Region:Middle East
Author(s):Dev
Product Code:KRAB1935
Pages:82
Published On:January 2026
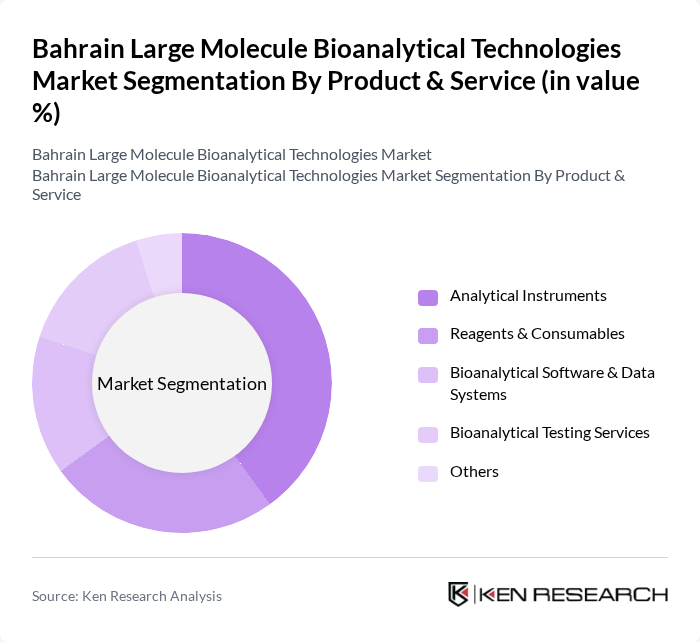
By Product & Service:The product and service segmentation of the market includes various categories such as analytical instruments, reagents and consumables, bioanalytical software and data systems, bioanalytical testing services, and others, which is consistent with global analytical and bioanalytical testing service taxonomies. Among these, analytical instruments are leading due to their essential role in enabling high?sensitivity and high?throughput large molecule assays (for example ligand-binding, LC?MS, flow cytometry, and cell?based potency tests) that are critical for biologics development, method validation, and regulatory compliance.
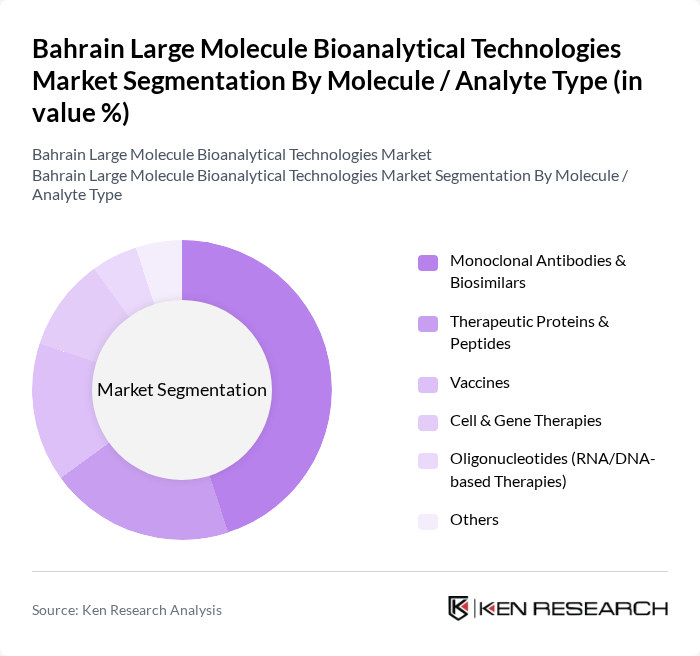
By Molecule / Analyte Type:This segmentation includes monoclonal antibodies and biosimilars, therapeutic proteins and peptides, vaccines, cell and gene therapies, oligonucleotides (RNA/DNA-based therapies), and others, which reflects prevailing global large molecule bioanalytical service categorizations. Monoclonal antibodies and biosimilars dominate this segment due to their extensive use in oncology, autoimmune, and inflammatory diseases, alongside sustained pipeline activity and patent expiries that drive intensive PK, immunogenicity, and comparability testing requirements.
