About the Report
Base Year 2024Qatar Clinical Trial Equipment Ancillary Solutions Market Overview
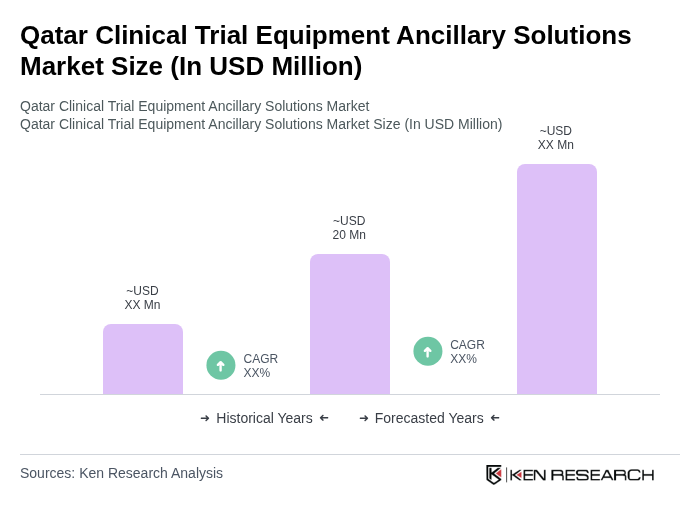
- The Qatar Clinical Trial Equipment Ancillary Solutions Market is valued at USD 20 million, based on a five-year historical analysis. This growth is primarily driven by the increasing number of clinical trials, rapid adoption of advanced medical technology, and a strong focus on patient-centric research approaches. The demand for ancillary solutions, including logistics and data management, has surged as stakeholders seek to enhance trial efficiency and compliance.
- Key players in this market include Doha, Al Rayyan, and Al Wakrah, which dominate due to their robust healthcare infrastructure, presence of leading hospitals and research institutions, and government support for clinical research initiatives. These cities are pivotal in attracting both local and international pharmaceutical and biotechnology firms, contributing to the overall growth of the market.
- In 2023, the Qatar government implemented a new regulation mandating that all clinical trials must adhere to the Good Clinical Practice (GCP) guidelines established by the World Health Organization. This regulation aims to ensure the safety and efficacy of clinical trials conducted within the country, thereby enhancing the credibility of research outcomes and fostering international collaboration.
Qatar Clinical Trial Equipment Ancillary Solutions Market Segmentation
By Type:The market is segmented into various types, including sourcing, supply/logistics, laboratory services, data management solutions, monitoring and compliance tools, clinical trial management systems, and others. Each of these segments plays a crucial role in supporting the clinical trial process, with specific sub-segments catering to distinct needs within the industry.

By End-User:The end-user segmentation includes pharmaceutical companies, biotechnology firms, contract research organizations (CROs), academic and research institutions, hospitals and medical centers, and others. Each end-user category has unique requirements and contributes differently to the market dynamics.
Qatar Clinical Trial Equipment Ancillary Solutions Market Competitive Landscape
The Qatar Clinical Trial Equipment Ancillary Solutions Market is characterized by a dynamic mix of regional and international players. Leading participants such as Qatar Medical Devices Company, Doha Clinical Trials Center (Hamad Medical Corporation), Qatar Biobank, Gulf Clinical Research Organization (GCRO), Qatar University Biomedical Research Center, Al Ahli Hospital, Hamad Medical Corporation, Qatar Pharma, Qatar Clinical Research Institute (QCRI), Medtronic, Novartis, Pfizer, Roche, Merck, AstraZeneca contribute to innovation, geographic expansion, and service delivery in this space.
Qatar Clinical Trial Equipment Ancillary Solutions Market Industry Analysis
Growth Drivers
- Increasing Demand for Clinical Trials:The demand for clinical trials in Qatar is projected to rise significantly, with over 200 active trials reported in future. This surge is driven by the country's strategic focus on becoming a regional hub for medical research, supported by a healthcare expenditure of approximately USD 6.2 billion. The growing prevalence of chronic diseases, such as diabetes and cardiovascular conditions, further fuels the need for innovative clinical solutions, enhancing the market for ancillary equipment.
- Government Support for Healthcare Innovation:The Qatari government has allocated around USD 1.5 billion for healthcare innovation initiatives, aiming to enhance research capabilities. This funding supports the establishment of advanced clinical trial facilities and encourages partnerships with international organizations. Additionally, the Qatar National Vision 2030 emphasizes healthcare as a priority sector, fostering an environment conducive to clinical trial advancements and ancillary solutions development.
