About the Report
Base Year 2024Australia Noninvasive Prenatal Testing Market Overview
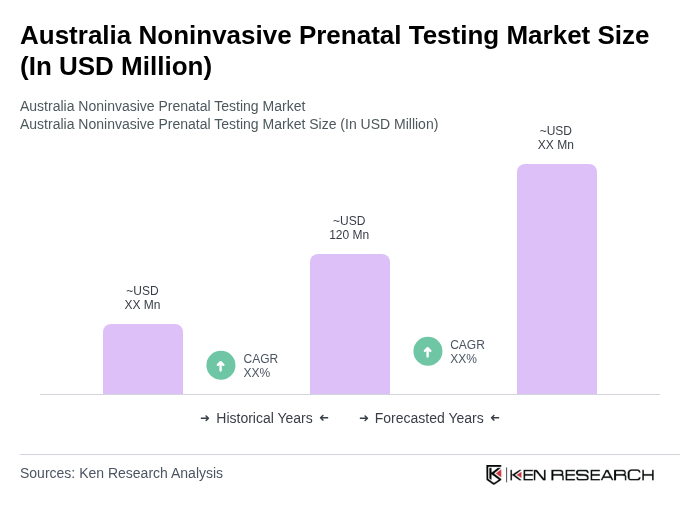
- The Australia Noninvasive Prenatal Testing market is valued at USD 120 million, based on a five-year historical analysis. This growth is primarily driven by increasing awareness of prenatal health, rising maternal age, advancements in genetic testing technologies, and a growing preference among expectant mothers for noninvasive procedures over traditional invasive methods. The market has seen a significant uptick in demand for accurate and early detection of genetic disorders, supported by the wider availability of NIPT in both public and private settings and growing integration into routine prenatal care pathways.
- Key regional contributors in this market include New South Wales, Victoria, and Queensland, which dominate due to their advanced healthcare infrastructure, higher population density, and a greater concentration of hospitals, specialist obstetric practices, and pathology laboratories offering prenatal testing services. These regions benefit from a robust network of public and private healthcare providers and better access to maternal–fetal medicine specialists, making them pivotal in the delivery and uptake of noninvasive prenatal testing services.
- In 2023, quality and safety requirements for prenatal genetic testing services in Australia continued to be governed by national accreditation and regulatory frameworks, including the National Pathology Accreditation Advisory Council (NPAAC) Requirements for Genomic Testing for Heritable Disorders and Variants in Somatic Tumours, 2018, issued by the Australian Government Department of Health and Aged Care. Under this framework, laboratories performing prenatal tests such as NIPT must operate within the National Association of Testing Authorities (NATA)/Royal College of Pathologists of Australasia (RCPA) accreditation system, implement validated assays, participate in external quality assurance programs, and ensure clear reporting standards to support the accuracy and reliability of test results and enhance patient safety.
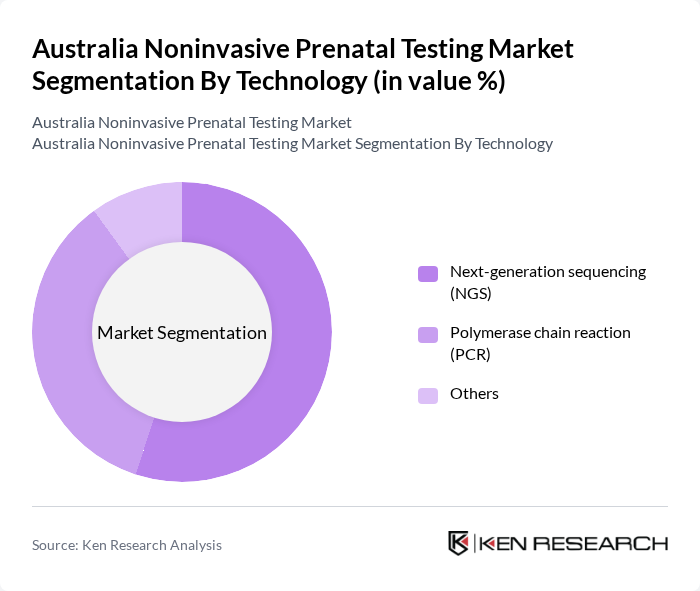
Australia Noninvasive Prenatal Testing Market Segmentation
By Technology:The technology segment of the market includes various methods used for noninvasive prenatal testing. The leading technologies are Next-generation sequencing (NGS), Polymerase chain reaction (PCR), and other emerging technologies. NGS is particularly dominant due to its high accuracy and ability to analyze multiple genetic conditions simultaneously, making it the preferred choice among healthcare providers for trisomy screening and broader genomic assessments.
