About the Report
Base Year 2024Bahrain Noninvasive Prenatal Testing Market Overview
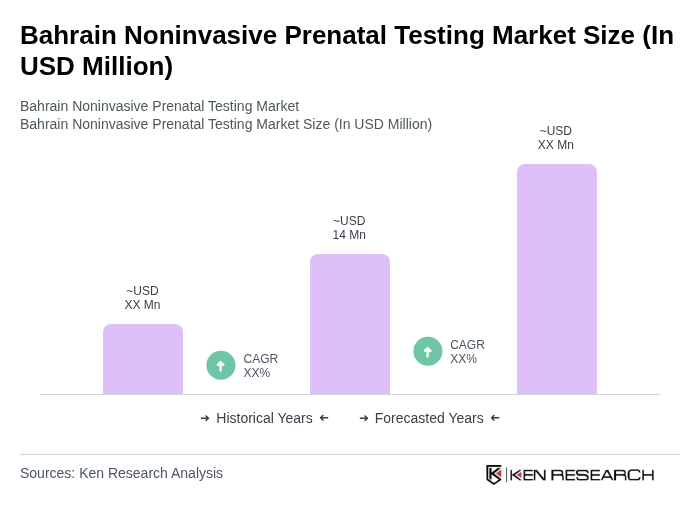
- The Bahrain Noninvasive Prenatal Testing market is valued at USD 14 million, based on a five-year historical analysis and Bahrain’s share within the Middle East and Africa noninvasive prenatal testing market, which generated around USD 132–133 million in revenue regionally. This growth is primarily driven by increasing awareness of prenatal health, advancements in genetic testing technologies, and a rising number of expectant mothers seeking early screening options, in line with broader regional trends of rising maternal age, higher uptake of genetic screening, and rapid adoption of next-generation sequencing-based prenatal tests. The demand for noninvasive prenatal testing (NIPT) has surged as it offers a safer alternative to invasive procedures, with global studies and market analyses highlighting its higher accuracy for common aneuploidies and strong preference among expectant mothers for noninvasive options to improve maternal and fetal health outcomes.
- Key players in this market include Manama, the capital city, which serves as a hub for healthcare services, and other regions like Muharraq and the Southern Governorate. The concentration of advanced medical facilities and specialized clinics in these areas contributes to their dominance in the NIPT market, as they provide comprehensive prenatal care and access to cutting-edge testing technologies, consistent with Bahrain’s broader pattern of centralizing tertiary and specialized healthcare services in Manama and major governorates.
- In 2023, the Bahrain government implemented regulations mandating that all prenatal testing facilities must adhere to strict quality control standards. This regulation aligns with the requirements of the National Health Regulatory Authority (NHRA) under instruments such as the Regulations of Private Health Care Facilities issued by the NHRA in 2016 and subsequent amendments, which require licensed laboratories and diagnostic centers, including those performing prenatal and genetic tests, to comply with defined quality management systems, maintain updated licenses, follow proficiency testing and external quality assessment schemes, and undergo periodic inspections and audits to ensure accuracy, reliability, and patient safety in diagnostic services.