- Rising Investment in Healthcare Infrastructure:Qatar's healthcare infrastructure is undergoing significant expansion, with investments exceeding USD 3 billion in new facilities and technologies planned. This includes the construction of specialized research centers and hospitals equipped with state-of-the-art clinical trial equipment. Such investments not only enhance the capacity for conducting trials but also attract global pharmaceutical companies, thereby increasing the demand for ancillary solutions in the clinical trial sector.
Market Challenges
- Regulatory Hurdles and Compliance Issues:Navigating the regulatory landscape in Qatar poses significant challenges for clinical trial operations. The country has stringent guidelines that require extensive documentation and approval processes, which can delay trial initiation. In future, the average approval time for clinical trials is estimated to be around 6-12 months, impacting the timely deployment of clinical trial equipment and ancillary solutions, thus hindering market growth.
- High Costs Associated with Clinical Trial Equipment:The financial burden of acquiring advanced clinical trial equipment remains a critical challenge. In future, the average cost of essential ancillary solutions is projected to be around USD 500,000 per trial. This high expenditure can deter smaller research organizations from participating in clinical trials, limiting the overall market potential. Additionally, the need for ongoing maintenance and training further escalates costs, impacting profitability.
Qatar Clinical Trial Equipment Ancillary Solutions Market Future Outlook
The future of the Qatar Clinical Trial Equipment Ancillary Solutions Market appears promising, driven by ongoing investments in healthcare infrastructure and a growing emphasis on patient-centric trial designs. As the country continues to enhance its regulatory framework, the market is likely to witness increased participation from both local and international stakeholders. Furthermore, the integration of digital technologies and data analytics will streamline trial processes, improving efficiency and patient engagement, ultimately fostering a more robust clinical trial ecosystem.
Market Opportunities
- Expansion of Clinical Trial Services in Emerging Markets:Qatar's strategic location positions it as a gateway to emerging markets in the Middle East and North Africa. By leveraging this advantage, local companies can expand their clinical trial services, tapping into a growing demand for innovative healthcare solutions in these regions, potentially increasing revenue streams significantly.
- Collaborations with International Research Organizations:Forming partnerships with global research institutions can enhance Qatar's clinical trial capabilities. Such collaborations can lead to knowledge transfer, access to advanced technologies, and shared resources, ultimately improving the quality and efficiency of clinical trials conducted in the region, thereby boosting market growth.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Sourcing (Procurement, Rental) Supply/Logistics (Transportation, Packaging, Direct-to-Patient Logistics) Laboratory Services (Bioanalytical Testing, Sample Management) Data Management Solutions (Electronic Data Capture, ePRO, eCOA) Monitoring and Compliance Tools (Remote Monitoring Devices, Temperature Monitoring, Calibration Services) Clinical Trial Management Systems (CTMS, RTSM) Others (Patient Recruitment Services, Ancillary Supplies) |
| By End-User | Pharmaceutical companies Biotechnology firms Contract research organizations (CROs) Academic and research institutions Hospitals and Medical Centers Others |
| By Application | Oncology trials Cardiovascular trials Neurology trials Infectious disease trials Autoimmune and Inflammatory Disease Trials Others |
| By Region | Doha Al Rayyan Umm Salal Al Wakrah Others |
| By Technology | Electronic data capture (EDC) Randomization and trial supply management (RTSM) Clinical trial management software (CTMS) Remote Monitoring Devices and IoT Solutions Others |
| By Investment Source | Private equity Government funding Venture capital Others |
| By Policy Support | Research grants Tax incentives Regulatory support programs Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Qatar Ministry of Public Health, Qatar Food Safety and Health Authority)
Clinical Research Organizations (CROs)
Pharmaceutical and Biotechnology Companies
Medical Device Manufacturers
Healthcare Providers and Hospitals
Logistics and Supply Chain Companies
Insurance Companies and Payers
Players Mentioned in the Report:
Qatar Medical Devices Company
Doha Clinical Trials Center (Hamad Medical Corporation)
Qatar Biobank
Gulf Clinical Research Organization (GCRO)
Qatar University Biomedical Research Center
Al Ahli Hospital
Hamad Medical Corporation
Qatar Pharma
Qatar Clinical Research Institute (QCRI)
Medtronic
Novartis
Pfizer
Roche
Merck
AstraZeneca
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Qatar Clinical Trial Equipment Ancillary Solutions Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Qatar Clinical Trial Equipment Ancillary Solutions Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Qatar Clinical Trial Equipment Ancillary Solutions Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for clinical trials in Qatar
3.1.2 Government support for healthcare innovation
3.1.3 Rising investment in healthcare infrastructure
3.1.4 Technological advancements in clinical trial processes
3.2 Market Challenges
3.2.1 Regulatory hurdles and compliance issues
3.2.2 High costs associated with clinical trial equipment
3.2.3 Limited availability of skilled professionals
3.2.4 Competition from established global players
3.3 Market Opportunities
3.3.1 Expansion of clinical trial services in emerging markets
3.3.2 Collaborations with international research organizations
3.3.3 Development of innovative ancillary solutions
3.3.4 Increased focus on patient-centric trial designs
3.4 Market Trends
3.4.1 Shift towards decentralized clinical trials
3.4.2 Integration of digital technologies in trial management
3.4.3 Growing emphasis on data analytics and real-time monitoring
3.4.4 Rise in patient engagement and recruitment strategies
3.5 Government Regulation
3.5.1 Stricter guidelines for clinical trial approvals
3.5.2 Enhanced focus on patient safety and ethical standards
3.5.3 Incentives for local manufacturing of clinical trial equipment
3.5.4 Policies promoting transparency in clinical trial processes
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Qatar Clinical Trial Equipment Ancillary Solutions Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Qatar Clinical Trial Equipment Ancillary Solutions Market Segmentation
