About the Report
Base Year 2024GCC Healthcare Contract Development Manufacturing Organization Market Overview
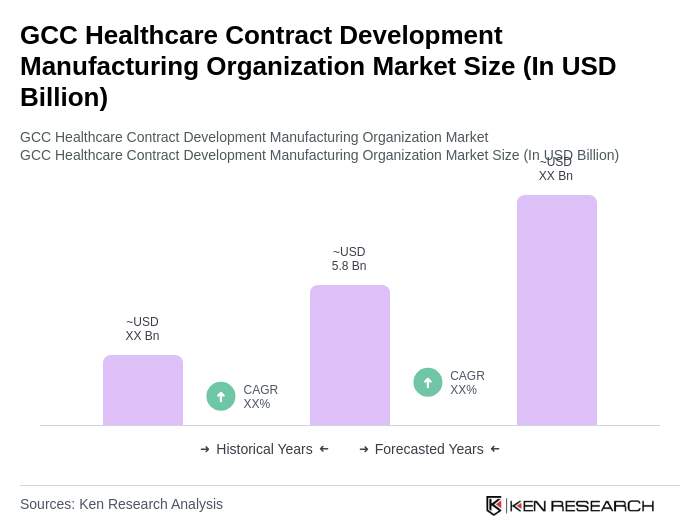
- The GCC Healthcare Contract Development Manufacturing Organization market is valued at USD 5.8 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for outsourcing services in the pharmaceutical and biotechnology sectors, as companies seek to reduce costs and enhance efficiency. The rise in chronic diseases, expansion of the generics market, and the need for innovative therapies have further propelled the market, leading to a surge in contract manufacturing and development activities. Additionally, the adoption of advanced manufacturing technologies and a focus on regulatory compliance are accelerating industry expansion .
- Key players in this market include Saudi Arabia and the United Arab Emirates, which dominate due to their advanced healthcare infrastructure, significant investments in healthcare technology, and supportive government policies. These countries have established themselves as regional hubs for pharmaceutical manufacturing and research, attracting both local and international companies to set up operations in the GCC region. The presence of specialized industrial zones and incentives for pharmaceutical investments further reinforce their leadership .
- In 2023, the Saudi Food and Drug Authority (SFDA) implemented the “Pharmaceutical Products Good Manufacturing Practice Regulations, 2023” issued by the Saudi Food and Drug Authority. This regulation mandates that all contract manufacturers comply with Good Manufacturing Practices (GMP) and undergo regular audits to ensure adherence to international standards. The initiative is designed to bolster consumer confidence and improve the overall quality of healthcare products in the region by enforcing stringent operational, documentation, and quality control requirements .
GCC Healthcare Contract Development Manufacturing Organization Market Segmentation
By Service Type:
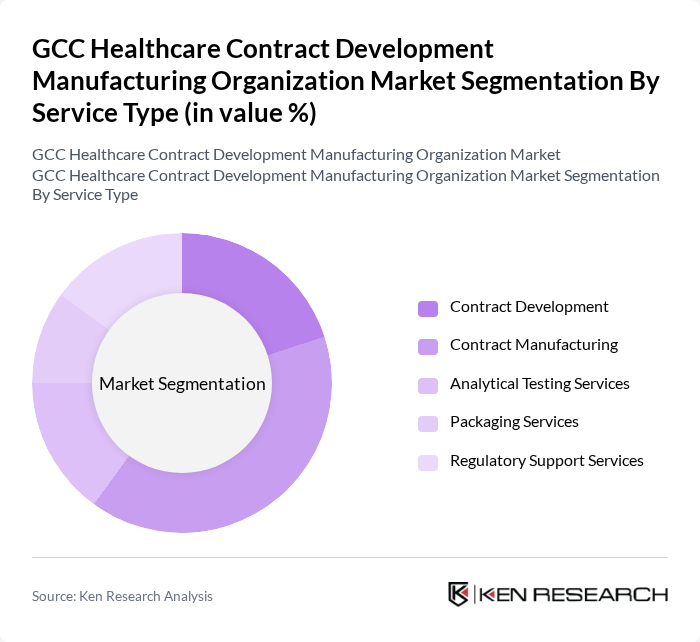
The service type segmentation includes Contract Development, Contract Manufacturing, Analytical Testing Services, Packaging Services, and Regulatory Support Services. Among these, Contract Manufacturing is the leading sub-segment, driven by the increasing trend of outsourcing production to specialized firms. Pharmaceutical companies are increasingly focusing on their core competencies, leading to a higher demand for contract manufacturing services. This trend is further supported by the need for cost-effective solutions, rapid scalability, and access to advanced manufacturing technologies in response to evolving market demands .