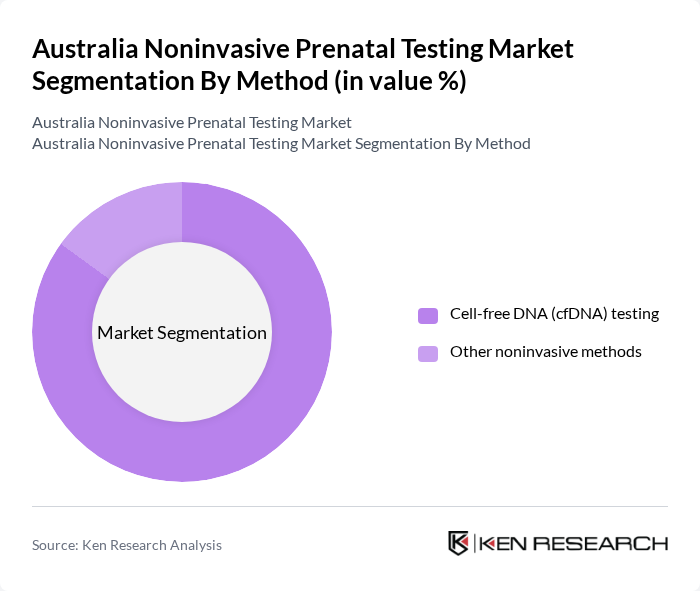
By Method:The method segment encompasses the various approaches to conducting noninvasive prenatal testing. The primary methods include Cell-free DNA (cfDNA) testing and other noninvasive methods. Cell-free DNA testing is the most widely adopted method due to its noninvasive nature, high sensitivity and specificity in detecting common chromosomal abnormalities such as trisomies 21, 18, and 13, and its growing inclusion in clinical guidelines as a first-line screening option for high?risk and, increasingly, average?risk pregnancies.
Australia Noninvasive Prenatal Testing Market Competitive Landscape
The Australia Noninvasive Prenatal Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sonic Healthcare Limited, Genetic Technologies Limited, F. Hoffmann-La Roche Ltd, Illumina, Inc., Natera, Inc., Laboratory Corporation of America Holdings (Labcorp), Quest Diagnostics Incorporated, Eurofins Scientific SE, BGI Genomics Co., Ltd., Fulgent Genetics, Inc., Myriad Genetics, Inc., Monash IVF Group, Virtus Health Limited, Roche Diagnostics Australia Pty Ltd, and other emerging Australia-based NIPT providers contribute to innovation, geographic expansion, and service delivery in this space.
Australia Noninvasive Prenatal Testing Market Industry Analysis
Growth Drivers
- Increasing Awareness of Prenatal Health:The Australian government allocated AUD 1.5 billion in the future to enhance maternal health services, significantly raising public awareness about prenatal health. This funding supports educational campaigns and community outreach programs, leading to a 30% increase in prenatal check-ups. As more expectant mothers become informed about the benefits of noninvasive prenatal testing (NIPT), the demand for these services is expected to rise, driving market growth.
- Rising Demand for Early Disease Detection:In the future, approximately 300,000 births are projected in Australia, with a growing emphasis on early disease detection. The prevalence of genetic disorders, such as Down syndrome, is estimated at 1 in 700 births. This statistic has prompted healthcare providers to recommend NIPT as a reliable screening method, leading to a projected increase in testing uptake by 25% over the next year, thereby boosting market demand.
