About the Report
Base Year 2024GCC Real World Evidence Solutions Market Overview
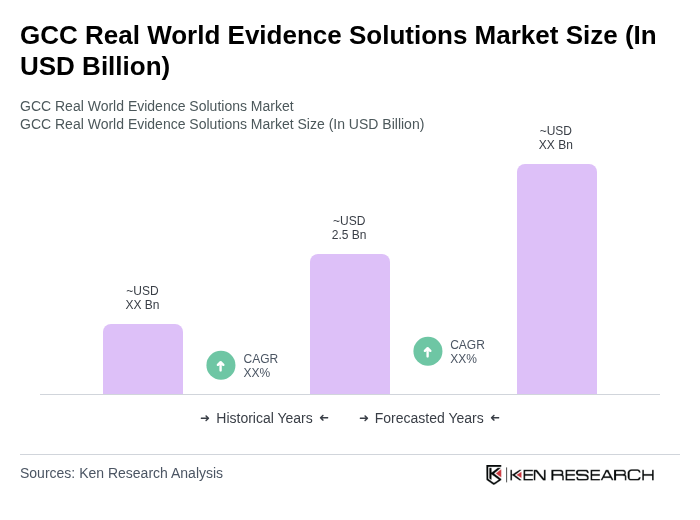
- The GCC Real World Evidence Solutions Market is valued at USD 2.5 billion, based on a five-year historical analysis. This reflects the region's rapid adoption of data-driven healthcare strategies and aligns with the latest global market sizing for real world evidence solutions, which is estimated between USD 2.3 billion and USD 3.3 billion for comparable regions and market segments. Growth is primarily driven by the increasing demand for data-backed insights in healthcare, the rising prevalence of chronic diseases such as diabetes and cardiovascular conditions, and the need for cost-effective healthcare delivery. The integration of advanced technologies—including artificial intelligence, big data analytics, and digital health platforms—continues to propel market expansion, enabling stakeholders to optimize clinical outcomes and operational efficiency.
- Key players in this market includeSaudi Arabiaand theUnited Arab Emirates (UAE), which dominate due to robust healthcare infrastructure, substantial investments in health technology, and a strategic focus on personalized medicine. Both countries have accelerated digital transformation in healthcare, with major pharmaceutical and biotechnology companies leveraging real-world evidence to enhance drug development, regulatory submissions, and market access strategies. The region's leadership is further supported by government initiatives promoting health data interoperability and clinical research partnerships.
- In 2023, the UAE government enacted theMinisterial Decision No. 28 of 2023 on the Use of Real-World Data in Clinical Trials and Post-Market Surveillance, issued by theMinistry of Health and Prevention (MOHAP). This regulation mandates the integration of real-world evidence in clinical trials and post-market surveillance for pharmaceuticals and medical devices. It requires healthcare providers and sponsors to utilize comprehensive data analytics, including electronic health records and patient registries, to improve patient safety and treatment outcomes. The regulation establishes operational standards for data collection, reporting, and compliance, thereby enhancing the overall quality of healthcare services across the UAE and setting a precedent for the broader GCC region.