By Molecule Type:
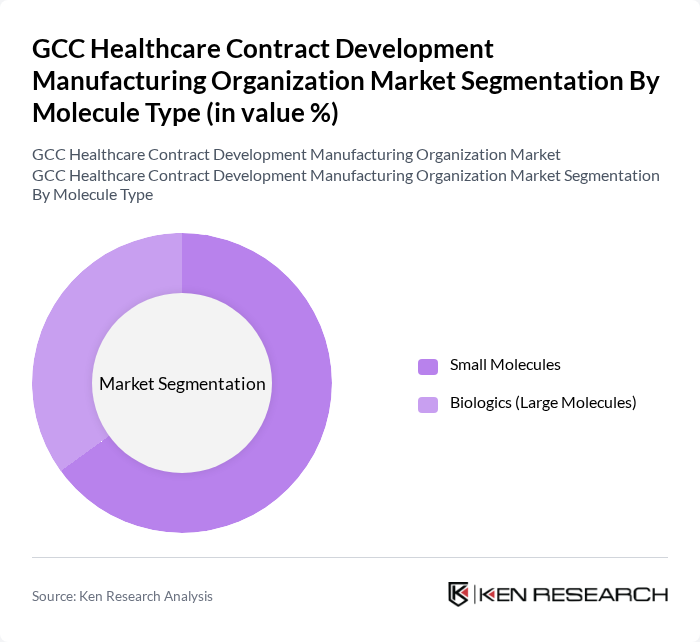
This segmentation includes Small Molecules and Biologics (Large Molecules). The Small Molecules sub-segment is currently dominating the market due to their widespread use in various therapeutic areas, including oncology and cardiovascular diseases. The established manufacturing processes, lower production costs, and robust supply chain infrastructure associated with small molecules make them a preferred choice for many pharmaceutical companies. Additionally, the growing pipeline of small molecule drugs and the continued demand for generic medications are expected to sustain this dominance in the near future .
GCC Healthcare Contract Development Manufacturing Organization Market Competitive Landscape
The GCC Healthcare Contract Development Manufacturing Organization Market is characterized by a dynamic mix of regional and international players. Leading participants such as SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation), Tabuk Pharmaceuticals Manufacturing Company, Julphar (Gulf Pharmaceutical Industries), Neopharma, Globalpharma (Sanofi Gulf), Pharma Solutions Industry (PSI), Jamjoom Pharma, Hikma Pharmaceuticals, Lonza Group AG, Catalent, Inc., Recipharm AB, Aenova Group, Vetter Pharma International GmbH, CordenPharma, and Almac Group contribute to innovation, geographic expansion, and service delivery in this space.
GCC Healthcare Contract Development Manufacturing Organization Market Industry Analysis
Growth Drivers
- Increasing Demand for Biologics:The GCC region is witnessing a significant rise in the demand for biologics, driven by an increase in chronic diseases. In future, the biologics market in the GCC is projected to reach approximately $5.5 billion, reflecting a compound annual growth rate (CAGR) of 8.5%. This surge is attributed to advancements in biotechnology and a growing preference for targeted therapies, which are more effective in treating complex health conditions, thus propelling the need for contract manufacturing services.
- Expansion of Healthcare Infrastructure:The GCC countries are investing heavily in healthcare infrastructure, with an estimated $80 billion allocated for healthcare projects in future. This investment aims to enhance healthcare delivery and accessibility, leading to increased demand for contract development manufacturing organizations (CDMOs). The establishment of new hospitals and clinics is expected to create a robust pipeline for pharmaceutical products, thereby driving the growth of the CDMO market in the region.
- Government Initiatives to Boost Local Manufacturing:Governments in the GCC are implementing policies to promote local manufacturing, with initiatives such as the Saudi Vision 2030 and UAE’s Industrial Strategy 2031. These strategies aim to reduce dependency on imports and enhance self-sufficiency in pharmaceuticals. In future, local manufacturing is expected to account for 45% of the pharmaceutical market, fostering a conducive environment for CDMOs to thrive and innovate in the region.
Market Challenges
- Stringent Regulatory Requirements:The GCC healthcare sector is characterized by stringent regulatory frameworks that govern the manufacturing and distribution of pharmaceuticals. Compliance with Good Manufacturing Practices (GMP) and other regulatory standards can be costly and time-consuming. In future, the average cost of compliance for CDMOs is estimated to be around $1.8 million, which poses a significant barrier to entry for new players in the market.
- High Initial Investment Costs:Establishing a CDMO facility in the GCC requires substantial initial investments, often exceeding $12 million. This includes costs for state-of-the-art equipment, facility construction, and workforce training. The high capital expenditure can deter potential investors, particularly in a market where returns may take several years to materialize, thus limiting the growth potential of new entrants in the industry.
