About the Report
Base Year 2024Bahrain Clinical Trial Equipment Ancillary Solutions Market Overview
- The Bahrain Clinical Trial Equipment Ancillary Solutions Market is valued at USD 180 million, based on a five-year historical analysis of the medical devices and clinical trial supplies sector in Bahrain. This growth is primarily driven by the increasing number of clinical trials, rapid advancements in medical technology, and a growing emphasis on patient-centric research methodologies. Additional market momentum is provided by the rising adoption of digital health solutions, remote patient monitoring, and telemedicine, reflecting global trends in clinical research and healthcare modernization . The demand for high-quality ancillary solutions remains essential for ensuring the integrity and efficiency of clinical trials.
- Key players in this market include Manama, the capital city, which serves as a hub for clinical research activities due to its strategic location and well-established healthcare infrastructure. Additionally, cities like Muharraq and Riffa contribute significantly to the market, supported by government initiatives aimed at enhancing the healthcare sector and attracting international research organizations. Bahrain’s government has invested in specialized medical centers, research institutions, and innovation hubs, further strengthening the country’s position in regional clinical research .
- In 2023, the Bahrain government implemented the “Medical Devices Regulatory Procedures, 2023” issued by the National Health Regulatory Authority (NHRA). This regulation mandates that all clinical trial equipment and medical devices must be registered with the NHRA before use in clinical trials. The regulation covers device registration, safety and efficacy evaluation, and compliance with international standards, thereby enhancing the overall quality and safety of research conducted in the country .
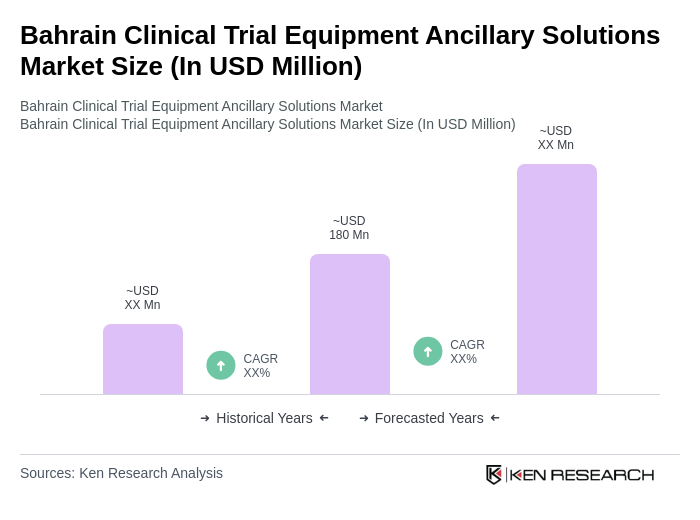
Bahrain Clinical Trial Equipment Ancillary Solutions Market Segmentation
By Type:This segmentation includes various types of equipment and solutions used in clinical trials, which are essential for ensuring the smooth operation and management of trials.

The Patient Monitoring Equipment segment is currently dominating the market due to the increasing need for real-time data collection and patient safety during clinical trials. This segment includes devices such as ECG monitors, blood pressure monitors, and pulse oximeters, which are essential for tracking patient health and ensuring compliance with trial protocols. The growing trend towards remote patient monitoring and telehealth solutions has further propelled the demand for these devices, making them a critical component of clinical trial operations. The adoption of wearable health trackers and connected devices is also accelerating segment growth in Bahrain .
By End-User:This segmentation focuses on the various entities that utilize clinical trial equipment and ancillary solutions, highlighting the diverse applications across different sectors.
