Region:Middle East
Author(s):Geetanshi
Product Code:KRAD4872
Pages:95
Published On:December 2025
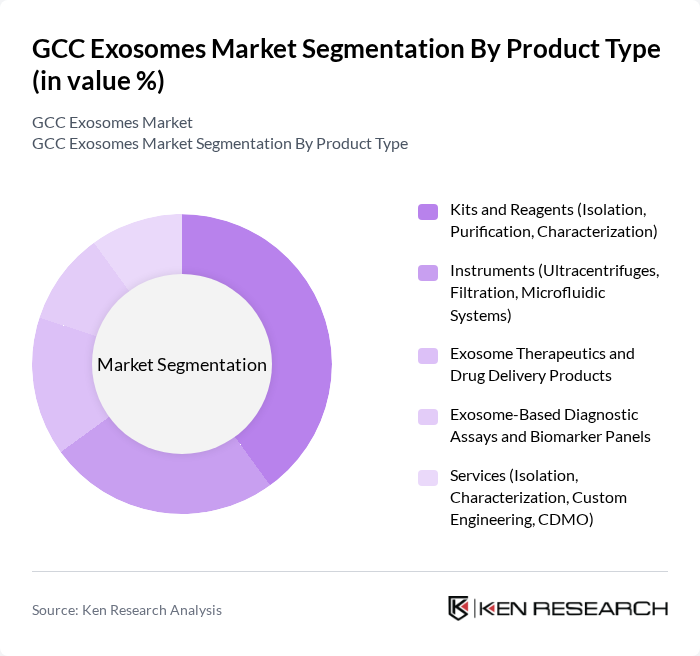
By Product Type:The product type segmentation includes various categories such as kits and reagents, instruments, exosome therapeutics, diagnostic assays, and services. Among these, kits and reagents are currently leading the market due to their essential role in the isolation, purification, and characterization of exosomes, consistent with global trends where kits and reagents represent the largest revenue segment. The increasing demand for research tools in academic and clinical settings drives this segment, as researchers seek reliable and efficient solutions for exosome analysis, including standardized isolation kits, characterization panels, and workflow-compatible reagents that support oncology, neurology, and regenerative medicine studies.
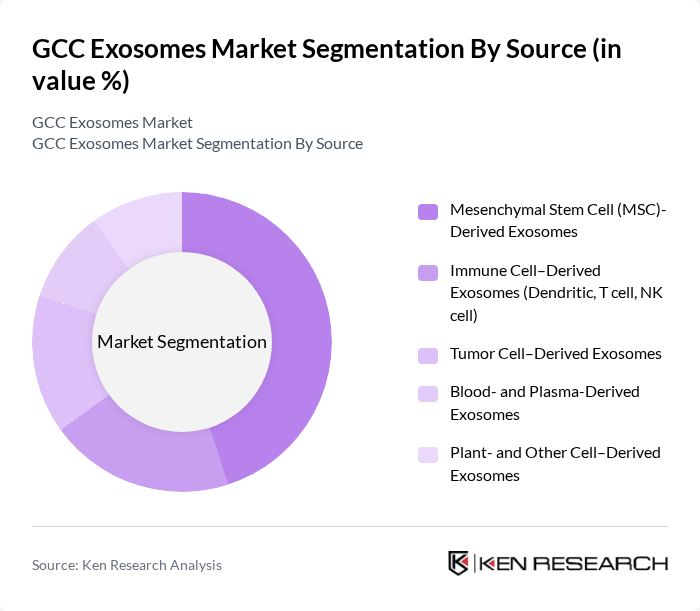
By Source:The source segmentation encompasses various types of exosomes derived from different cell types, including mesenchymal stem cells, immune cells, tumor cells, blood and plasma, and plant-derived exosomes. Mesenchymal stem cell-derived exosomes are currently the dominant source in the market, attributed to their therapeutic potential in regenerative medicine, their role in tissue repair and wound healing, and their ability to modulate immune responses, making them highly sought after in clinical and preclinical applications.
