Region:Middle East
Author(s):Dev
Product Code:KRAB1961
Pages:96
Published On:January 2026
By Service Type:The bioanalytical testing services market is segmented into various service types, including Pharmacokinetics (PK) testing, Pharmacodynamics (PD) and biomarker analysis, Immunogenicity and serology testing, Bioavailability and bioequivalence (BA/BE) studies, Cell-based assays and virology testing, Method development, validation, and stability testing, and others. These service categories are consistent with the global and regional structure of the bioanalytical testing services market, which commonly covers ADME/PK, PD, BA/BE, biomarker testing, immunogenicity, and cell-based/virology assays. Among these, Pharmacokinetics (PK) testing is positioned as a leading segment in Oman due to its critical role in drug development, helping to determine the absorption, distribution, metabolism, and excretion of drugs, mirroring its importance at the Middle East & Africa and global levels. The increasing focus on personalized medicine, expansion of clinical trial activity across the Gulf region, and greater outsourcing of analytical work by pharmaceutical and biopharmaceutical companies are further driving the demand for PK testing services.

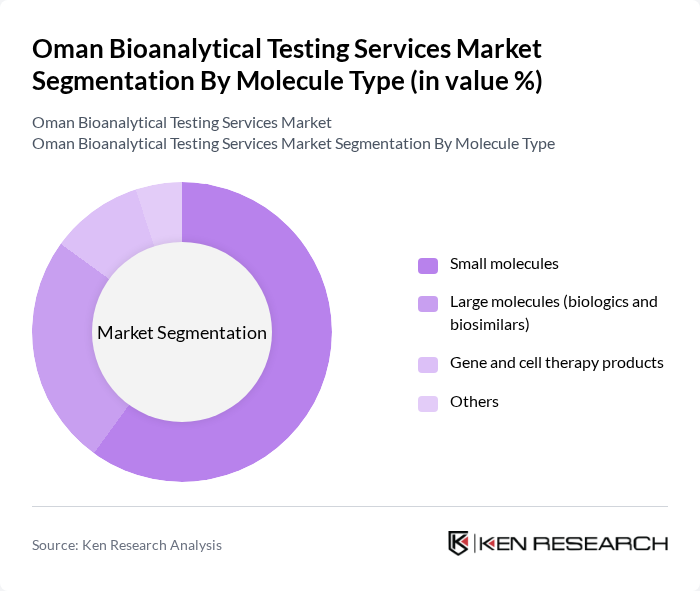
By Molecule Type:The market is also segmented by molecule type, which includes Small molecules, Large molecules (biologics and biosimilars), Gene and cell therapy products, and others. This segmentation aligns with standard global categorizations of bioanalytical testing services by molecule, where small molecules and large molecules (biologics and biosimilars) are the core segments. The small molecules segment dominates the market due to their widespread use in pharmaceuticals and the extensive research and development activities surrounding them, consistent with the finding that small molecules are the largest revenue-generating segment in the Middle East & Africa bioanalytical testing services market. The increasing prevalence of chronic diseases and the need for effective treatment options in Oman and the wider region are driving the demand for small molecule testing services, while interest in biologics, biosimilars, and emerging gene and cell therapies is gradually expanding the large molecule and advanced therapy testing segments.
