Region:Middle East
Author(s):Rebecca
Product Code:KRAC8561
Pages:80
Published On:November 2025
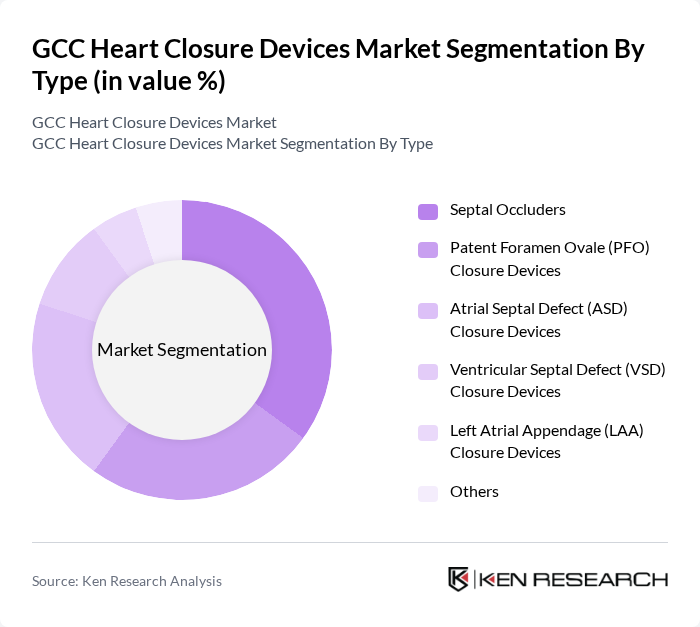
By Type:The market is segmented into various types of heart closure devices, including Septal Occluders, Patent Foramen Ovale (PFO) Closure Devices, Atrial Septal Defect (ASD) Closure Devices, Ventricular Septal Defect (VSD) Closure Devices, Left Atrial Appendage (LAA) Closure Devices, and Others. Among these,Septal Occludersare the most widely used due to their proven effectiveness in treating atrial septal defects, which are among the most common congenital heart conditions. The increasing preference for minimally invasive, catheter-based procedures and the availability of advanced occluder materials have further propelled the demand for these devices .
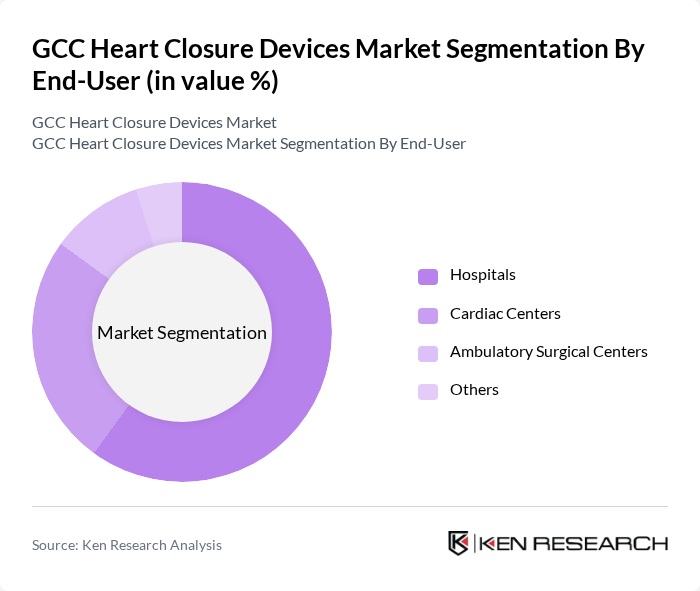
By End-User:The end-user segmentation includes Hospitals, Cardiac Centers, Ambulatory Surgical Centers, and Others.Hospitalsare the leading end-users of heart closure devices, primarily due to their comprehensive cardiac care facilities, access to advanced imaging and interventional technologies, and the availability of specialized medical staff. The increasing number of cardiac interventions performed in hospitals, coupled with the rising patient population and the expansion of tertiary care centers, has significantly contributed to the growth of this segment .
The GCC Heart Closure Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Boston Scientific Corporation, Medtronic plc, Edwards Lifesciences Corporation, Gore Medical, St. Jude Medical, Inc., AGA Medical Corporation, Terumo Corporation, LivaNova PLC, Cardia, Inc., Merit Medical Systems, Inc., Biotronik SE & Co. KG, Cook Medical, Philips Healthcare, SentreHEART, Inc., MicroPort Scientific Corporation, W.L. Gore & Associates, Teleflex Incorporated, B. Braun Melsungen AG, Stryker Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the GCC heart closure devices market appears promising, driven by ongoing technological advancements and increasing healthcare investments. As the region's healthcare infrastructure expands, the adoption of minimally invasive procedures is likely to rise, enhancing patient outcomes. Furthermore, the integration of digital health technologies will facilitate better monitoring and management of heart conditions, paving the way for innovative solutions that cater to the growing demand for effective heart closure devices in the GCC.
| Segment | Sub-Segments |
|---|---|
| By Type | Septal Occluders Patent Foramen Ovale (PFO) Closure Devices Atrial Septal Defect (ASD) Closure Devices Ventricular Septal Defect (VSD) Closure Devices Left Atrial Appendage (LAA) Closure Devices Others |
| By End-User | Hospitals Cardiac Centers Ambulatory Surgical Centers Others |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients Others |
| By Procedure Type | Elective Procedures Emergency Procedures Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Geography | Saudi Arabia UAE Qatar Kuwait Oman Bahrain |
| By Policy Support | Government Subsidies Tax Incentives Regulatory Support Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiologist Insights | 100 | Interventional Cardiologists, Electrophysiologists |
| Hospital Procurement Analysis | 70 | Procurement Managers, Supply Chain Managers |
| Clinical Research Feedback | 50 | Clinical Researchers, Medical Device Evaluators |
| Patient Experience Surveys | 60 | Patients with Heart Conditions, Caregivers |
| Regulatory Insights | 40 | Regulatory Affairs Specialists, Compliance Officers |
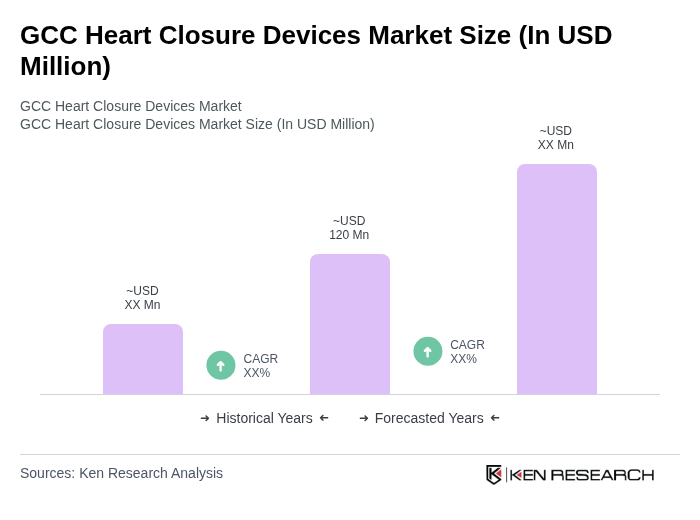
The GCC Heart Closure Devices Market is valued at approximately USD 120 million, driven by factors such as the rising prevalence of congenital heart defects and advancements in device technology, which enhance patient safety and efficacy.