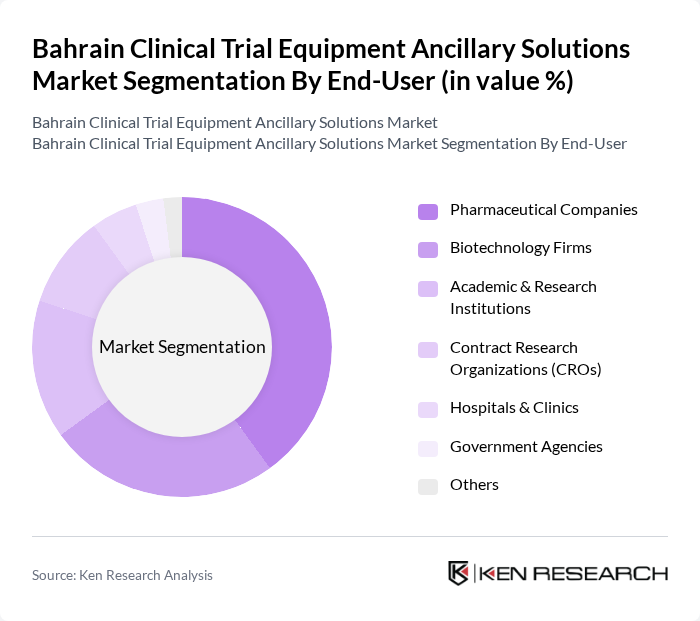
Pharmaceutical Companies are the leading end-users in the market, driven by their extensive involvement in clinical trials for drug development. These companies require a wide range of ancillary solutions to ensure compliance with regulatory standards and to enhance the efficiency of their research processes. The increasing investment in R&D by pharmaceutical firms, coupled with the rising number of clinical trials and the proliferation of biologics and biosimilars, has solidified their position as the dominant end-user segment .
Bahrain Clinical Trial Equipment Ancillary Solutions Market Competitive Landscape
The Bahrain Clinical Trial Equipment Ancillary Solutions Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Thermo Fisher Scientific, Siemens Healthineers, GE HealthCare, Philips, Abbott Laboratories, Stryker, Parexel International, ICON plc, Charles River Laboratories, PPD (part of Thermo Fisher Scientific), Syneos Health, WuXi AppTec, Al Jishi Corporation, Gulf House Medical System W.L.L. contribute to innovation, geographic expansion, and service delivery in this space.
Bahrain Clinical Trial Equipment Ancillary Solutions Market Industry Analysis
Growth Drivers
- Increasing Demand for Clinical Trials:The demand for clinical trials in Bahrain is projected to rise significantly, driven by a growing healthcare sector. In future, the healthcare expenditure in Bahrain is expected to reach approximately $2.1 billion, reflecting a moderate increase from previous periods. This surge is attributed to the increasing number of pharmaceutical companies establishing operations in the region, which necessitates enhanced clinical trial capabilities and ancillary solutions to support their research efforts.
- Government Support for Healthcare Innovation:The Bahraini government has committed to investing approximately $1 billion in healthcare innovation in future, fostering an environment conducive to clinical research. This investment includes funding for clinical trial infrastructure and regulatory improvements, which are essential for attracting international clinical research organizations. Such initiatives are expected to enhance the local clinical trial landscape, driving demand for ancillary solutions and equipment.
- Rising Prevalence of Chronic Diseases:Chronic diseases are on the rise in Bahrain, with diabetes and cardiovascular diseases affecting over 30% of the adult population. The World Health Organization reported that the prevalence of diabetes in Bahrain is projected to reach approximately 15% of adults in future. This alarming trend necessitates increased clinical trials focused on innovative treatments, thereby driving demand for clinical trial equipment and ancillary solutions to support these initiatives.
Market Challenges
- Regulatory Hurdles:Navigating the regulatory landscape in Bahrain poses significant challenges for clinical trial operations. The Bahrain National Health Regulatory Authority has stringent requirements for clinical trial approvals, which can delay project timelines. In future, it is estimated that the average approval time for clinical trials will remain around 6-12 months, hindering the swift execution of research and increasing operational costs for clinical trial sponsors.
- High Costs of Clinical Trial Equipment:The financial burden associated with acquiring clinical trial equipment is a major challenge for stakeholders in Bahrain. The average cost of essential clinical trial equipment, such as imaging devices and laboratory instruments, can exceed $500,000. This high capital requirement limits access for smaller research organizations and may deter investment in clinical trials, ultimately impacting the growth of the ancillary solutions market.
Bahrain Clinical Trial Equipment Ancillary Solutions Market Future Outlook
The future of the Bahrain Clinical Trial Equipment Ancillary Solutions Market appears promising, driven by ongoing investments in healthcare infrastructure and a focus on innovative research methodologies. As the government continues to support healthcare initiatives, the market is likely to witness an influx of international collaborations and partnerships. Additionally, the integration of advanced technologies, such as artificial intelligence and data analytics, will enhance the efficiency of clinical trials, paving the way for more patient-centric approaches and improved outcomes.
Market Opportunities
- Expansion of Clinical Research Organizations:The growth of clinical research organizations (CROs) in Bahrain presents a significant opportunity for ancillary solutions providers. With an estimated 15 new CROs expected to enter the market in future, there will be increased demand for specialized equipment and services tailored to support diverse clinical trials, enhancing the overall research landscape.
