Region:Global
Author(s):Rebecca
Product Code:KRAC4703
Pages:93
Published On:October 2025
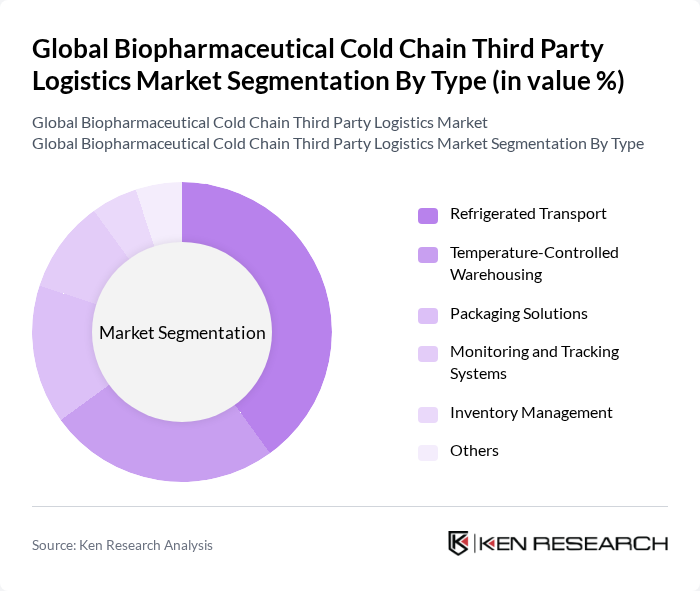
By Type:The market is segmented into various types, including Refrigerated Transport, Temperature-Controlled Warehousing, Packaging Solutions, Monitoring and Tracking Systems, Inventory Management, and Others. Among these, Refrigerated Transport is the leading sub-segment due to the increasing demand for efficient and reliable transportation of temperature-sensitive products. The rise in global trade of biopharmaceuticals necessitates advanced refrigerated transport solutions to maintain product integrity and compliance with regulatory standards. The integration of IoT devices enables real-time monitoring of environmental conditions during transit, while AI and blockchain technologies are increasingly adopted for predictive analytics and enhanced traceability across the supply chain.
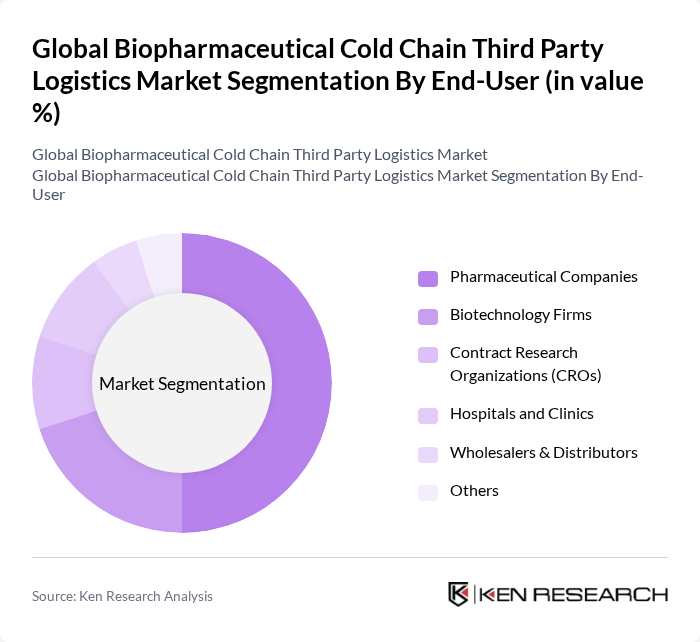
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Contract Research Organizations (CROs), Hospitals and Clinics, Wholesalers & Distributors, and Others. Pharmaceutical Companies dominate this segment, driven by the increasing production of biologics and the need for stringent temperature control during distribution. The growing focus on research and development in the pharmaceutical sector, globalization of clinical trials, and the rise of personalized medicines further propel the demand for specialized cold chain logistics services.