Bahrain Noninvasive Prenatal Testing Market Segmentation
By Test Type:
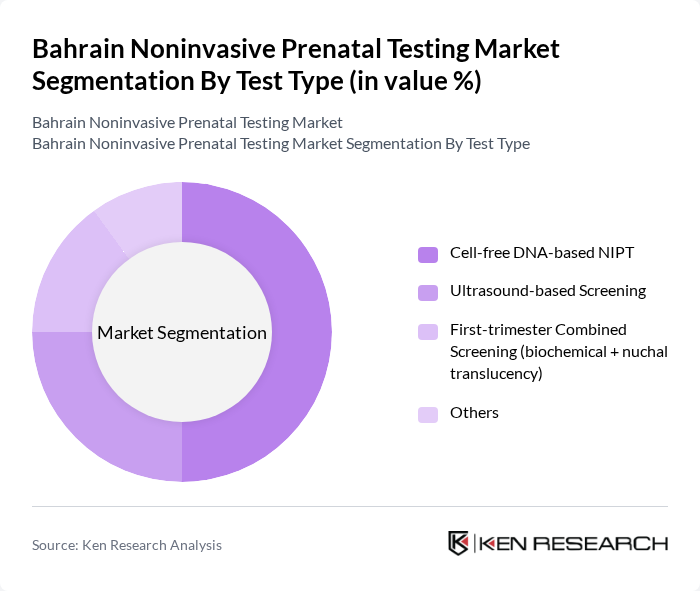
The test type segmentation includes Cell-free DNA-based NIPT, Ultrasound-based Screening, First-trimester Combined Screening (biochemical + nuchal translucency), and Others. Among these, Cell-free DNA-based NIPT is the leading subsegment due to its high accuracy and noninvasive nature, making it the preferred choice for expectant mothers; global and regional market reports indicate that cell-free DNA-based NIPT captures the largest revenue share in prenatal screening because of its superior sensitivity and specificity for trisomies compared with traditional serum and ultrasound screening. The increasing adoption of this technology is driven by its ability to detect chromosomal abnormalities early in pregnancy and by broader trends in the Middle East and Africa toward early genetic risk assessment, improved reimbursement, and greater availability of NGS-based NIPT panels, thus enhancing maternal-fetal health management.
By Technology:
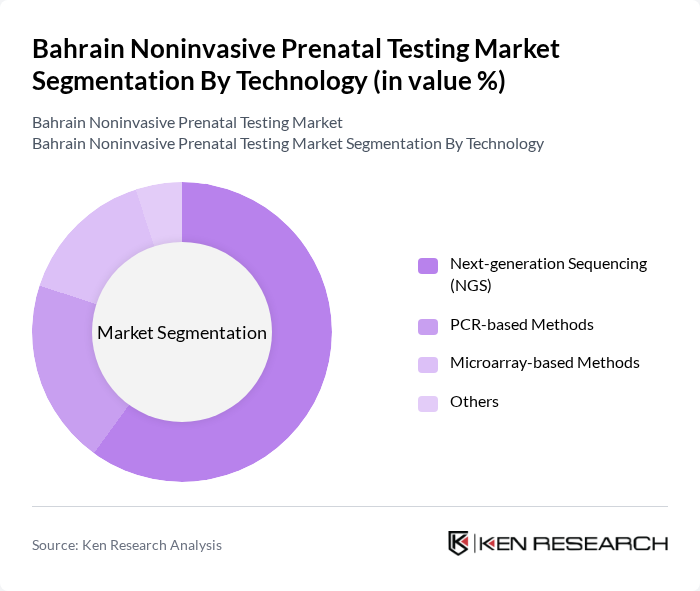
This segmentation includes Next-generation Sequencing (NGS), PCR-based Methods, Microarray-based Methods, and Others. The Next-generation Sequencing (NGS) technology dominates the market due to its ability to analyze multiple genetic markers simultaneously, providing comprehensive results with high sensitivity and specificity; global NIPT market analyses consistently identify NGS-based platforms as the primary technology underpinning commercial NIPT assays. The growing preference for NGS is attributed to its efficiency, scalability, and steadily decreasing costs, which have made high-throughput sequencing more accessible for routine clinical prenatal screening across regions including the Middle East and Africa.
Bahrain Noninvasive Prenatal Testing Market Competitive Landscape
The Bahrain Noninvasive Prenatal Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Natera, Inc., Illumina, Inc., Laboratory Corporation of America Holdings (LabCorp), Quest Diagnostics Incorporated, F. Hoffmann-La Roche Ltd (Roche Diagnostics), PerkinElmer, Inc., Eurofins Scientific, Myriad Genetics, Inc., BGI Genomics Co., Ltd., MedGenome, Fetal Medicine Foundation, Sequenom, Inc., Ambry Genetics, Sema4, and other regional and local NIPT providers in Bahrain contribute to innovation, geographic expansion, and service delivery in this space, reflecting the broad global competitive landscape for NIPT solutions described in international market analyses.