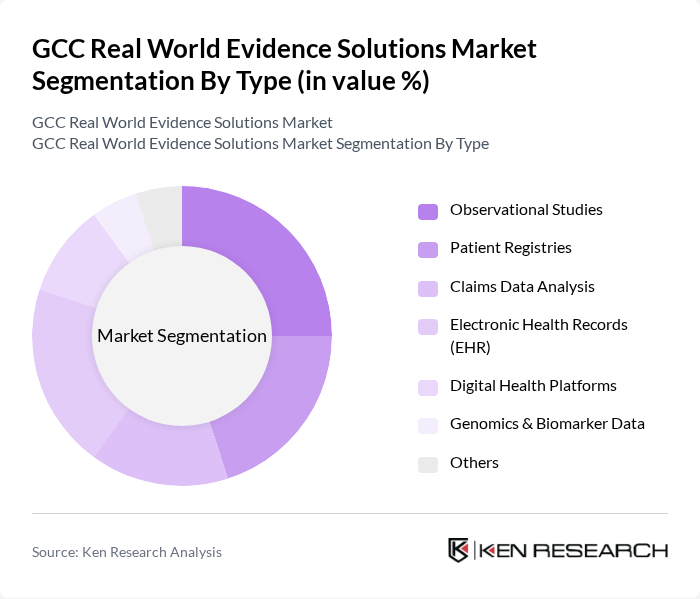
GCC Real World Evidence Solutions Market Segmentation
By Type:The market is segmented into Observational Studies, Patient Registries, Claims Data Analysis, Electronic Health Records (EHR), Digital Health Platforms, Genomics & Biomarker Data, and Others. Each sub-segment plays a pivotal role in generating actionable insights for healthcare decision-making. Observational studies and patient registries are widely used for post-market surveillance and outcomes research, while claims data analysis and EHRs support real-time monitoring and population health management. Digital health platforms and genomics data are increasingly utilized for precision medicine and advanced analytics.
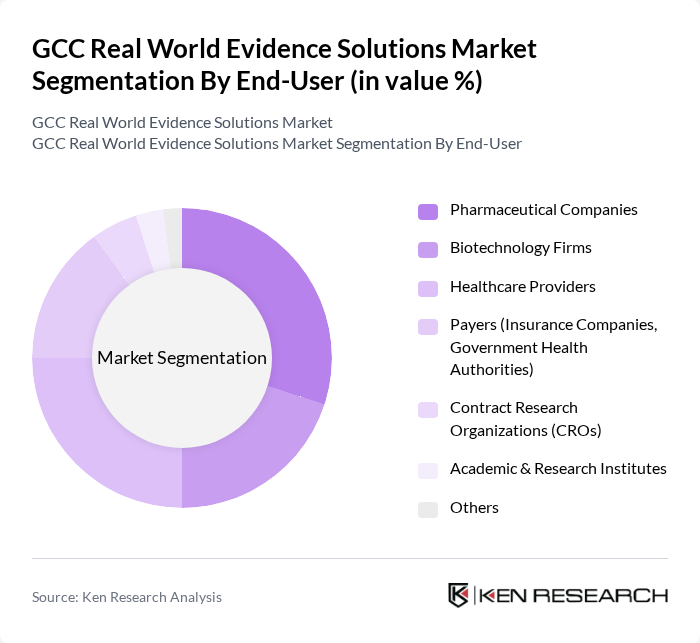
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Healthcare Providers, Payers (Insurance Companies, Government Health Authorities), Contract Research Organizations (CROs), Academic & Research Institutes, and Others. Pharmaceutical and biotechnology firms are the largest adopters, leveraging real-world evidence for regulatory submissions and market access. Healthcare providers utilize RWE for clinical decision support and quality improvement, while payers and government authorities increasingly rely on RWE for reimbursement and policy decisions. CROs and academic institutes contribute through research collaborations and data analytics services.
GCC Real World Evidence Solutions Market Competitive Landscape
The GCC Real World Evidence Solutions Market is characterized by a dynamic mix of regional and international players. Leading participants such as IQVIA, Optum, Syneos Health, Parexel International, Medpace, Labcorp (formerly Covance), PPD (part of Thermo Fisher Scientific), Oracle Health (formerly Cerner Corporation), Flatiron Health, Aetion, Evidera (a PPD business, part of Thermo Fisher Scientific), HealthCore, Cegedim Health Data, ICON plc, Verily Life Sciences contribute to innovation, geographic expansion, and service delivery in this space.