GCC Healthcare Contract Development Manufacturing Organization Market Future Outlook
The future of the GCC healthcare contract development manufacturing organization market appears promising, driven by ongoing investments in healthcare infrastructure and a growing emphasis on local manufacturing. As the demand for personalized medicine increases, CDMOs are likely to adapt their services to meet these evolving needs. Additionally, technological advancements in manufacturing processes will enhance efficiency and reduce costs, positioning the region as a competitive player in the global pharmaceutical landscape.
Market Opportunities
- Growth in Personalized Medicine:The rise of personalized medicine presents a significant opportunity for CDMOs in the GCC. In future, the personalized medicine market is expected to reach $3.5 billion, driven by advancements in genomics and biotechnology. CDMOs can leverage this trend by offering tailored manufacturing solutions that cater to specific patient needs, enhancing their market position.
- Strategic Partnerships with Local Firms:Forming strategic partnerships with local firms can provide CDMOs with access to established distribution networks and market insights. Collaborations are expected to increase by 35% in future, enabling CDMOs to enhance their operational capabilities and expand their market reach, ultimately driving growth in the region.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Service Type | Contract Development Contract Manufacturing Analytical Testing Services Packaging Services Regulatory Support Services |
| By Molecule Type | Small Molecules Biologics (Large Molecules) |
| By Phase | Preclinical Clinical Commercial |
| By End-User | Pharmaceutical Companies Biotechnology Firms Medical Device Companies Research Institutions |
| By Country | Saudi Arabia United Arab Emirates (UAE) Qatar Kuwait Oman Bahrain |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Saudi Food and Drug Authority)
Pharmaceutical Manufacturers
Biotechnology Companies
Medical Device Manufacturers
Healthcare Providers and Hospitals
Supply Chain and Logistics Companies
Insurance Companies and Payers
Players Mentioned in the Report:
SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation)
Tabuk Pharmaceuticals Manufacturing Company
Julphar (Gulf Pharmaceutical Industries)
Neopharma
Globalpharma (Sanofi Gulf)
Pharma Solutions Industry (PSI)
Jamjoom Pharma
Hikma Pharmaceuticals
Lonza Group AG
Catalent, Inc.
Recipharm AB
Aenova Group
Vetter Pharma International GmbH
CordenPharma
Almac Group
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. GCC Healthcare Contract Development Manufacturing Organization Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 GCC Healthcare Contract Development Manufacturing Organization Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. GCC Healthcare Contract Development Manufacturing Organization Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Biologics
3.1.2 Expansion of Healthcare Infrastructure
3.1.3 Rising Prevalence of Chronic Diseases
3.1.4 Government Initiatives to Boost Local Manufacturing
3.2 Market Challenges
3.2.1 Stringent Regulatory Requirements
3.2.2 High Initial Investment Costs
3.2.3 Limited Skilled Workforce
3.2.4 Competition from Established Global Players
3.3 Market Opportunities
3.3.1 Growth in Personalized Medicine
3.3.2 Strategic Partnerships with Local Firms
3.3.3 Technological Advancements in Manufacturing
3.3.4 Expansion into Emerging Markets
3.4 Market Trends
3.4.1 Shift Towards Outsourcing Manufacturing
3.4.2 Increased Focus on Sustainability
3.4.3 Adoption of Advanced Manufacturing Technologies
3.4.4 Growth of Telehealth Services
3.5 Government Regulation
3.5.1 Implementation of GMP Standards
3.5.2 Regulatory Framework for Clinical Trials
3.5.3 Policies Supporting Local Manufacturing
3.5.4 Import Regulations on Pharmaceutical Products
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. GCC Healthcare Contract Development Manufacturing Organization Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. GCC Healthcare Contract Development Manufacturing Organization Market Segmentation
8.1 By Service Type
8.1.1 Contract Development
8.1.2 Contract Manufacturing
8.1.3 Analytical Testing Services
8.1.4 Packaging Services
8.1.5 Regulatory Support Services
8.2 By Molecule Type
8.2.1 Small Molecules
8.2.2 Biologics (Large Molecules)
8.3 By Phase
8.3.1 Preclinical
8.3.2 Clinical
8.3.3 Commercial
8.4 By End-User
8.4.1 Pharmaceutical Companies
8.4.2 Biotechnology Firms
8.4.3 Medical Device Companies
8.4.4 Research Institutions
8.5 By Country
8.5.1 Saudi Arabia
8.5.2 United Arab Emirates (UAE)
8.5.3 Qatar
8.5.4 Kuwait
8.5.5 Oman
8.5.6 Bahrain
9. GCC Healthcare Contract Development Manufacturing Organization Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue (USD Million)
9.2.4 Revenue Growth Rate (%)
9.2.5 Market Share (%)
9.2.6 Number of Manufacturing Facilities (GCC/Global)
9.2.7 Capacity Utilization Rate (%)
9.2.8 R&D Investment as % of Revenue
9.2.9 Time to Market (Months)
9.2.10 Regulatory Compliance Track Record (GMP/EMA/FDA Approvals)
9.2.11 Client Diversification (Top 5 Clients % of Revenue)
9.2.12 Contract Renewal Rate (%)
9.2.13 Quality Audit Pass Rate (%)
9.2.14 Employee Headcount (GCC/Global)
9.2.15 ESG/Sustainability Metrics
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation)