- Collaborations with International Firms:Collaborating with international pharmaceutical and biotech companies can open new avenues for growth in Bahrain's clinical trial market. In future, partnerships with global firms are anticipated to increase by 25%, facilitating knowledge transfer and access to advanced technologies, which will enhance local capabilities and drive demand for ancillary solutions.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Patient Monitoring Equipment Sample Collection Devices Data Management Systems Laboratory Equipment Storage & Cold Chain Solutions Transport & Logistics Equipment Packaging and Labelling Solutions Ancillary Supplies (e.g., IV sets, PPE, disposables) Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic & Research Institutions Contract Research Organizations (CROs) Hospitals & Clinics Government Agencies Others |
| By Application | Oncology Trials Cardiovascular Trials Neurology Trials Infectious Disease Trials Rare Disease Trials Others |
| By Distribution Channel | Direct Sales Regional Distributors Online Sales Tender-Based Procurement Others |
| By Component | Hardware Software Services |
| By Pricing Strategy | Premium Pricing Competitive Pricing Value-Based Pricing |
| By Regulatory Compliance | NHRA Registration ISO Certification CE Marking FDA Approval Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Bahrain National Health Regulatory Authority)
Clinical Research Organizations (CROs)
Pharmaceutical and Biotechnology Companies
Medical Device Manufacturers
Healthcare Providers and Hospitals
Supply Chain and Logistics Companies
Insurance Companies and Payers
Players Mentioned in the Report:
Medtronic
Thermo Fisher Scientific
Siemens Healthineers
GE HealthCare
Philips
Abbott Laboratories
Stryker
Parexel International
ICON plc
Charles River Laboratories
PPD (part of Thermo Fisher Scientific)
Syneos Health
WuXi AppTec
Al Jishi Corporation
Gulf House Medical System W.L.L.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Bahrain Clinical Trial Equipment Ancillary Solutions Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Bahrain Clinical Trial Equipment Ancillary Solutions Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Bahrain Clinical Trial Equipment Ancillary Solutions Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for clinical trials
3.1.2 Government support for healthcare innovation
3.1.3 Rising prevalence of chronic diseases
3.1.4 Advancements in clinical trial methodologies
3.2 Market Challenges
3.2.1 Regulatory hurdles
3.2.2 High costs of clinical trial equipment
3.2.3 Limited local expertise
3.2.4 Competition from established markets
3.3 Market Opportunities
3.3.1 Expansion of clinical research organizations
3.3.2 Collaborations with international firms
3.3.3 Growth in personalized medicine
3.3.4 Technological advancements in data management
3.4 Market Trends
3.4.1 Increased focus on patient-centric trials
3.4.2 Adoption of digital health technologies
3.4.3 Integration of artificial intelligence in trials
3.4.4 Shift towards decentralized clinical trials
3.5 Government Regulation
3.5.1 Clinical trial registration requirements
3.5.2 Ethical guidelines for clinical research
3.5.3 Data protection regulations
3.5.4 Import/export regulations for clinical equipment
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Bahrain Clinical Trial Equipment Ancillary Solutions Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Bahrain Clinical Trial Equipment Ancillary Solutions Market Segmentation
8.1 By Type
8.1.1 Patient Monitoring Equipment
8.1.2 Sample Collection Devices
8.1.3 Data Management Systems
8.1.4 Laboratory Equipment
8.1.5 Storage & Cold Chain Solutions
8.1.6 Transport & Logistics Equipment
8.1.7 Packaging and Labelling Solutions
8.1.8 Ancillary Supplies (e.g., IV sets, PPE, disposables)
8.1.9 Others
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Biotechnology Firms
8.2.3 Academic & Research Institutions
8.2.4 Contract Research Organizations (CROs)
8.2.5 Hospitals & Clinics
8.2.6 Government Agencies
8.2.7 Others
8.3 By Application
8.3.1 Oncology Trials
8.3.2 Cardiovascular Trials
8.3.3 Neurology Trials
8.3.4 Infectious Disease Trials
8.3.5 Rare Disease Trials
8.3.6 Others
8.4 By Distribution Channel
8.4.1 Direct Sales
8.4.2 Regional Distributors
8.4.3 Online Sales
8.4.4 Tender-Based Procurement
8.4.5 Others
8.5 By Component
8.5.1 Hardware
8.5.2 Software
8.5.3 Services
8.6 By Pricing Strategy
8.6.1 Premium Pricing
8.6.2 Competitive Pricing
8.6.3 Value-Based Pricing
8.7 By Regulatory Compliance