8.1 By Type
8.1.1 Sourcing (Procurement, Rental)
8.1.2 Supply/Logistics (Transportation, Packaging, Direct-to-Patient Logistics)
8.1.3 Laboratory Services (Bioanalytical Testing, Sample Management)
8.1.4 Data Management Solutions (Electronic Data Capture, ePRO, eCOA)
8.1.5 Monitoring and Compliance Tools (Remote Monitoring Devices, Temperature Monitoring, Calibration Services)
8.1.6 Clinical Trial Management Systems (CTMS, RTSM)
8.1.7 Others (Patient Recruitment Services, Ancillary Supplies)
8.2 By End-User
8.2.1 Pharmaceutical companies
8.2.2 Biotechnology firms
8.2.3 Contract research organizations (CROs)
8.2.4 Academic and research institutions
8.2.5 Hospitals and Medical Centers
8.2.6 Others
8.3 By Application
8.3.1 Oncology trials
8.3.2 Cardiovascular trials
8.3.3 Neurology trials
8.3.4 Infectious disease trials
8.3.5 Autoimmune and Inflammatory Disease Trials
8.3.6 Others
8.4 By Region
8.4.1 Doha
8.4.2 Al Rayyan
8.4.3 Umm Salal
8.4.4 Al Wakrah
8.4.5 Others
8.5 By Technology
8.5.1 Electronic data capture (EDC)
8.5.2 Randomization and trial supply management (RTSM)
8.5.3 Clinical trial management software (CTMS)
8.5.4 Remote Monitoring Devices and IoT Solutions
8.5.5 Others
8.6 By Investment Source
8.6.1 Private equity
8.6.2 Government funding
8.6.3 Venture capital
8.6.4 Others
8.7 By Policy Support
8.7.1 Research grants
8.7.2 Tax incentives
8.7.3 Regulatory support programs
8.7.4 Others
9. Qatar Clinical Trial Equipment Ancillary Solutions Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (Qatar Market)
9.2.4 Market Penetration Rate (Number of Trials Supported/Equipment Supplied in Qatar)
9.2.5 Customer Retention Rate (Repeat Contracts/Client Loyalty)
9.2.6 Pricing Strategy (Premium, Value-based, Volume Discount, etc.)
9.2.7 Product Development Cycle Time (Time to Market for New Solutions)
9.2.8 Average Contract Value (Qatar Clinical Trial Segment)
9.2.9 Customer Satisfaction Score (NPS or Equivalent)
9.2.10 Market Share Percentage (Qatar Clinical Trial Equipment Ancillary Solutions)
9.2.11 Local Partnerships/Presence (Qatar-based Operations, JVs, or Distribution)
9.2.12 Regulatory Compliance Track Record (Qatar MOPH, International Standards)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Qatar Medical Devices Company
9.5.2 Doha Clinical Trials Center (Hamad Medical Corporation)
9.5.3 Qatar Biobank
9.5.4 Gulf Clinical Research Organization (GCRO)
9.5.5 Qatar University Biomedical Research Center
9.5.6 Al Ahli Hospital
9.5.7 Hamad Medical Corporation
9.5.8 Qatar Pharma
9.5.9 Qatar Clinical Research Institute (QCRI)
9.5.10 Medtronic
9.5.11 Novartis
9.5.12 Pfizer
9.5.13 Roche
9.5.14 Merck
9.5.15 AstraZeneca
10. Qatar Clinical Trial Equipment Ancillary Solutions Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Public Health
10.1.2 Ministry of Education and Higher Education
10.1.3 Ministry of Finance
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in clinical trial facilities
10.2.2 Funding for research and development
10.2.3 Budget allocation for technology upgrades
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Pharmaceutical companies
10.3.2 CROs
10.3.3 Academic institutions
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of clinical trial benefits
10.4.2 Training and support requirements
10.4.3 Technology adoption barriers
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of trial success rates
10.5.2 Cost savings analysis
10.5.3 Expansion into new therapeutic areas
10.5.4 Others
11. Qatar Clinical Trial Equipment Ancillary Solutions Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Business model development
1.3 Value proposition analysis
1.4 Competitive landscape assessment
1.5 Customer segmentation
1.6 Revenue stream identification
1.7 Cost structure analysis
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience identification
2.4 Communication strategies
2.5 Digital marketing initiatives
2.6 Partnership opportunities
2.7 Market positioning tactics
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 Online distribution channels
3.4 Direct sales approaches
3.5 Logistics and supply chain management
3.6 Distribution partnerships
3.7 Inventory management strategies
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing strategies
4.4 Customer willingness to pay
