focus on testing new treatments and therapies, while observational and registry trials support long-term data collection and real-world evidence generation. Expanded access trials provide investigational therapies to patients outside of formal clinical studies.
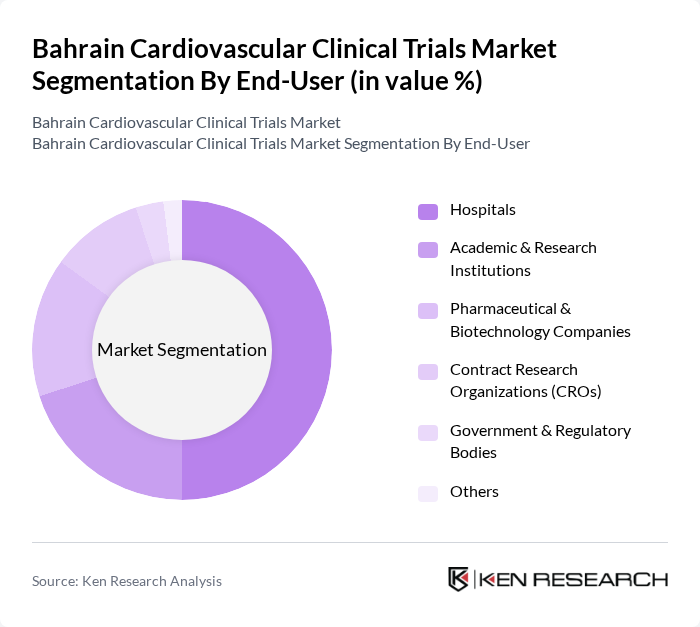
By End-User:The end-users of clinical trials in Bahrain include hospitals, academic and research institutions, pharmaceutical and biotechnology companies, contract research organizations (CROs), government and regulatory bodies, and others. Hospitals are the dominant end-user segment, providing comprehensive infrastructure and patient access for conducting trials. Academic and research institutions contribute to protocol design and data analysis, while pharmaceutical and biotechnology companies drive drug and device development. CROs support operational management and regulatory compliance.
The Bahrain Cardiovascular Clinical Trials Market is characterized by a dynamic mix of regional and international players. Leading participants such as Salmaniya Medical Complex, King Hamad University Hospital Clinical Research Center, Bahrain Defence Force Hospital Research Center, Royal Bahrain Hospital Clinical Trials Unit, Bahrain Specialist Hospital Clinical Research Unit, American Mission Hospital, Ibn Al-Nafis Hospital, Al Hilal Hospital Clinical Research, KIMS Bahrain Medical Centre, National Health Regulatory Authority (NHRA), Arabian Gulf University - Clinical Research Center, Gulf Diabetes Specialist Center, Bahrain Medical Society, Bahrain Oncology Center, Bahrain Red Crescent Society (Clinical Research Division) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Bahrain cardiovascular clinical trials market appears promising, driven by advancements in technology and a growing emphasis on patient-centric approaches. The integration of digital health technologies and telemedicine is expected to enhance patient engagement and streamline trial processes. Additionally, the focus on personalized medicine will likely lead to more targeted therapies, improving patient outcomes and attracting further investment in clinical research initiatives within the region.
| Segment | Sub-Segments |
|---|---|
| By Type | Interventional Trials Observational Trials Registry Trials Expanded Access Trials |
| By End-User | Hospitals Academic & Research Institutions Pharmaceutical & Biotechnology Companies Contract Research Organizations (CROs) Government & Regulatory Bodies Others |
| By Phase of Trial | Phase I Phase II Phase III Phase IV |
| By Therapeutic Area | Coronary Artery Disease Heart Failure Arrhythmias Hypertension Stroke Others |
| By Study Design | Randomized Controlled Trials (RCTs) Non-Randomized Trials Cross-Sectional Studies Cohort Studies Others |
| By Patient Demographics | Age Groups (Pediatric, Adult, Geriatric) Gender Comorbid Conditions (e.g., Diabetes, Obesity) Others |
| By Funding Source | Government Funding Private Sector Funding Non-Profit & International Organizations Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiovascular Clinical Trial Coordinators | 60 | Clinical Research Associates, Site Managers |
| Cardiologists in Bahrain | 50 | Interventional Cardiologists, General Cardiologists |
| Pharmaceutical Companies Conducting Trials | 40 | Clinical Operations Managers, Regulatory Affairs Specialists |
| Patients with Cardiovascular Conditions | 60 | Trial Participants, Patient Advocacy Group Members |
| Healthcare Policy Makers | 40 | Health Economists, Public Health Officials |
The Bahrain Cardiovascular Clinical Trials Market is valued at approximately USD 20 million, reflecting a five-year historical analysis. This growth is attributed to the rising prevalence of cardiovascular diseases and advancements in medical technology.