Bahrain Noninvasive Prenatal Testing Market Industry Analysis
Growth Drivers
- Increasing Awareness of Prenatal Health:The awareness of prenatal health in Bahrain has significantly increased, with over 70% of expectant mothers now seeking prenatal care, according to the Ministry of Health. This rise in awareness is attributed to educational campaigns and improved access to information. The government has invested approximately $5 million in maternal health initiatives, which has led to a greater understanding of the importance of early testing and monitoring, thereby driving demand for noninvasive prenatal testing (NIPT).
- Rising Demand for Early Disease Detection:The demand for early disease detection among pregnant women in Bahrain is on the rise, with an estimated 15% increase in NIPT usage reported in future. This trend is driven by the growing prevalence of genetic disorders, with around 1 in 150 births affected by conditions such as Down syndrome. The increasing focus on preventive healthcare is prompting healthcare providers to recommend NIPT as a reliable method for early diagnosis, further propelling market growth.
- Technological Advancements in Testing Methods:Technological advancements in NIPT have led to more accurate and efficient testing methods. In future, the introduction of next-generation sequencing (NGS) technology is expected to enhance the accuracy of tests to over 99%. This innovation is supported by a $2 million investment from local biotech firms, which is expected to improve the reliability of results and reduce turnaround times, making NIPT more appealing to healthcare providers and patients alike.
Market Challenges
- High Cost of Testing Procedures:The high cost of NIPT procedures remains a significant barrier to widespread adoption in Bahrain. The average cost of a single NIPT test is approximately $1,500, which is prohibitive for many families, especially in a country where the average household income is around $30,000 annually. This financial burden limits access to testing, particularly among lower-income populations, hindering overall market growth.
- Limited Access to Healthcare Facilities:Access to healthcare facilities offering NIPT is limited, particularly in rural areas of Bahrain. Approximately 40% of the population lives outside urban centers, where specialized prenatal care is scarce. This geographical disparity results in a significant portion of expectant mothers being unable to access NIPT services, thereby restricting the market's potential and contributing to unequal healthcare outcomes across the region.
Bahrain Noninvasive Prenatal Testing Market Future Outlook
The future of the NIPT market in Bahrain appears promising, driven by ongoing advancements in technology and increasing public awareness of prenatal health. As healthcare infrastructure expands, more facilities are expected to offer NIPT services, enhancing accessibility. Additionally, collaborations with international health organizations are likely to facilitate knowledge transfer and improve testing standards. These developments will create a more favorable environment for the adoption of NIPT, ultimately benefiting maternal and fetal health outcomes in the region.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Bahraini government is investing approximately $10 million in expanding healthcare facilities, which will include prenatal care services. This expansion is expected to increase the availability of NIPT, making it more accessible to a larger segment of the population, particularly in underserved areas.
- Development of Cost-Effective Testing Solutions:There is a growing opportunity for the development of cost-effective NIPT solutions, with local startups focusing on reducing testing costs by up to 30%. This innovation could significantly increase the adoption rate among families, making prenatal testing more affordable and accessible to a broader demographic.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Test Type | Cell-free DNA-based NIPT Ultrasound-based Screening First-trimester Combined Screening (biochemical + nuchal translucency) Others |
| By Technology | Next-generation Sequencing (NGS) PCR-based Methods Microarray-based Methods Others |
| By Clinical Application | Trisomy 21 (Down syndrome) Trisomy 18 and Trisomy 13 Sex Chromosome Aneuploidies Microdeletion and Microduplication Syndromes Others |
| By Gestational Age | –12 Weeks –24 Weeks –36 Weeks Others |
| By End-User | Hospitals & Maternity Hospitals Diagnostic & Reference Laboratories Specialized Fertility & Prenatal Clinics Others |
| By Risk Category | High-Risk Pregnancies Average / Low-Risk Pregnancies Others |
| By Geographic Distribution | Northern Governorate Southern Governorate Capital Governorate Muharraq Governorate |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Bahrain Ministry of Health)
Manufacturers and Producers of Noninvasive Prenatal Testing Kits
Healthcare Providers and Hospitals
Diagnostic Laboratories
Medical Device Distributors
Health Insurance Companies
Pharmaceutical Companies involved in Maternal Health
Players Mentioned in the Report:
Natera, Inc.