The GCC Exosomes Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific Inc., Danaher Corporation (Beckman Coulter Life Sciences), QIAGEN N.V., Lonza Group Ltd., FUJIFILM Holdings Corporation (FUJIFILM Wako / Irvine Scientific), Sistemic Ltd., System Biosciences, LLC (SBI), AMSBIO (AMS Biotechnology (Europe) Limited), NanoView Biosciences, Exopharm Limited, NurExone Biologic Inc., ExoCoBio Inc., Kimera Labs Inc., ReGenTree FZ-LLC (GCC-based Regenerative Medicine Entity), Dubai Stem Cell Centre (DSCC) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the GCC exosomes market appears promising, driven by ongoing advancements in biotechnology and a growing emphasis on personalized medicine. As healthcare systems increasingly adopt non-invasive diagnostic methods, the demand for exosome applications is expected to rise. Furthermore, collaborations between academic institutions and industry players are likely to accelerate research and development, paving the way for innovative therapies and expanding the market's potential in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Kits and Reagents (Isolation, Purification, Characterization) Instruments (Ultracentrifuges, Filtration, Microfluidic Systems) Exosome Therapeutics and Drug Delivery Products Exosome-Based Diagnostic Assays and Biomarker Panels Services (Isolation, Characterization, Custom Engineering, CDMO) |
| By Source | Mesenchymal Stem Cell (MSC)-Derived Exosomes Immune Cell–Derived Exosomes (Dendritic, T cell, NK cell) Tumor Cell–Derived Exosomes Blood- and Plasma-Derived Exosomes Plant- and Other Cell–Derived Exosomes |
| By Application | Oncology (Diagnosis, Prognosis, Therapy Monitoring) Neurology and Neurodegenerative Diseases Cardiology and Metabolic Disorders Regenerative Medicine and Orthopedics Aesthetic and Dermatology (Skin & Hair Rejuvenation) Infectious and Autoimmune Diseases Others |
| By Workflow | Isolation and Purification Downstream Analysis (Proteomics, Genomics, Lipidomics) Exosome Engineering and Loading GMP-Grade Manufacturing and Quality Control |
| By End-User | Academic and Research Institutes Hospitals and Specialty Clinics Pharmaceutical and Biotechnology Companies Contract Research Organizations (CROs) and CDMOs Cosmetic and Aesthetic Clinics Others |
| By GCC Country | Saudi Arabia United Arab Emirates Qatar Kuwait Oman Bahrain |
| By Research and Clinical Stage | Preclinical Research Early-Phase Clinical Trials (Phase I/II) Late-Phase Clinical Development (Phase III and Pivotal Studies) Commercially Available Products and Services |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Applications of Exosomes | 100 | Clinical Researchers, Medical Directors |
| Exosome-based Diagnostic Tools | 80 | Laboratory Managers, Diagnostic Product Developers |
| Therapeutic Applications in Oncology | 70 | Oncologists, Treatment Protocol Coordinators |
| Regulatory Perspectives on Exosome Products | 60 | Regulatory Affairs Specialists, Compliance Officers |
| Market Trends in Biotech Startups | 90 | Startup Founders, Venture Capital Analysts |
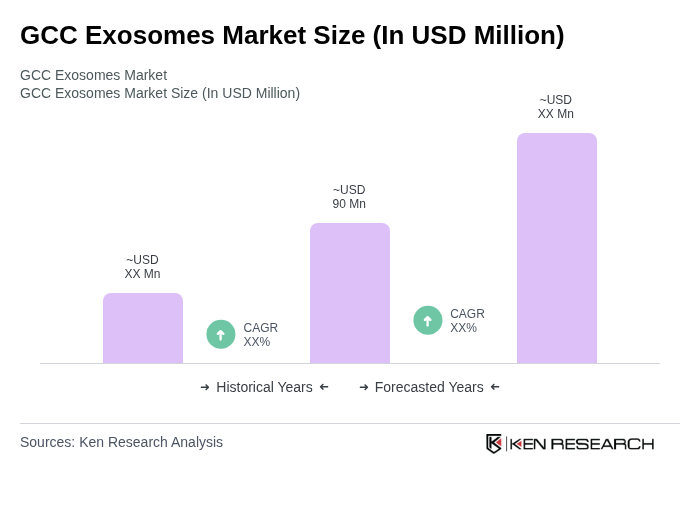
The GCC Exosomes Market is valued at approximately USD 90 million, reflecting a significant share of the global exosomes market. This valuation is based on a five-year historical analysis and highlights the region's growing investment in biotechnology and healthcare innovation.