The Global Biopharmaceutical Cold Chain Third Party Logistics Market is characterized by a dynamic mix of regional and international players. Leading participants such as DHL Supply Chain, FedEx Corporation, UPS Healthcare, Kuehne + Nagel, DB Schenker, SF Express, Cardinal Health, Thermo Fisher Scientific, GEODIS, Cencora (formerly AmerisourceBergen), Maersk, Kerry Logistics Network Limited, World Courier, BioCare Copenhagen, Agility Logistics contribute to innovation, geographic expansion, and service delivery in this space.
The future of the biopharmaceutical cold chain logistics market appears promising, driven by technological advancements and increasing global demand for biopharmaceuticals. As the industry embraces IoT and smart technologies, operational efficiencies are expected to improve significantly. Additionally, the rise of personalized medicine will further necessitate sophisticated cold chain solutions, ensuring that temperature-sensitive products are delivered safely and efficiently. The market is poised for transformative growth as stakeholders adapt to evolving regulatory landscapes and consumer needs.
| Segment | Sub-Segments |
|---|---|
| By Type | Refrigerated Transport Temperature-Controlled Warehousing Packaging Solutions Monitoring and Tracking Systems Inventory Management Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Contract Research Organizations (CROs) Hospitals and Clinics Wholesalers & Distributors Others |
| By Distribution Mode | Direct Distribution Third-Party Distribution E-commerce Distribution Others |
| By Packaging Type | Insulated Containers Refrigerated Pallets Temperature-Controlled Boxes Phase Change Materials Packaging Others |
| By Service Type | Transportation Services Warehousing Services Packaging Services Monitoring Services Inventory Management Services Others |
| By Temperature Range | Refrigerated (2°C to 8°C) Frozen (-20°C to -80°C) Ultra-Frozen/Deep-Frozen (below -80°C) Controlled Room Temperature (15°C to 25°C) Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Price Range | Budget Mid-Range Premium Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Cold Chain Logistics | 60 | Logistics Managers, Supply Chain Executives |
| Biotech Product Distribution | 50 | Operations Directors, Quality Control Managers |
| Clinical Trial Supply Chain | 40 | Clinical Operations Managers, Regulatory Affairs Specialists |
| Vaccine Storage and Transport | 45 | Cold Chain Specialists, Warehouse Managers |
| Temperature-Sensitive Drug Delivery | 40 | Pharmaceutical Logistics Coordinators, Compliance Officers |
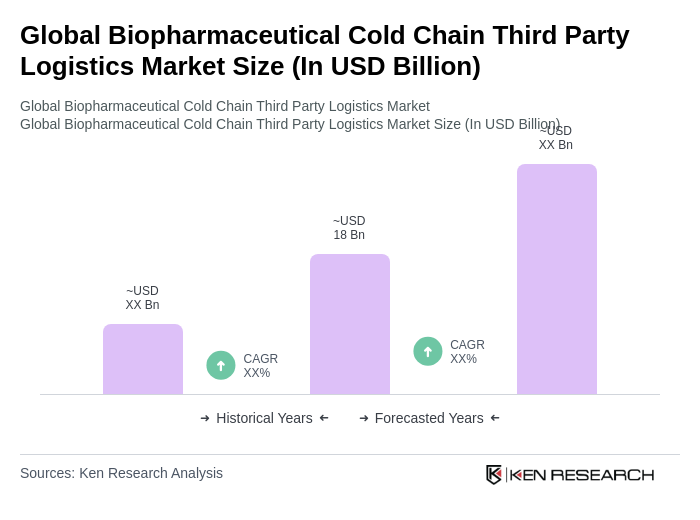
The Global Biopharmaceutical Cold Chain Third Party Logistics Market is valued at approximately USD 18 billion, driven by the increasing demand for temperature-sensitive pharmaceuticals and the rise in biopharmaceutical production.