9.5.2 Tabuk Pharmaceuticals Manufacturing Company
9.5.3 Julphar (Gulf Pharmaceutical Industries)
9.5.4 Neopharma
9.5.5 Globalpharma (Sanofi Gulf)
9.5.6 Pharma Solutions Industry (PSI)
9.5.7 Jamjoom Pharma
9.5.8 Hikma Pharmaceuticals
9.5.9 Lonza Group AG
9.5.10 Catalent, Inc.
9.5.11 Recipharm AB
9.5.12 Aenova Group
9.5.13 Vetter Pharma International GmbH
9.5.14 CordenPharma
9.5.15 Almac Group
10. GCC Healthcare Contract Development Manufacturing Organization Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Industry
10.1.3 Ministry of Finance
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Healthcare Facilities
10.2.2 Funding for Research and Development
10.2.3 Expenditure on Medical Equipment
10.3 Pain Point Analysis by End-User Category
10.3.1 Pharmaceutical Companies
10.3.2 Biotech Firms
10.3.3 Research Institutions
10.4 User Readiness for Adoption
10.4.1 Awareness of Contract Manufacturing Benefits
10.4.2 Training and Skill Development Needs
10.4.3 Financial Readiness for Outsourcing
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Cost Savings
10.5.2 Evaluation of Service Quality Improvements
10.5.3 Expansion into New Markets
11. GCC Healthcare Contract Development Manufacturing Organization Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Identification of Market Gaps
1.2 Value Proposition Development
1.3 Revenue Model Structuring
1.4 Key Partnerships and Resources
1.5 Customer Segmentation
1.6 Cost Structure Analysis
1.7 Channels for Market Entry
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Audience Identification
2.4 Communication Strategy
2.5 Digital Marketing Approaches
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 E-commerce Integration
3.4 Logistics and Supply Chain Management
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments Analysis
5.3 Emerging Trends and Needs
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Feedback Mechanisms
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Competitive Advantages
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Solutions
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships Evaluation
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of healthcare market reports from regional health authorities and industry publications
- Review of regulatory frameworks and compliance guidelines from the GCC health ministries
- Examination of existing contracts and agreements within the GCC healthcare sector
Primary Research
- Interviews with executives from leading Contract Development Manufacturing Organizations (CDMOs) in the GCC
- Surveys targeting healthcare procurement officers and supply chain managers
- Field interviews with quality assurance and regulatory affairs professionals in healthcare manufacturing
Validation & Triangulation
- Cross-validation of findings through multiple data sources including industry reports and expert opinions
- Triangulation of market data with insights from healthcare conferences and seminars
- Sanity checks conducted through expert panel reviews and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and growth rates
- Segmentation of the market by therapeutic areas and manufacturing capabilities
- Incorporation of government healthcare initiatives and funding allocations
Bottom-up Modeling
- Collection of data on production capacities and output from key CDMOs in the region
- Operational cost analysis based on service pricing and contract terms
- Volume x cost calculations for various healthcare products and services
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating demographic trends and healthcare demands
- Scenario modeling based on potential regulatory changes and market entry of new players
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturing Contracts | 100 | Procurement Managers, Contract Specialists |
| Biologics and Biosimilars Production | 80 | R&D Directors, Quality Control Managers |
| Medical Device Manufacturing Agreements | 70 | Operations Managers, Regulatory Affairs Officers |
| Clinical Trial Supply Chain Management | 50 | Clinical Operations Managers, Supply Chain Managers |
| Healthcare Product Packaging Solutions | 90 | Packaging Engineers, Product Development Managers |
Frequently Asked Questions
What is the current value of the GCC Healthcare Contract Development Manufacturing Organization market?
The GCC Healthcare Contract Development Manufacturing Organization market is valued at approximately USD 5.8 billion, driven by increasing demand for outsourcing services in the pharmaceutical and biotechnology sectors, as well as the rise in chronic diseases and the expansion of the generics market.