4.5 Price elasticity considerations
4.6 Discounting strategies
4.7 Value-based pricing models
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
5.3 Emerging trends and needs
5.4 Product development opportunities
5.5 Customer feedback mechanisms
5.6 Market research insights
5.7 Innovation opportunities
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer engagement strategies
6.4 Feedback and improvement processes
6.5 Relationship management tools
6.6 Customer support initiatives
6.7 Community building efforts
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Unique selling propositions
7.4 Customer-centric approaches
7.5 Competitive advantages
7.6 Value delivery mechanisms
7.7 Brand loyalty factors
8. Key Activities
8.1 Regulatory compliance
8.2 Branding efforts
8.3 Distribution setup
8.4 Marketing campaigns
8.5 Training and development
8.6 Performance monitoring
8.7 Stakeholder engagement
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band analysis
9.1.3 Packaging strategies
9.1.4 Distribution channels
9.1.5 Marketing approaches
9.1.6 Regulatory compliance
9.1.7 Risk assessment
9.2 Export Entry Strategy
9.2.1 Target countries identification
9.2.2 Compliance roadmap development
9.2.3 Market entry barriers
9.2.4 Distribution partnerships
9.2.5 Marketing strategies
9.2.6 Risk management
9.2.7 Financial projections
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
10.5 Strategic Alliances
10.6 Licensing Agreements
10.7 Franchising Opportunities
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines for market entry
11.3 Funding sources
11.4 Budget allocation
11.5 Financial modeling
11.6 Risk assessment
11.7 Contingency planning
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnership dynamics
12.3 Risk management strategies
12.4 Control mechanisms
12.5 Decision-making processes
12.6 Performance monitoring
12.7 Exit strategies
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
13.3 Profit margin analysis
13.4 Revenue projections
13.5 Cost management strategies</h
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from healthcare regulatory bodies in Qatar
- Review of published studies on clinical trial equipment usage and trends
- Examination of industry publications and white papers on ancillary solutions
Primary Research
- Interviews with clinical trial coordinators and project managers in Qatar
- Surveys targeting equipment suppliers and service providers in the clinical trial sector
- Field interviews with healthcare professionals involved in clinical trials
Validation & Triangulation
- Cross-validation of findings through multiple data sources including academic journals
- Triangulation of insights from primary interviews and secondary data analysis
- Sanity checks conducted through expert panel discussions with industry veterans
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and clinical trial budgets
- Segmentation of market by type of clinical trial equipment and ancillary services
- Incorporation of government initiatives promoting clinical research in Qatar
Bottom-up Modeling
- Data collection from leading clinical trial equipment suppliers on sales volumes
- Operational cost analysis based on service pricing models in the market
- Volume x cost calculations for various types of ancillary solutions
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating growth rates in clinical trials and healthcare investments
- Scenario modeling based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Trial Equipment Suppliers | 100 | Sales Managers, Product Development Leads |
| Clinical Research Organizations (CROs) | 80 | Project Managers, Clinical Operations Directors |
| Healthcare Professionals in Clinical Trials | 75 | Clinical Trial Coordinators, Principal Investigators |
| Regulatory Affairs Experts | 50 | Regulatory Managers, Compliance Officers |
| Pharmaceutical Companies | 90 | Clinical Development Managers, R&D Directors |
Frequently Asked Questions
What is the current value of the Qatar Clinical Trial Equipment Ancillary Solutions Market?
The Qatar Clinical Trial Equipment Ancillary Solutions Market is valued at approximately USD 20 million, reflecting a significant growth trend driven by the increasing number of clinical trials and advancements in medical technology.