Global Breast Cancer Liquid Biopsy Market Overview
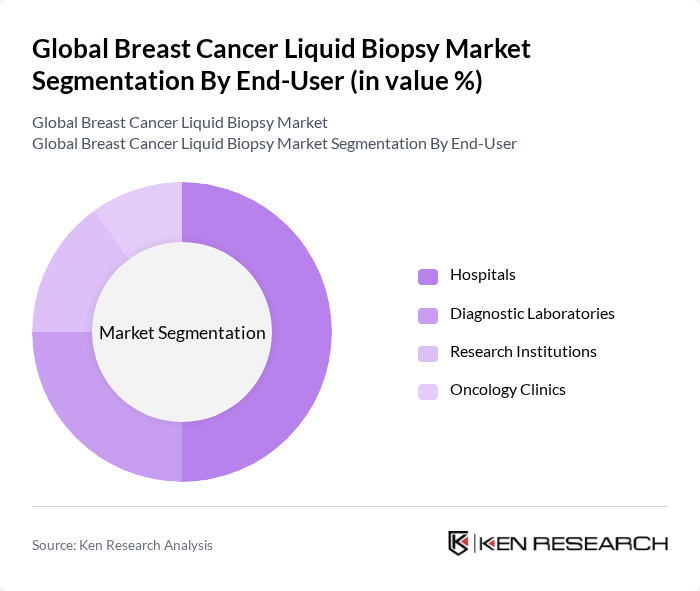
- The Global Breast Cancer Liquid Biopsy Market is valued at USD 1.3 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of breast cancer, rapid advancements in liquid biopsy technologies, and the rising demand for non-invasive diagnostic methods. The market is further supported by the growing adoption of early detection strategies, the expansion of personalized medicine, and the integration of real-time cancer monitoring into clinical workflows, all of which enhance patient outcomes .
- Key players in this market are predominantly located in North America and Europe, with the United States being a major contributor due to its advanced healthcare infrastructure, high R&D investments, and a strong focus on cancer research. Additionally, countries such as Germany and the United Kingdom are significant contributors, driven by robust healthcare systems and the increasing adoption of innovative diagnostic solutions .
- In 2023, the U.S. Food and Drug Administration (FDA) issued updated guidance under the Breakthrough Devices Program, streamlining the approval process for liquid biopsy tests. This regulatory framework, formalized in the “Breakthrough Devices Program Guidance” (U.S. FDA, 2023), provides an expedited pathway for clinical validation and market entry of innovative diagnostic tools, thereby enhancing the availability of liquid biopsy solutions for breast cancer detection and monitoring. The program emphasizes early and frequent communication with manufacturers, prioritized review, and flexible clinical evidence requirements for devices addressing unmet medical needs .
Global Breast Cancer Liquid Biopsy Market Segmentation
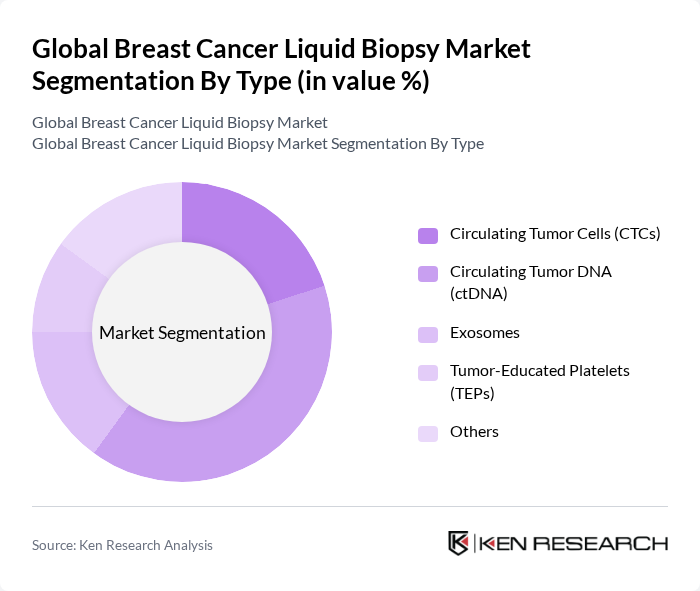
By Type:The market is segmented into various types of liquid biopsy technologies, including Circulating Tumor Cells (CTCs), Circulating Tumor DNA (ctDNA), Exosomes, Tumor-Educated Platelets (TEPs), and Others. Among these, Circulating Tumor DNA (ctDNA) is currently dominating the market due to its high sensitivity and specificity in detecting genetic mutations associated with breast cancer. The increasing adoption of ctDNA for early detection and treatment monitoring is driven by its non-invasive nature, the ability to track tumor evolution in real time, and the growing trend towards personalized medicine .
