About the Report
Base Year 2024Bahrain Continuous Manufacturing Small Molecule APIs Market Overview
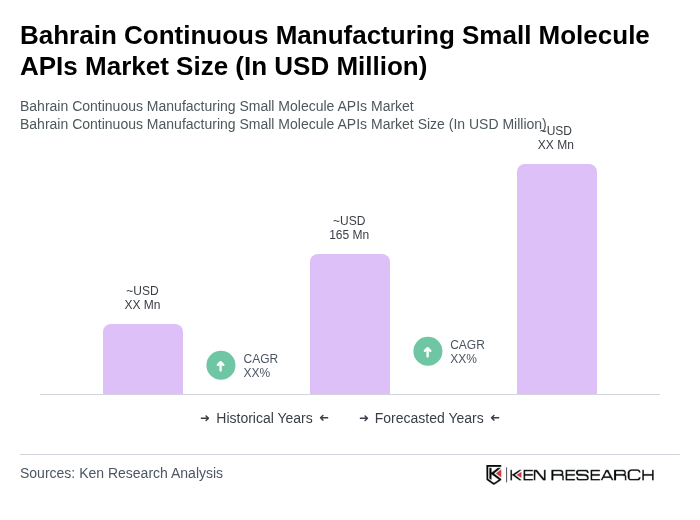
- The Bahrain Continuous Manufacturing Small Molecule APIs Market is valued at USD 165 million, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for efficient and cost-effective manufacturing processes in the pharmaceutical sector, alongside the rising prevalence of chronic diseases that necessitate the development of new drugs. The global pharmaceutical continuous manufacturing market is expanding rapidly, with the sector valued at USD 3.4 billion in 2025 and projected to reach USD 8.4 billion by 2034, demonstrating strong industry momentum that extends to regional markets like Bahrain.
- Key players in this market include Manama, which is the capital and largest city, and other significant contributors like Muharraq and Riffa. These cities dominate the market due to their strategic locations, robust infrastructure, and the presence of pharmaceutical manufacturing facilities that cater to both local and international markets.
- Bahrain's pharmaceutical manufacturing sector operates under the Bahrain Pharmaceutical Manufacturing Regulations issued by the National Medicines and Food Administration (NMFA), which establishes comprehensive standards for API production facilities. These regulations mandate adherence to Good Manufacturing Practice (GMP) standards, require facility licensing and periodic inspections, and establish quality control thresholds for all pharmaceutical manufacturing operations. The regulatory framework ensures that pharmaceutical companies maintain production efficiency standards and implement sustainable manufacturing practices, with compliance verification conducted through regular audits and facility assessments.
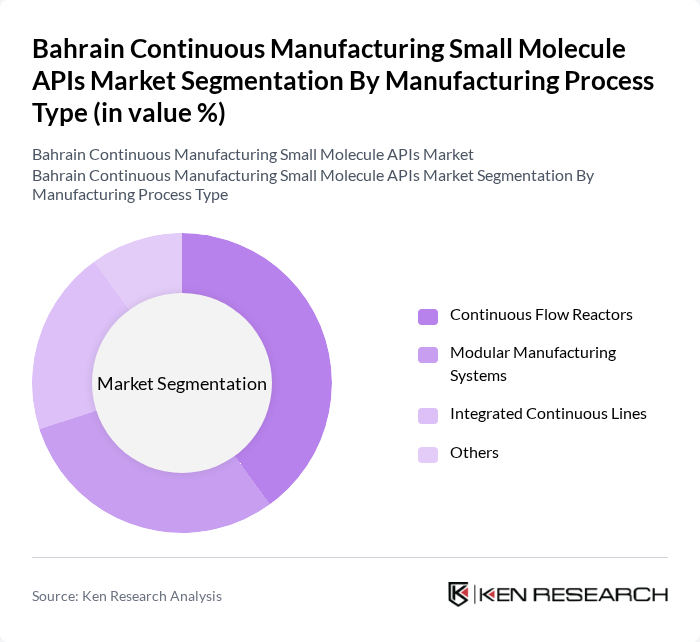
Bahrain Continuous Manufacturing Small Molecule APIs Market Segmentation
By Manufacturing Process Type:The manufacturing process type segment includes various methods employed in the production of small molecule APIs. Continuous flow reactors are gaining traction due to their efficiency and scalability, while modular manufacturing systems offer flexibility and rapid deployment. Integrated continuous lines are also becoming popular as they streamline the production process. The "Others" category includes alternative methods that are less commonly used but still relevant in niche applications.