Illumina, Inc.
Laboratory Corporation of America Holdings (LabCorp)
Quest Diagnostics Incorporated
F. Hoffmann-La Roche Ltd (Roche Diagnostics)
PerkinElmer, Inc.
Eurofins Scientific
Myriad Genetics, Inc.
BGI Genomics Co., Ltd.
MedGenome
Fetal Medicine Foundation
Sequenom, Inc.
Ambry Genetics
Sema4
Other Regional and Local NIPT Providers in Bahrain
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Bahrain Noninvasive Prenatal Testing Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Bahrain Noninvasive Prenatal Testing Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Bahrain Noninvasive Prenatal Testing Market Analysis
3.1 Growth Drivers
3.1.1 Increasing awareness of prenatal health
3.1.2 Rising demand for early disease detection
3.1.3 Technological advancements in testing methods
3.1.4 Government initiatives promoting maternal health
3.2 Market Challenges
3.2.1 High cost of testing procedures
3.2.2 Limited access to healthcare facilities
3.2.3 Regulatory hurdles in testing approvals
3.2.4 Cultural resistance to prenatal testing
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Collaborations with international health organizations
3.3.3 Development of cost-effective testing solutions
3.3.4 Increasing investment in maternal health programs
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Growth in telemedicine and remote testing
3.4.3 Rising popularity of home-based testing kits
3.4.4 Integration of AI in prenatal testing analysis
3.5 Government Regulation
3.5.1 Guidelines for prenatal testing procedures
3.5.2 Licensing requirements for testing facilities
3.5.3 Quality control standards for testing kits
3.5.4 Data privacy regulations for patient information
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Bahrain Noninvasive Prenatal Testing Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Bahrain Noninvasive Prenatal Testing Market Segmentation
8.1 By Test Type
8.1.1 Cell-free DNA-based NIPT
8.1.2 Ultrasound-based Screening
8.1.3 First-trimester Combined Screening (biochemical + nuchal translucency)
8.1.4 Others
8.2 By Technology
8.2.1 Next-generation Sequencing (NGS)
8.2.2 PCR-based Methods
8.2.3 Microarray-based Methods
8.2.4 Others
8.3 By Clinical Application
8.3.1 Trisomy 21 (Down syndrome)
8.3.2 Trisomy 18 and Trisomy 13
8.3.3 Sex Chromosome Aneuploidies
8.3.4 Microdeletion and Microduplication Syndromes
8.3.5 Others
8.4 By Gestational Age
8.4.1 0–12 Weeks
8.4.2 13–24 Weeks
8.4.3 25–36 Weeks
8.4.4 Others
8.5 By End-User
8.5.1 Hospitals & Maternity Hospitals
8.5.2 Diagnostic & Reference Laboratories
8.5.3 Specialized Fertility & Prenatal Clinics
8.5.4 Others
8.6 By Risk Category
8.6.1 High-Risk Pregnancies
8.6.2 Average / Low-Risk Pregnancies
8.6.3 Others
8.7 By Geographic Distribution
8.7.1 Northern Governorate
8.7.2 Southern Governorate
8.7.3 Capital Governorate
8.7.4 Muharraq Governorate
9. Bahrain Noninvasive Prenatal Testing Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Bahrain NIPT Revenue (USD million)
9.2.3 3-year Revenue CAGR in Bahrain (%)