- Technological Advancements in Testing Methods:The introduction of next-generation sequencing (NGS) technology has revolutionized NIPT, enhancing accuracy rates to over 99%. In the future, the Australian healthcare sector is expected to invest AUD 200 million in advanced testing technologies. This investment is anticipated to improve the efficiency and reliability of NIPT, making it more accessible and appealing to expectant parents, thus driving market growth.
Market Challenges
- High Cost of Testing Procedures:The average cost of NIPT in Australia ranges from AUD 400 to AUD 1,000, which can be prohibitive for many families. With healthcare expenditure per capita projected to reach AUD 7,000 in the future, the high costs associated with NIPT may deter potential users, limiting market penetration. This financial barrier poses a significant challenge to the widespread adoption of noninvasive testing methods.
- Limited Access in Rural Areas:Approximately 30% of Australia's population resides in rural areas, where access to specialized prenatal testing services is limited. In the future, only 50% of rural healthcare facilities are expected to offer NIPT, compared to 90% in urban centers. This disparity in access creates a significant challenge for the market, as many expectant mothers in remote regions may not receive timely testing and care.
Australia Noninvasive Prenatal Testing Market Future Outlook
The future of the Australia noninvasive prenatal testing market appears promising, driven by technological advancements and increasing public awareness. As healthcare providers adopt innovative testing methods, the accuracy and reliability of NIPT will improve, attracting more expectant parents. Additionally, the expansion of telehealth services is expected to enhance access to prenatal care, particularly in underserved areas, fostering greater adoption of NIPT. Overall, these trends indicate a robust growth trajectory for the market in the coming years.
Market Opportunities
- Expansion of Telehealth Services:The rise of telehealth services in Australia presents a significant opportunity for NIPT providers. With an estimated 40% increase in telehealth consultations in the future, expectant mothers can access prenatal testing remotely, improving convenience and accessibility. This trend is likely to drive higher adoption rates of NIPT, particularly among those in rural areas.
- Development of New Testing Technologies:Continuous innovation in genetic testing technologies offers substantial market opportunities. In the future, investments in research and development are projected to exceed AUD 150 million, focusing on enhancing NIPT accuracy and reducing costs. This advancement will likely attract more healthcare providers to offer NIPT, expanding the market and improving patient outcomes.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Technology | Next-generation sequencing (NGS) Polymerase chain reaction (PCR) Others |
| By Method | Cell-free DNA (cfDNA) testing Other noninvasive methods |
| By Application | Trisomy detection (T21, T18, T13) Sex chromosome aneuploidy detection Microdeletion and microduplication detection Other genetic condition screening |
| By Gestational Age | –12 weeks –24 weeks –36 weeks |
| By End User | Hospitals and clinics Diagnostic laboratories Others |
| By Component | Instruments Kits and reagents Services |
| By Geographic Region | New South Wales Victoria Queensland Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Therapeutic Goods Administration, National Health and Medical Research Council)
Manufacturers and Producers of NIPT Kits
Healthcare Providers and Clinics Offering Prenatal Testing
Laboratories Specializing in Genetic Testing
Health Insurance Companies
Industry Associations (e.g., Australian Medical Association)
Pharmaceutical Companies Involved in Prenatal Care
Players Mentioned in the Report:
Sonic Healthcare Limited
Genetic Technologies Limited
F. Hoffmann-La Roche Ltd
Illumina, Inc.
Natera, Inc.
Laboratory Corporation of America Holdings (Labcorp)
Quest Diagnostics Incorporated
Eurofins Scientific SE
BGI Genomics Co., Ltd.
Fulgent Genetics, Inc.
Myriad Genetics, Inc.