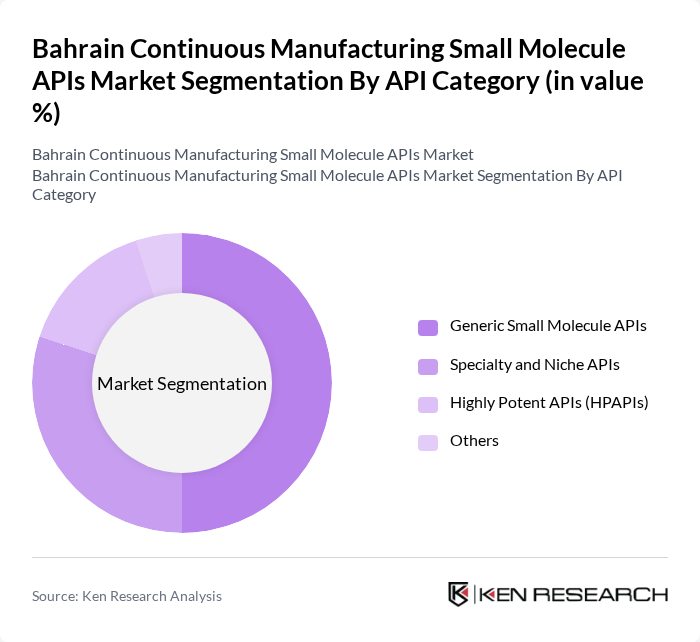
By API Category:The API category segment encompasses various types of small molecule APIs, including generic small molecule APIs, which dominate the market due to their widespread use and lower production costs. Specialty and niche APIs are also significant, catering to specific therapeutic areas. Highly potent APIs (HPAPIs) are gaining attention due to their effectiveness in treating complex diseases, while the "Others" category includes less common APIs that still play a role in the market.
Bahrain Continuous Manufacturing Small Molecule APIs Market Competitive Landscape
The Bahrain Continuous Manufacturing Small Molecule APIs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Gulf Pharmaceutical Industries (Julphar), Bahrain Pharma, United Pharmaceutical Manufacturing Company, Al-Hekma Pharmaceuticals, Badr Pharma, Noor Pharmaceuticals, Gulf Biotech, Bahrain International Investment Park (BIIP) - Pharmaceutical Zone, Al-Muhaidib Pharmaceutical Company, Ameen Al-Fahad Group contribute to innovation, geographic expansion, and service delivery in this space.
Bahrain Continuous Manufacturing Small Molecule APIs Market Industry Analysis
Growth Drivers
- Increasing Demand for Cost-Effective Production Methods:The Bahrain pharmaceutical sector is projected to grow at a rate of 6.5% annually, driven by the need for cost-effective production methods. Continuous manufacturing offers a streamlined process that reduces waste and operational costs. In future, the average cost savings from adopting continuous manufacturing is estimated to be around $1.5 million per facility, making it an attractive option for local manufacturers aiming to enhance profitability while meeting rising demand.
- Technological Advancements in Continuous Manufacturing:The integration of advanced technologies in continuous manufacturing is transforming the production landscape. In future, investments in automation and real-time monitoring systems are expected to exceed $600 million in Bahrain. These innovations enhance efficiency and product quality, allowing manufacturers to produce small molecule APIs with greater precision and speed, thus meeting the increasing market demand for high-quality pharmaceuticals.
- Rising Focus on Regulatory Compliance and Quality Assurance:Bahrain's pharmaceutical industry is increasingly prioritizing regulatory compliance, with the government investing approximately $250 million in enhancing quality assurance frameworks. In future, the implementation of stringent Good Manufacturing Practices (GMP) is expected to drive the adoption of continuous manufacturing, as it aligns with the need for consistent quality and safety in drug production, thereby boosting market growth.
Market Challenges
- High Initial Investment Costs:The transition to continuous manufacturing requires significant upfront capital, estimated at around $3.5 million per facility. This high initial investment poses a barrier for many small and medium-sized enterprises in Bahrain. In future, the financial burden of adopting new technologies may deter potential entrants, limiting competition and innovation within the small molecule APIs market.
- Limited Skilled Workforce in Continuous Manufacturing:The lack of a skilled workforce proficient in continuous manufacturing techniques is a significant challenge. Currently, only 20% of the workforce in Bahrain's pharmaceutical sector possesses the necessary skills. In future, this skills gap may hinder the effective implementation of advanced manufacturing processes, impacting productivity and the overall growth of the small molecule APIs market.
Bahrain Continuous Manufacturing Small Molecule APIs Market Future Outlook
The future of the Bahrain continuous manufacturing small molecule APIs market appears promising, driven by technological advancements and a growing emphasis on regulatory compliance. As the industry embraces automation and real-time monitoring, production efficiency is expected to improve significantly. Additionally, the government's support for local production initiatives will likely foster a conducive environment for growth. By future, the market is anticipated to witness increased collaboration between pharmaceutical companies and research institutions, further enhancing innovation and competitiveness in the region.