8.7.1 NHRA Registration
8.7.2 ISO Certification
8.7.3 CE Marking
8.7.4 FDA Approval
8.7.5 Others
9. Bahrain Clinical Trial Equipment Ancillary Solutions Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (Bahrain clinical trial segment)
9.2.4 Market Penetration Rate (share of clinical trial equipment supplied in Bahrain)
9.2.5 Number of NHRA-Registered Products
9.2.6 Local Service & Support Presence
9.2.7 Customer Retention Rate (Bahrain healthcare clients)
9.2.8 Pricing Strategy (premium, value, tender-based)
9.2.9 Product Diversification Index (range of clinical trial solutions offered)
9.2.10 Operational Efficiency Ratio (delivery lead times, SLA adherence)
9.2.11 Research and Development Investment (% of revenue)
9.2.12 Brand Equity Score (Bahrain market recognition)
9.2.13 Tender Awards Won (Bahrain Tender Board/NHRA)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medtronic
9.5.2 Thermo Fisher Scientific
9.5.3 Siemens Healthineers
9.5.4 GE HealthCare
9.5.5 Philips
9.5.6 Abbott Laboratories
9.5.7 Stryker
9.5.8 Parexel International
9.5.9 ICON plc
9.5.10 Charles River Laboratories
9.5.11 PPD (part of Thermo Fisher Scientific)
9.5.12 Syneos Health
9.5.13 WuXi AppTec
9.5.14 Al Jishi Corporation
9.5.15 Gulf House Medical System W.L.L.
10. Bahrain Clinical Trial Equipment Ancillary Solutions Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Industry and Commerce
10.1.3 Ministry of Education
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Clinical Research Facilities
10.2.2 Funding for Equipment Upgrades
10.2.3 Budget Allocation for Trials
10.3 Pain Point Analysis by End-User Category
10.3.1 Equipment Availability
10.3.2 Training and Support
10.3.3 Compliance with Regulations
10.4 User Readiness for Adoption
10.4.1 Awareness of New Technologies
10.4.2 Training Needs
10.4.3 Financial Readiness
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Success Metrics
10.5.2 Case Studies of Successful Implementations
10.5.3 Future Investment Plans
11. Bahrain Clinical Trial Equipment Ancillary Solutions Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from Bahrain's Ministry of Health and local regulatory bodies
- Review of published studies on clinical trial equipment usage in the Middle East
- Examination of market trends and forecasts from healthcare journals and trade publications
Primary Research
- Interviews with clinical trial coordinators and project managers in Bahrain
- Surveys targeting equipment suppliers and manufacturers in the clinical trial sector
- Field interviews with healthcare professionals involved in clinical research
Validation & Triangulation
- Cross-validation of findings through multiple data sources including government and industry reports
- Triangulation of qualitative insights from interviews with quantitative data from surveys
- Sanity checks conducted through expert panel reviews comprising industry veterans
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and clinical trial budgets
- Segmentation by types of clinical trial equipment and ancillary services
- Incorporation of growth rates from regional clinical trial activity reports
Bottom-up Modeling
- Data collection from leading clinical trial equipment suppliers regarding sales volumes
- Operational cost analysis based on pricing models of equipment and services
- Volume x cost calculations for various types of clinical trials conducted in Bahrain
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as regulatory changes and technological advancements
- Scenario modeling based on potential increases in clinical trial activity and funding
- Baseline, optimistic, and pessimistic projections through 2030 based on market dynamics
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Trial Equipment Suppliers | 60 | Sales Managers, Product Development Leads |
| Clinical Research Organizations (CROs) | 50 | Project Managers, Clinical Research Associates |
| Healthcare Institutions Conducting Trials | 40 | Clinical Trial Coordinators, Research Directors |
| Regulatory Bodies and Ethics Committees | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Investors in Clinical Research | 40 | Venture Capitalists, Healthcare Investment Analysts |
Frequently Asked Questions
What is the current value of the Bahrain Clinical Trial Equipment Ancillary Solutions Market?
The Bahrain Clinical Trial Equipment Ancillary Solutions Market is valued at approximately USD 180 million, reflecting a significant growth driven by the increasing number of clinical trials and advancements in medical technology.