GCC Real World Evidence Solutions Market Industry Analysis
Growth Drivers
- Increasing Demand for Patient-Centric Data:The GCC region is witnessing a significant shift towards patient-centric healthcare, with a projected increase in patient engagement initiatives. In future, healthcare expenditure in the GCC is expected to reach approximately $104.6 billion, driven by the need for personalized treatment plans. This demand is further supported by the World Health Organization's emphasis on integrating patient feedback into healthcare systems, which is anticipated to enhance the quality of care and improve health outcomes across the region.
- Advancements in Data Analytics Technologies:The rapid evolution of data analytics technologies is transforming the healthcare landscape in the GCC. In future, the healthcare analytics market in the region is projected to exceed $1.8 billion, fueled by innovations in artificial intelligence and machine learning. These technologies enable healthcare providers to analyze vast amounts of patient data efficiently, leading to improved decision-making and enhanced patient outcomes, thereby driving the adoption of real-world evidence solutions.
- Rising Focus on Value-Based Healthcare:The GCC countries are increasingly adopting value-based healthcare models, which prioritize patient outcomes over service volume. In future, it is estimated that approximately 35% of healthcare providers in the region will implement value-based care initiatives. This shift is supported by government policies aimed at improving healthcare quality and efficiency, encouraging the use of real-world evidence to assess treatment effectiveness and optimize resource allocation in healthcare systems.
Market Challenges
- Data Privacy and Security Concerns:As the adoption of real-world evidence solutions increases, so do concerns regarding data privacy and security. In future, it is projected that data breaches in the healthcare sector could cost the GCC region over $1 billion. This financial impact, coupled with stringent data protection regulations, poses a significant challenge for healthcare organizations seeking to implement real-world evidence solutions while ensuring compliance and safeguarding patient information.
- Integration Issues with Existing Systems:Many healthcare providers in the GCC face challenges integrating new real-world evidence solutions with their existing IT infrastructure. In future, it is estimated that around 55% of healthcare organizations will struggle with interoperability issues, leading to inefficiencies and increased operational costs. This challenge hinders the seamless flow of data and limits the potential benefits of real-world evidence in enhancing patient care and operational efficiency.
GCC Real World Evidence Solutions Market Future Outlook
The future of the GCC real-world evidence solutions market appears promising, driven by technological advancements and a growing emphasis on patient-centric care. As healthcare providers increasingly adopt digital health solutions, the integration of artificial intelligence and real-time data analytics will enhance decision-making processes. Furthermore, collaboration between healthcare organizations and academic institutions is expected to foster innovation, leading to improved patient outcomes and more efficient healthcare delivery systems across the region.
Market Opportunities
- Expansion of Digital Health Solutions:The digital health market in the GCC is projected to reach approximately $4.5 billion in future, presenting significant opportunities for real-world evidence solutions. This growth is driven by increased smartphone penetration and the rising adoption of telehealth services, enabling healthcare providers to collect and analyze patient data more effectively.
- Collaborations with Academic Institutions:Partnerships between healthcare organizations and academic institutions are expected to grow, with over 25 collaborative projects anticipated in future. These collaborations will facilitate research and development in real-world evidence methodologies, enhancing the quality of healthcare services and fostering innovation in treatment approaches across the GCC.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Observational Studies Patient Registries Claims Data Analysis Electronic Health Records (EHR) Digital Health Platforms Genomics & Biomarker Data Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Healthcare Providers Payers (Insurance Companies, Government Health Authorities) Contract Research Organizations (CROs) Academic & Research Institutes Others |