Market Opportunities
- Growing Demand for Personalized Medicine:The shift towards personalized medicine presents a significant opportunity for the continuous manufacturing sector. In future, the market for personalized medicine in Bahrain is projected to reach $200 million, creating demand for tailored small molecule APIs that can be efficiently produced through continuous processes, thus enhancing patient outcomes.
- Potential for Export to Neighboring GCC Markets:Bahrain's strategic location offers a gateway to the GCC markets, with an export potential estimated at $250 million by future. Continuous manufacturing can enable local companies to meet the growing demand for high-quality APIs in these markets, positioning Bahrain as a key player in the regional pharmaceutical supply chain.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Manufacturing Process Type | Continuous Flow Reactors Modular Manufacturing Systems Integrated Continuous Lines Others |
| By API Category | Generic Small Molecule APIs Specialty and Niche APIs Highly Potent APIs (HPAPIs) Others |
| By End-User | Pharmaceutical Manufacturers Contract Development and Manufacturing Organizations (CDMOs) Research and Development Institutions Others |
| By Therapeutic Application | Oncology Cardiovascular Diseases Neurology and Psychiatry Infectious Diseases Others |
| By Production Capacity Scale | Pilot Scale (kg-100kg) Small Scale (100kg-1 ton) Medium Scale (1-10 tons) Large Scale (>10 tons) |
| By Distribution Channel | Direct Supply to Pharmaceutical Companies CDMO and Contract Manufacturers Pharmaceutical Distributors Others |
| By Government Support Mechanism | Government Subsidies and Grants Tax Incentives and Exemptions Research and Development Funding Infrastructure Support Programs |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Bahrain National Oil and Gas Authority, Bahrain Economic Development Board)
Manufacturers and Producers of Small Molecule APIs
Pharmaceutical Companies
Contract Manufacturing Organizations (CMOs)
Supply Chain and Logistics Providers
Industry Associations (e.g., Bahrain Pharmaceutical Society)
Financial Institutions and Banks
Players Mentioned in the Report:
Gulf Pharmaceutical Industries (Julphar)
Bahrain Pharma
United Pharmaceutical Manufacturing Company
Al-Hekma Pharmaceuticals
Badr Pharma
Noor Pharmaceuticals
Gulf Biotech
Bahrain International Investment Park (BIIP) - Pharmaceutical Zone
Al-Muhaidib Pharmaceutical Company
Ameen Al-Fahad Group
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Bahrain Continuous Manufacturing Small Molecule APIs Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Bahrain Continuous Manufacturing Small Molecule APIs Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Bahrain Continuous Manufacturing Small Molecule APIs Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for cost-effective production methods
3.1.2 Technological advancements in continuous manufacturing
3.1.3 Rising focus on regulatory compliance and quality assurance
3.1.4 Expansion of pharmaceutical companies in Bahrain
3.2 Market Challenges
3.2.1 High initial investment costs
3.2.2 Limited skilled workforce in continuous manufacturing
3.2.3 Regulatory hurdles and compliance issues
3.2.4 Competition from traditional batch manufacturing processes
3.3 Market Opportunities
3.3.1 Growing demand for personalized medicine
3.3.2 Potential for export to neighboring GCC markets
3.3.3 Collaborations with research institutions
3.3.4 Adoption of Industry 4.0 technologies
3.4 Market Trends
3.4.1 Shift towards sustainable manufacturing practices
3.4.2 Increasing investment in R&D for small molecule APIs
3.4.3 Integration of AI and machine learning in production
3.4.4 Focus on supply chain resilience and flexibility
3.5 Government Regulation
3.5.1 Implementation of GMP (Good Manufacturing Practices)
3.5.2 Regulatory support for continuous manufacturing initiatives
3.5.3 Incentives for local production of APIs
3.5.4 Compliance with international quality standards
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Bahrain Continuous Manufacturing Small Molecule APIs Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Bahrain Continuous Manufacturing Small Molecule APIs Market Segmentation
8.1 By Manufacturing Process Type
8.1.1 Continuous Flow Reactors
8.1.2 Modular Manufacturing Systems
8.1.3 Integrated Continuous Lines
8.1.4 Others
8.2 By API Category
8.2.1 Generic Small Molecule APIs
8.2.2 Specialty and Niche APIs
8.2.3 Highly Potent APIs (HPAPIs)
8.2.4 Others
8.3 By End-User
8.3.1 Pharmaceutical Manufacturers
8.3.2 Contract Development and Manufacturing Organizations (CDMOs)
8.3.3 Research and Development Institutions
8.3.4 Others
8.4 By Therapeutic Application
8.4.1 Oncology