The Bahrain Large Molecule Bioanalytical Technologies Market is characterized by a dynamic mix of regional and international players, consistent with Bahrain’s broader large molecule drug discovery outsourcing and pharmaceutical testing landscape. Leading participants such as Abbott Laboratories, F. Hoffmann-La Roche Ltd (Roche Diagnostics), Thermo Fisher Scientific Inc., Merck KGaA, Bio-Rad Laboratories, Inc., Charles River Laboratories International, Inc., Lonza Group Ltd, Agilent Technologies, Inc., Bruker Corporation, Waters Corporation, SCIEX, Shimadzu Corporation, Eurofins Scientific, IQVIA Inc., and local and regional players (for example, Gulf Biotech, Bahrain Pharma) contribute through provision of analytical platforms, reagents, contract bioanalytical services, clinical trial support, and technology transfer that collectively support large molecule assay development and validation in Bahrain.
The future of the bioanalytical technologies market in Bahrain appears promising, driven by ongoing advancements in technology and increasing healthcare investments. As the demand for biologics continues to rise, local companies are likely to enhance their capabilities through collaborations and technology adoption. Furthermore, the integration of artificial intelligence in bioanalytical processes is expected to streamline operations, improve accuracy, and reduce turnaround times, positioning Bahrain as a competitive player in the regional bioanalytical landscape.
| Segment | Sub-Segments |
|---|---|
| By Product & Service | Analytical Instruments Reagents & Consumables Bioanalytical Software & Data Systems Bioanalytical Testing Services Others |
| By Molecule / Analyte Type | Monoclonal Antibodies & Biosimilars Therapeutic Proteins & Peptides Vaccines Cell & Gene Therapies Oligonucleotides (RNA/DNA-based Therapies) Others |
| By Application | Pharmacokinetics (PK) & Pharmacodynamics (PD) Immunogenicity Testing Biomarker Discovery & Validation Bioavailability & Bioequivalence Studies QC Release & Stability Testing Others |
| By Technology | Ligand Binding Assays (ELISA, ECL, etc.) LC-MS/MS & Hybrid Mass Spectrometry Techniques Chromatography & Electrophoresis Flow Cytometry & Cell-based Assays PCR & NGS-based Quantification Others |
| By Service Workflow | Method Development & Optimization Method Validation Sample Preparation & Handling Sample Analysis & Reporting Bioinformatics & Data Management Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic & Research Institutions Contract Research / Contract Development & Manufacturing Organizations (CROs/CDMOs) Government & Public Health Laboratories Others |
| By Region | Central Bahrain Northern Governorate Southern Governorate Muharraq Governorate |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Development | 100 | Clinical Research Managers, Bioanalytical Scientists |
| Regulatory Compliance | 80 | Regulatory Affairs Specialists, Quality Assurance Managers |
| Biotechnology Research | 70 | Research Scientists, Laboratory Directors |
| Contract Research Organizations (CROs) | 60 | CRO Managers, Business Development Executives |
| Clinical Trial Management | 90 | Clinical Trial Coordinators, Project Managers |
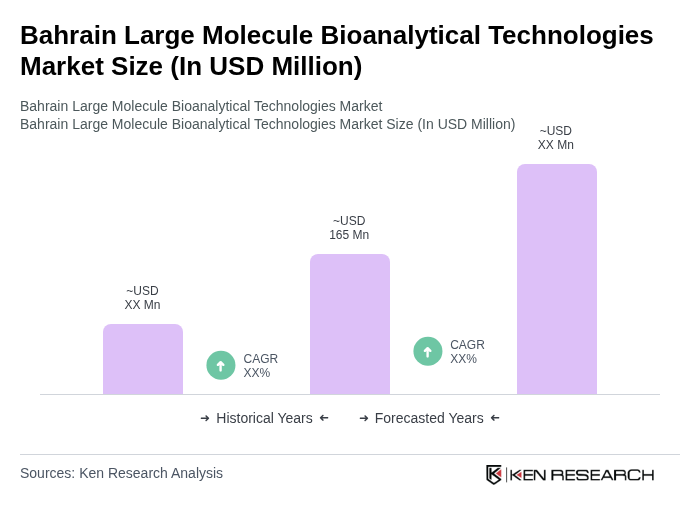
The Bahrain Large Molecule Bioanalytical Technologies Market is valued at approximately USD 165 million, reflecting a growing demand for biologics and advanced therapies in drug development, as well as the adoption of sophisticated bioanalytical methods.