| By Application | Drug Development & Approvals Market Access & Reimbursement Post-Market Surveillance & Safety Monitoring Health Economics and Outcomes Research (HEOR) Epidemiology Studies Personalized & Precision Medicine Others |
| By Data Source | Clinical Trials Patient Surveys & Reported Outcomes Wearable Devices & Remote Monitoring Social Media & Digital Health Analytics Claims & Billing Data Genomic Data Others |
| By Region | Saudi Arabia United Arab Emirates (UAE) Qatar Kuwait Oman Bahrain |
| By Technology | Data Analytics Tools Machine Learning & AI Algorithms Cloud Computing Solutions Data Visualization & Reporting Software Blockchain & Data Security Solutions Others |
| By Policy Support | Government Grants Tax Incentives Research Funding Regulatory Support Initiatives Public-Private Partnerships Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Gulf Cooperation Council, Ministry of Health and Prevention)
Pharmaceutical and Biotechnology Companies
Healthcare Providers and Hospitals
Health Insurance Companies
Clinical Research Organizations
Data Analytics and Technology Firms
Public Health Agencies
Players Mentioned in the Report:
IQVIA
Optum
Syneos Health
Parexel International
Medpace
Labcorp (formerly Covance)
PPD (part of Thermo Fisher Scientific)
Oracle Health (formerly Cerner Corporation)
Flatiron Health
Aetion
Evidera (a PPD business, part of Thermo Fisher Scientific)
HealthCore
Cegedim Health Data
ICON plc
Verily Life Sciences
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. GCC Real World Evidence Solutions Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 GCC Real World Evidence Solutions Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. GCC Real World Evidence Solutions Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Patient-Centric Data
3.1.2 Advancements in Data Analytics Technologies
3.1.3 Rising Focus on Value-Based Healthcare
3.1.4 Government Initiatives Supporting RWE Adoption
3.2 Market Challenges
3.2.1 Data Privacy and Security Concerns
3.2.2 Limited Awareness Among Stakeholders
3.2.3 Integration Issues with Existing Systems
3.2.4 Regulatory Hurdles in Data Utilization
3.3 Market Opportunities
3.3.1 Expansion of Digital Health Solutions
3.3.2 Collaborations with Academic Institutions
3.3.3 Growth in Personalized Medicine
3.3.4 Increasing Investment in Healthcare IT
3.4 Market Trends
3.4.1 Shift Towards Real-Time Data Collection
3.4.2 Rise of Artificial Intelligence in RWE
3.4.3 Emphasis on Patient Engagement Platforms
3.4.4 Adoption of Blockchain for Data Integrity
3.5 Government Regulation
3.5.1 Data Protection Laws and Compliance
3.5.2 Guidelines for RWE in Clinical Trials
3.5.3 Policies Promoting Health Data Sharing
3.5.4 Regulatory Framework for Digital Health Solutions
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. GCC Real World Evidence Solutions Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. GCC Real World Evidence Solutions Market Segmentation
8.1 By Type
8.1.1 Observational Studies
8.1.2 Patient Registries
8.1.3 Claims Data Analysis
8.1.4 Electronic Health Records (EHR)
8.1.5 Digital Health Platforms
8.1.6 Genomics & Biomarker Data
8.1.7 Others
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Biotechnology Firms
8.2.3 Healthcare Providers
8.2.4 Payers (Insurance Companies, Government Health Authorities)
8.2.5 Contract Research Organizations (CROs)
8.2.6 Academic & Research Institutes
8.2.7 Others
8.3 By Application
8.3.1 Drug Development & Approvals
8.3.2 Market Access & Reimbursement
8.3.3 Post-Market Surveillance & Safety Monitoring
8.3.4 Health Economics and Outcomes Research (HEOR)
8.3.5 Epidemiology Studies
8.3.6 Personalized & Precision Medicine
8.3.7 Others
8.4 By Data Source
8.4.1 Clinical Trials
8.4.2 Patient Surveys & Reported Outcomes
8.4.3 Wearable Devices & Remote Monitoring
8.4.4 Social Media & Digital Health Analytics
8.4.5 Claims & Billing Data
8.4.6 Genomic Data
8.4.7 Others
8.5 By Region
8.5.1 Saudi Arabia
8.5.2 United Arab Emirates (UAE)
8.5.3 Qatar
8.5.4 Kuwait
8.5.5 Oman
8.5.6 Bahrain
8.6 By Technology
8.6.1 Data Analytics Tools
