Region:Global
Author(s):Geetanshi
Product Code:KRAA2279
Pages:80
Published On:August 2025
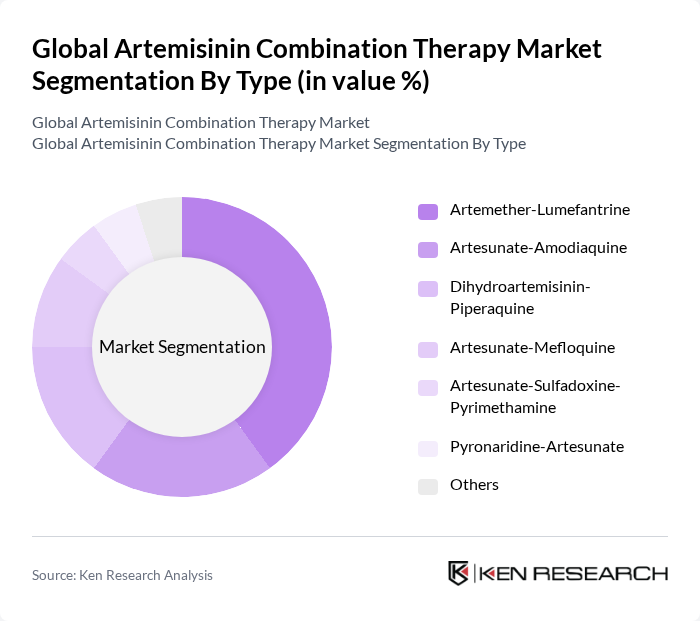
By Type:The market is segmented into various types of artemisinin combination therapies, including Artemether-Lumefantrine, Artesunate-Amodiaquine, Dihydroartemisinin-Piperaquine, Artesunate-Mefloquine, Artesunate-Sulfadoxine-Pyrimethamine, Pyronaridine-Artesunate, and Others. Among these, Artemether-Lumefantrine is the leading sub-segment due to its widespread use and proven efficacy in treating malaria. The preference for this combination is driven by its favorable safety profile and effectiveness against Plasmodium falciparum, the most lethal malaria parasite. The increasing awareness of malaria treatment options and the push for effective therapies in endemic regions further bolster the demand for this sub-segment .
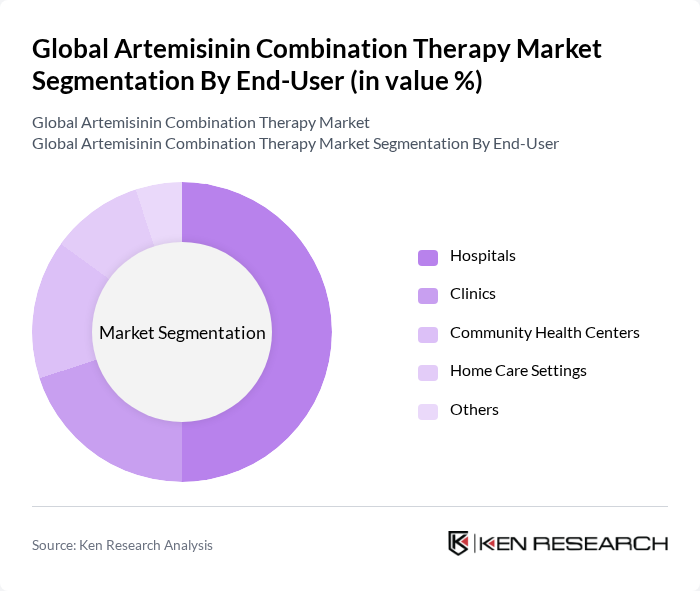
By End-User:The market is segmented by end-users, including Hospitals, Clinics, Community Health Centers, Home Care Settings, and Others. Hospitals are the dominant end-user segment, primarily due to their capacity to provide comprehensive healthcare services and access to advanced treatment options. The increasing number of malaria cases requiring hospitalization, coupled with the availability of specialized medical staff and facilities, drives the demand for artemisinin combination therapies in this segment. Additionally, hospitals often serve as the primary point of care for severe malaria cases, further solidifying their role in the market .
The Global Artemisinin Combination Therapy Market is characterized by a dynamic mix of regional and international players. Leading participants such as Novartis AG, Sanofi S.A., GlaxoSmithKline plc, Bayer AG, Medicines for Malaria Venture (MMV), Cipla Limited, Viatris Inc., Ipca Laboratories Limited, Hetero Labs Limited, Aurobindo Pharma Limited, Zydus Lifesciences Limited, Emcure Pharmaceuticals Ltd., Fosun Pharma (Shanghai Fosun Pharmaceutical Group Co., Ltd.), Guilin Pharmaceutical Co., Ltd., Ajanta Pharma Limited contribute to innovation, geographic expansion, and service delivery in this space.
The future of the artemisinin combination therapy market appears promising, driven by ongoing advancements in drug formulation and increased funding for malaria research. As governments and NGOs collaborate to enhance treatment accessibility, the market is likely to witness a surge in innovative therapies. Additionally, the integration of digital health solutions is expected to improve patient monitoring and adherence, further supporting the fight against malaria and ensuring that effective treatments reach those in need.
| Segment | Sub-Segments |
|---|---|
| By Type | Artemether-Lumefantrine Artesunate-Amodiaquine Dihydroartemisinin-Piperaquine Artesunate-Mefloquine Artesunate-Sulfadoxine-Pyrimethamine Pyronaridine-Artesunate Others |
| By End-User | Hospitals Clinics Community Health Centers Home Care Settings Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies NGO & Government Distribution Programs Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients |
| By Treatment Duration | Short-term Treatment Long-term Treatment |
| By Pricing Strategy | Premium Pricing Competitive Pricing Value-based Pricing |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers in Malaria Treatment | 100 | Doctors, Pharmacists, Healthcare Administrators |
| Pharmaceutical Distributors | 60 | Distribution Managers, Sales Representatives |
| NGO Representatives Focused on Malaria | 50 | Program Managers, Field Coordinators |
| Patients Using Artemisinin Combination Therapies | 80 | Patients, Caregivers, Advocacy Group Members |
| Regulatory Bodies and Health Authorities | 40 | Policy Makers, Health Inspectors |
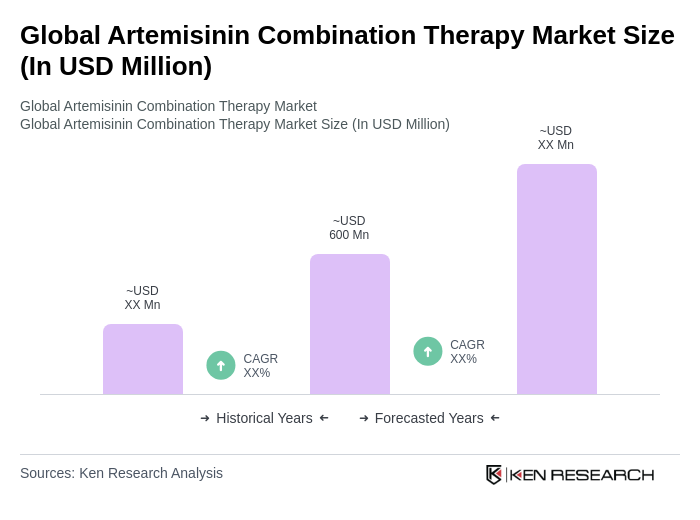
The Global Artemisinin Combination Therapy Market is valued at approximately USD 600 million, reflecting a significant growth driven by the rising incidence of malaria and the adoption of artemisinin-based therapies as the first-line treatment in endemic regions.