Monash IVF Group
Virtus Health Limited
Roche Diagnostics Australia Pty Ltd
Other emerging Australia-based NIPT providers
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Australia Noninvasive Prenatal Testing Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Australia Noninvasive Prenatal Testing Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Australia Noninvasive Prenatal Testing Market Analysis
3.1 Growth Drivers
3.1.1 Increasing awareness of prenatal health
3.1.2 Rising demand for early disease detection
3.1.3 Technological advancements in testing methods
3.1.4 Growing healthcare expenditure
3.2 Market Challenges
3.2.1 High cost of testing procedures
3.2.2 Limited access in rural areas
3.2.3 Regulatory hurdles
3.2.4 Ethical concerns regarding genetic testing
3.3 Market Opportunities
3.3.1 Expansion of telehealth services
3.3.2 Development of new testing technologies
3.3.3 Partnerships with healthcare providers
3.3.4 Increasing government support for maternal health
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Integration of AI in testing processes
3.4.3 Growing focus on noninvasive methods
3.4.4 Rise in consumer demand for genetic counseling
3.5 Government Regulation
3.5.1 Guidelines for prenatal testing
3.5.2 Licensing requirements for testing facilities
3.5.3 Data protection regulations
3.5.4 Reimbursement policies for noninvasive tests
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Australia Noninvasive Prenatal Testing Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Australia Noninvasive Prenatal Testing Market Segmentation
8.1 By Technology
8.1.1 Next-generation sequencing (NGS)
8.1.2 Polymerase chain reaction (PCR)
8.1.3 Others
8.2 By Method
8.2.1 Cell-free DNA (cfDNA) testing
8.2.2 Other noninvasive methods
8.3 By Application
8.3.1 Trisomy detection (T21, T18, T13)
8.3.2 Sex chromosome aneuploidy detection
8.3.3 Microdeletion and microduplication detection
8.3.4 Other genetic condition screening
8.4 By Gestational Age
8.4.1 0–12 weeks
8.4.2 13–24 weeks
8.4.3 25–36 weeks
8.5 By End User
8.5.1 Hospitals and clinics
8.5.2 Diagnostic laboratories
8.5.3 Others
8.6 By Component
8.6.1 Instruments
8.6.2 Kits and reagents
8.6.3 Services
8.7 By Geographic Region
8.7.1 New South Wales
8.7.2 Victoria
8.7.3 Queensland
8.7.4 Others
9. Australia Noninvasive Prenatal Testing Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company name
9.2.2 Australia NIPT revenue (latest year, US$ million)
9.2.3 3-year Australia NIPT revenue CAGR (%)
9.2.4 Australia NIPT market share (%)
9.2.5 Test volume (annual NIPT tests conducted)
9.2.6 Average revenue per test (ARPT)
9.2.7 Gross margin from NIPT business (%)
9.2.8 R&D spend on NIPT as % of revenue
9.2.9 Geographic coverage within Australia (states/territories served)
9.2.10 Payer mix (public vs private reimbursement share)
9.2.11 Turnaround time (median days per test)
9.2.12 Test menu breadth (number of indications covered)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Sonic Healthcare Limited