9.2.4 Test Volume Share (%)
9.2.5 Installed Test Capacity Utilization (%)
9.2.6 Average Realized Price per Test (USD)
9.2.7 Gross Margin (%) on NIPT Portfolio
9.2.8 R&D Spend on NIPT as % of Revenue
9.2.9 Number of NIPT Panels / SKUs Offered
9.2.10 Share of Direct vs Referred Patients (%)
9.2.11 Contracted Accounts (Hospitals / Clinics) – Number
9.2.12 Patient Net Promoter Score (NPS)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Natera, Inc.
9.5.2 Illumina, Inc.
9.5.3 Laboratory Corporation of America Holdings (LabCorp)
9.5.4 Quest Diagnostics Incorporated
9.5.5 F. Hoffmann-La Roche Ltd (Roche Diagnostics)
9.5.6 PerkinElmer, Inc.
9.5.7 Eurofins Scientific
9.5.8 Myriad Genetics, Inc.
9.5.9 BGI Genomics Co., Ltd.
9.5.10 MedGenome
9.5.11 Fetal Medicine Foundation
9.5.12 Sequenom, Inc.
9.5.13 Ambry Genetics
9.5.14 Sema4
9.5.15 Other Regional and Local NIPT Providers in Bahrain
10. Bahrain Noninvasive Prenatal Testing Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation for Prenatal Health
10.1.2 Decision-Making Process
10.1.3 Preferred Suppliers
10.1.4 Evaluation Criteria
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Healthcare Facilities
10.2.2 Funding for Research and Development
10.2.3 Partnerships with Private Sector
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to Quality Testing
10.3.2 Affordability of Services
10.3.3 Awareness and Education
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Technology Acceptance
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Success
10.5.2 Scalability of Solutions
10.5.3 Feedback Mechanisms
10.5.4 Others
11. Bahrain Noninvasive Prenatal Testing Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams
1.4 Cost Structure Analysis
1.5 Key Partnerships
1.6 Customer Segments
1.7 Channels
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of existing market reports and publications on noninvasive prenatal testing (NIPT) in Bahrain
- Review of demographic and health statistics from Bahrain's Ministry of Health and relevant health organizations
- Examination of regulatory frameworks and guidelines pertaining to prenatal testing in Bahrain
Primary Research
- Interviews with obstetricians and gynecologists practicing in Bahrain to understand clinical perspectives on NIPT
- Surveys with laboratory managers and technicians involved in NIPT to gather insights on operational practices
- Focus group discussions with expectant parents to assess awareness and acceptance of NIPT services
Validation & Triangulation
- Cross-validation of findings through comparison with international NIPT market trends and data
- Triangulation of insights from healthcare professionals, laboratory data, and patient feedback
- Sanity checks conducted through expert panel reviews comprising healthcare professionals and market analysts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market for NIPT based on birth rates and prenatal testing uptake in Bahrain
- Segmentation of the market by types of tests offered and demographic factors influencing demand
- Incorporation of healthcare expenditure trends and government health initiatives promoting prenatal care
Bottom-up Modeling
- Collection of data on the number of NIPT tests conducted by leading laboratories in Bahrain
- Analysis of pricing structures for NIPT services across different healthcare providers
- Volume x price calculations to derive revenue estimates for the NIPT market
Forecasting & Scenario Analysis
- Development of forecasting models based on historical growth rates and projected changes in healthcare policies
- Scenario analysis considering factors such as technological advancements and shifts in consumer preferences
- Creation of baseline, optimistic, and pessimistic forecasts for the NIPT market through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Obstetricians and Gynecologists | 60 | Healthcare Providers, Specialists in Prenatal Care |
| Laboratory Technicians and Managers | 50 | Laboratory Staff, Quality Control Managers |
| Expectant Parents | 120 | Parents-to-be, Couples Considering NIPT |
| Healthcare Policy Makers | 40 | Government Officials, Health Program Directors |
| Insurance Providers | 45 | Insurance Analysts, Policy Underwriters |
Frequently Asked Questions
What is the current value of the Bahrain Noninvasive Prenatal Testing market?
The Bahrain Noninvasive Prenatal Testing market is valued at approximately USD 14 million, reflecting its share within the broader Middle East and Africa NIPT market, which generates around USD 132133 million in revenue regionally.