The Oman Bioanalytical Testing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as Oman Pharmaceuticals and Biological Industries Company (OmanPBI), Oman Pharmaceutical Products Co. LLC (OPP), National Pharmaceutical Industries Co. SAOG (NPI), Sultan Qaboos University – Central Analytical & Research Laboratories, Oman Ministry of Health – Central Public Health Laboratory, Muscat Private Hospital – Laboratory & Diagnostics Services, Starcare Hospital Muscat – Laboratory Services, Badr Al Samaa Group of Hospitals – Laboratory Network, Al Harub Medical Laboratory, Oman International Hospital – Clinical Laboratory, Oman Medical College / University of Buraimi – Research Laboratories, Dhofar University – Biomedical & Health Sciences Labs, Nizwa Hospital – Clinical & Research Laboratory, Royal Hospital, Muscat – Advanced Diagnostics & Research Labs, Private Independent Diagnostic Laboratories and CROs in Oman (Representative Profiles) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the bioanalytical testing services market in Oman appears promising, driven by increasing investments in healthcare and research. The government's commitment to enhancing clinical trial capabilities and fostering innovation is expected to create a conducive environment for market growth. Furthermore, the integration of advanced technologies such as artificial intelligence and machine learning will likely streamline testing processes, improving efficiency and accuracy. As the biopharmaceutical sector expands, the demand for bioanalytical services will continue to rise, presenting significant growth opportunities.
| Segment | Sub-Segments |
|---|---|
| By Service Type | **Pharmacokinetics (PK) testing** **Pharmacodynamics (PD) and biomarker analysis** **Immunogenicity and serology testing** **Bioavailability and bioequivalence (BA/BE) studies** **Cell-based assays and virology testing** **Method development, validation, and stability testing** Others |
| By Molecule Type | **Small molecules** **Large molecules (biologics and biosimilars)** **Gene and cell therapy products** Others |
| By End-User | **Pharmaceutical and biopharmaceutical companies** **Contract Research Organizations (CROs)** **Contract Development and Manufacturing Organizations (CDMOs)** **Academic and research institutions** **Hospitals and clinical laboratories** Others |
| By Application / Disease Area | **Oncology** **Cardiovascular and metabolic disorders** **Infectious diseases and virology** **Neurology and autoimmune disorders** **Respiratory and gastroenterology** Others |
| By Testing Technology | **LC-MS/MS and chromatography-based methods** **Immunoassays (ELISA and related platforms)** **Molecular assays (PCR and sequencing)** **Cell-based and flow cytometry assays** Others |
| By Region | **Muscat** **Dhofar** **Al Batinah** **Al Dakhiliyah** Others |
| By Sponsorship / Investment Source | **Multinational and regional pharmaceutical sponsors** **Government and public sector funding** **Private equity and venture capital** **Public–private partnerships (PPPs)** Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Testing Services | 60 | Laboratory Managers, Quality Assurance Officers |
| Clinical Research Organizations | 50 | Clinical Trial Managers, Regulatory Affairs Specialists |
| Biotechnology Firms | 40 | Research Scientists, Product Development Managers |
| Academic Research Institutions | 40 | Principal Investigators, Research Coordinators |
| Government Health Agencies | 40 | Policy Makers, Health Program Directors |
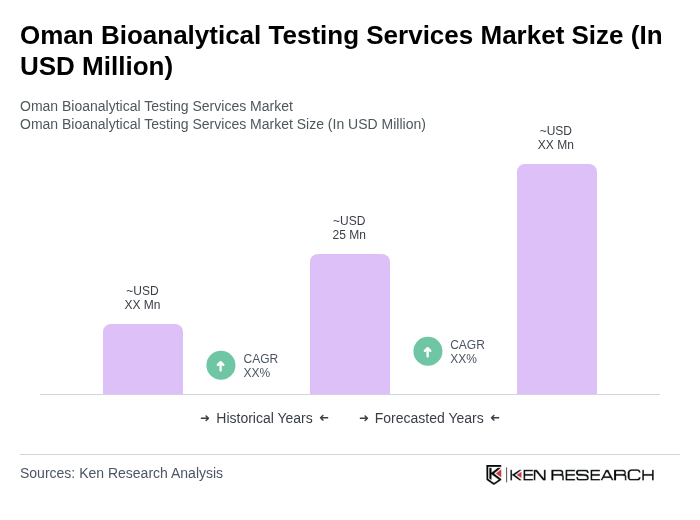
The Oman Bioanalytical Testing Services Market is valued at approximately USD 25 million, reflecting a five-year historical analysis and the country's share within the broader Middle East & Africa bioanalytical testing services market.