9.5.2 Genetic Technologies Limited
9.5.3 F. Hoffmann-La Roche Ltd
9.5.4 Illumina, Inc.
9.5.5 Natera, Inc.
9.5.6 Laboratory Corporation of America Holdings (Labcorp)
9.5.7 Quest Diagnostics Incorporated
9.5.8 Eurofins Scientific SE
9.5.9 BGI Genomics Co., Ltd.
9.5.10 Fulgent Genetics, Inc.
9.5.11 Myriad Genetics, Inc.
9.5.12 Monash IVF Group
9.5.13 Virtus Health Limited
9.5.14 Roche Diagnostics Australia Pty Ltd
9.5.15 Other emerging Australia-based NIPT providers
10. Australia Noninvasive Prenatal Testing Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget allocation for prenatal testing
10.1.2 Decision-making processes
10.1.3 Preferred suppliers
10.1.4 Compliance requirements
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in healthcare infrastructure
10.2.2 Funding for research and development
10.2.3 Expenditure on technology upgrades
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to testing services
10.3.2 Affordability of tests
10.3.3 Awareness and education
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Familiarity with noninvasive testing
10.4.2 Willingness to pay for services
10.4.3 Trust in testing accuracy
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of testing outcomes
10.5.2 Expansion into new markets
10.5.3 Long-term cost savings
10.5.4 Others
11. Australia Noninvasive Prenatal Testing Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation analysis
1.5 Competitive landscape assessment
1.6 Key partnerships identification
1.7 Risk assessment
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience definition
2.4 Communication strategy
2.5 Digital marketing approach
2.6 Offline marketing tactics
2.7 Performance metrics
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 Online distribution channels
3.4 Partnerships with healthcare providers
3.5 Logistics and supply chain management
3.6 Inventory management
3.7 Customer service strategies
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing comparison
4.4 Value-based pricing strategies
4.5 Discounts and promotions
4.6 Customer feedback integration
4.7 Pricing strategy adjustments
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
5.3 Product development opportunities
5.4 Service enhancement suggestions
5.5 Market entry strategies
5.6 Customer engagement initiatives
5.7 Future trends forecasting
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer feedback mechanisms
6.4 Community engagement
6.5 Relationship management tools
6.6 Customer education initiatives
6.7 Retention strategies
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Unique selling points
7.4 Customer-centric approach
7.5 Competitive advantages
7.6 Value delivery mechanisms
7.7 Long-term vision alignment
8. Key Activities
8.1 Regulatory compliance
8.2 Branding efforts
8.3 Distribution setup
8.4 Training and development
8.5 Performance monitoring
8.6 Stakeholder engagement
8.7 Continuous improvement processes
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band analysis
9.1.3 Packaging strategies
9.2 Export Entry Strategy
9.2.1 Target countries identification
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
10.5 Risk assessment
10.6 Market entry feasibility
10.7 Strategic partnerships
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines for market entry
11.3 Budget allocation
11.4 Financial projections
11.5 Funding sources
11.6 Cost management strategies
11.7 Milestone tracking
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnership dynamics
12.3 Risk management strategies
12.4 Control mechanisms
12.5 Decision-making processes
12.6 Long-term sustainability
12.7 Exit strategies
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
13.3 Profit margin analysis
13.4 Revenue forecasting
13.5 Cost structure evaluation
13.6 Financial health indicators
13.7 Investment return analysis
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition targets
14.4 Strategic alliances
14.5 Research collaborations
14.6 Technology partners
14.7 Market entry facilitators
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity tracking
15.2.2 Milestone achievements
15.2.3 Performance evaluation
15.2.4 Adjustments and pivots
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of existing market reports and publications on noninvasive prenatal testing (NIPT) in Australia
- Review of government health statistics and demographic data relevant to prenatal care
- Examination of academic journals and clinical studies focusing on NIPT technologies and methodologies
Primary Research
- Interviews with obstetricians and gynecologists to understand clinical adoption rates and patient preferences
- Surveys with laboratory managers and technicians involved in NIPT processing and analysis
- Focus groups with expectant parents to gauge awareness and perceptions of NIPT
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including healthcare providers and industry reports
- Triangulation of qualitative insights from interviews with quantitative data from surveys
- Sanity checks through expert panel reviews comprising healthcare professionals and market analysts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on national birth rates and prenatal testing uptake
- Segmentation of the market by testing type, including genetic screening and diagnostic tests
- Incorporation of trends in healthcare policy and insurance coverage for NIPT
Bottom-up Modeling
- Collection of data on the number of NIPT tests conducted by leading laboratories in Australia
- Estimation of average pricing for NIPT services across different providers
- Calculation of market size based on volume of tests multiplied by average test pricing
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as maternal age trends and technological advancements
- Scenario modeling based on potential changes in healthcare regulations and public awareness campaigns
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Obstetricians and Gynecologists | 100 | Healthcare Providers, Clinical Practitioners |
| Laboratory Technicians and Managers | 80 | Laboratory Staff, Quality Control Managers |
| Expectant Parents | 150 | First-time Parents, Couples Considering NIPT |
| Healthcare Policy Makers | 50 | Government Officials, Health Administrators |
| Genetic Counselors | 70 | Genetic Counselors, Patient Advisors |
Frequently Asked Questions
What is the current value of the Australia Noninvasive Prenatal Testing market?
The Australia Noninvasive Prenatal Testing market is valued at approximately USD 120 million, reflecting a significant growth trend driven by increased awareness of prenatal health and advancements in genetic testing technologies.