8.6.2 Machine Learning & AI Algorithms
8.6.3 Cloud Computing Solutions
8.6.4 Data Visualization & Reporting Software
8.6.5 Blockchain & Data Security Solutions
8.6.6 Others
8.7 By Policy Support
8.7.1 Government Grants
8.7.2 Tax Incentives
8.7.3 Research Funding
8.7.4 Regulatory Support Initiatives
8.7.5 Public-Private Partnerships
8.7.6 Others
9. GCC Real World Evidence Solutions Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Regional Presence (GCC Coverage, Local Partnerships)
9.2.4 Revenue Growth Rate (GCC Operations)
9.2.5 Number of Active RWE Projects in GCC
9.2.6 Market Penetration Rate (GCC Healthcare Institutions Served)
9.2.7 Customer Retention Rate
9.2.8 Average Deal Size (GCC Contracts)
9.2.9 Strategic Partnerships (with Ministries, Hospitals, Pharma)
9.2.10 Compliance with GCC Data Regulations
9.2.11 Technology Adoption Level (AI, Cloud, Interoperability)
9.2.12 Customer Satisfaction Score (GCC Clients)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 IQVIA
9.5.2 Optum
9.5.3 Syneos Health
9.5.4 Parexel International
9.5.5 Medpace
9.5.6 Labcorp (formerly Covance)
9.5.7 PPD (part of Thermo Fisher Scientific)
9.5.8 Oracle Health (formerly Cerner Corporation)
9.5.9 Flatiron Health
9.5.10 Aetion
9.5.11 Evidera (a PPD business, part of Thermo Fisher Scientific)
9.5.12 HealthCore
9.5.13 Cegedim Health Data
9.5.14 ICON plc
9.5.15 Verily Life Sciences
10. GCC Real World Evidence Solutions Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Vendors
10.1.4 Evaluation Criteria
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Priorities
10.2.2 Spending Patterns
10.2.3 Impact of Economic Conditions
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges Faced by Healthcare Providers
10.3.2 Issues Encountered by Payers
10.3.3 Concerns of Pharmaceutical Companies
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Technology Adoption Barriers
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Metrics for Success
10.5.2 Future Use Cases
11. GCC Real World Evidence Solutions Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from healthcare analytics firms and market research publications
- Review of government health statistics and publications from the GCC health ministries
- Examination of academic journals and white papers on real-world evidence methodologies
Primary Research
- Interviews with healthcare professionals and clinical researchers in the GCC region
- Surveys targeting pharmaceutical companies and biotech firms utilizing real-world evidence
- Focus groups with healthcare policymakers and regulatory bodies in the GCC
Validation & Triangulation
- Cross-validation of findings through multiple data sources including clinical trial registries
- Triangulation of insights from primary interviews with secondary data trends
- Sanity checks conducted through expert panels comprising industry veterans
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on healthcare expenditure trends in the GCC
- Segmentation by therapeutic areas and types of real-world evidence solutions
- Incorporation of government initiatives promoting the use of real-world evidence in healthcare
Bottom-up Modeling
- Data collection from leading real-world evidence solution providers on service offerings
- Estimation of market penetration rates based on current adoption levels in healthcare
- Volume and pricing analysis of real-world evidence services across different sectors
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating healthcare trends and technology adoption rates
- Scenario modeling based on potential regulatory changes and market dynamics
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Companies | 60 | Clinical Research Directors, Data Analysts |
| Healthcare Providers | 50 | Hospital Administrators, Medical Directors |
| Regulatory Bodies | 40 | Policy Makers, Regulatory Affairs Specialists |
| Biotech Firms | 40 | Product Development Managers, Market Access Specialists |
| Health Insurance Companies | 40 | Underwriters, Claims Analysts |
Frequently Asked Questions
What is the current value of the GCC Real World Evidence Solutions Market?
The GCC Real World Evidence Solutions Market is valued at approximately USD 2.5 billion, reflecting the region's rapid adoption of data-driven healthcare strategies and the increasing demand for data-backed insights in healthcare.