8.4.2 Cardiovascular Diseases
8.4.3 Neurology and Psychiatry
8.4.4 Infectious Diseases
8.4.5 Others
8.5 By Production Capacity Scale
8.5.1 Pilot Scale (kg-100kg)
8.5.2 Small Scale (100kg-1 ton)
8.5.3 Medium Scale (1-10 tons)
8.5.4 Large Scale (>10 tons)
8.6 By Distribution Channel
8.6.1 Direct Supply to Pharmaceutical Companies
8.6.2 CDMO and Contract Manufacturers
8.6.3 Pharmaceutical Distributors
8.6.4 Others
8.7 By Government Support Mechanism
8.7.1 Government Subsidies and Grants
8.7.2 Tax Incentives and Exemptions
8.7.3 Research and Development Funding
8.7.4 Infrastructure Support Programs
9. Bahrain Continuous Manufacturing Small Molecule APIs Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name and Headquarters Location
9.2.2 Organization Size (Large, Medium, or Small)
9.2.3 Annual Revenue Growth Rate (%)
9.2.4 Continuous Manufacturing Capacity (tons/year)
9.2.5 Market Share in Bahrain Region (%)
9.2.6 Production Efficiency Ratio (Output per unit input)
9.2.7 R&D Investment as % of Revenue
9.2.8 Regulatory Compliance Score (GMP, ISO certifications)
9.2.9 Time-to-Market for New APIs (months)
9.2.10 Customer Retention Rate (%)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Gulf Pharmaceutical Industries (Julphar)
9.5.2 Bahrain Pharma
9.5.3 United Pharmaceutical Manufacturing Company
9.5.4 Al-Hekma Pharmaceuticals
9.5.5 Badr Pharma
9.5.6 Noor Pharmaceuticals
9.5.7 Gulf Biotech
9.5.8 Bahrain International Investment Park (BIIP) - Pharmaceutical Zone
9.5.9 Al-Muhaidib Pharmaceutical Company
9.5.10 Ameen Al-Fahad Group
10. Bahrain Continuous Manufacturing Small Molecule APIs Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Industry and Commerce
10.1.3 Ministry of Finance
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Technology
10.2.1 Pharmaceutical Sector Investments
10.2.2 R&D Expenditure
10.2.3 Technology Upgrades and Equipment
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Cost Management and Operational Efficiency
10.3.2 Quality Assurance and Batch Consistency
10.3.3 Regulatory Compliance and Documentation
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training and Development Needs
10.4.2 Technology Familiarity and Expertise
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics and KPIs
10.5.2 Scalability Potential
10.5.3 User Feedback Mechanisms
10.5.4 Others
11. Bahrain Continuous Manufacturing Small Molecule APIs Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging Considerations
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Industry reports from Bahrain's Ministry of Industry and Commerce
- Market analysis publications from local and regional pharmaceutical associations
- Scientific journals and articles focusing on continuous manufacturing technologies
Primary Research
- Interviews with executives from leading pharmaceutical companies in Bahrain
- Surveys targeting R&D managers and production leads in API manufacturing
- Focus groups with industry experts and regulatory bodies
Validation & Triangulation
- Cross-validation of data from multiple industry sources and expert opinions
- Triangulation of market trends with historical data and future projections
- Sanity checks through peer reviews and expert panel discussions
Phase 2: Market Size Estimation1
Top-down Assessment
- Analysis of Bahrain's overall pharmaceutical market size and growth rates
- Segmentation of the market by therapeutic areas and API types
- Incorporation of government initiatives promoting local manufacturing
Bottom-up Modeling
- Volume estimates based on production capacities of local manufacturers
- Cost analysis derived from operational expenses and pricing strategies
- Estimation of market share based on firm-level sales data
Forecasting & Scenario Analysis
- Multi-variable regression analysis considering market drivers such as healthcare spending
- Scenario planning based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Small Molecule API Manufacturers | 45 | Production Managers, Quality Assurance Heads |
| Regulatory Affairs in Pharmaceuticals | 38 | Regulatory Compliance Officers, Legal Advisors |
| Research & Development in Continuous Manufacturing | 42 | R&D Directors, Process Engineers |
| Supply Chain Management in API Distribution | 50 | Supply Chain Managers, Logistics Coordinators |
| Market Access and Pricing Strategy | 35 | Market Access Managers, Pricing Analysts |
Frequently Asked Questions
What is the current value of the Bahrain Continuous Manufacturing Small Molecule APIs Market?
The Bahrain Continuous Manufacturing Small Molecule APIs Market is valued at approximately USD 165 million, reflecting a five-year historical analysis that highlights the sector's growth driven by demand for efficient manufacturing processes in pharmaceuticals.