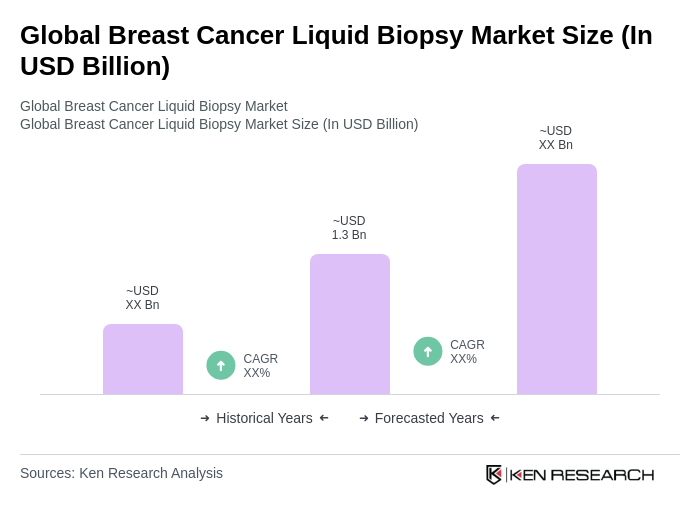
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Research Institutions, and Oncology Clinics. Hospitals are the leading end-users in the liquid biopsy market, primarily due to their comprehensive cancer care services and the increasing integration of advanced diagnostic technologies in clinical settings. The demand for liquid biopsy tests in hospitals is further fueled by the need for timely and accurate cancer diagnosis and monitoring, as well as the expansion of multidisciplinary oncology programs .
Global Breast Cancer Liquid Biopsy Market Competitive Landscape
The Global Breast Cancer Liquid Biopsy Market is characterized by a dynamic mix of regional and international players. Leading participants such as Guardant Health, Inc., Exact Sciences Corporation, Biocept, Inc., Foundation Medicine, Inc., Roche Diagnostics, Thermo Fisher Scientific Inc., Illumina, Inc., QIAGEN N.V., Sysmex Corporation, Siemens Healthineers, Myriad Genetics, Inc., Freenome Holdings, Inc., GRAIL, Inc., C2i Genomics, Inc., OncoCyte Corporation contribute to innovation, geographic expansion, and service delivery in this space.
Global Breast Cancer Liquid Biopsy Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Breast Cancer:The incidence of breast cancer is projected to reach approximately 2.3 million new cases globally in future, according to the World Health Organization. This rising prevalence is a significant driver for the liquid biopsy market, as healthcare systems seek innovative diagnostic solutions. The increasing number of diagnosed cases necessitates advanced testing methods, leading to a higher demand for liquid biopsies, which offer a less invasive alternative to traditional methods.
- Advancements in Liquid Biopsy Technologies:The liquid biopsy market is experiencing rapid technological advancements, with over 50 new products expected to enter the market in future. Innovations such as next-generation sequencing and digital PCR are enhancing the sensitivity and specificity of these tests. These advancements are crucial in improving early detection rates and monitoring treatment responses, thereby driving market growth as healthcare providers adopt these cutting-edge technologies to improve patient outcomes.
- Rising Demand for Non-Invasive Diagnostic Methods:The global shift towards non-invasive diagnostic methods is evident, with a projected increase in demand for liquid biopsies expected to reach 1.5 million tests annually in future. Patients and healthcare providers favor these methods due to their reduced risk and discomfort compared to traditional biopsies. This trend is further supported by a growing body of evidence demonstrating the effectiveness of liquid biopsies in detecting cancer biomarkers, thus propelling market growth.
Market Challenges
- High Costs Associated with Liquid Biopsy Tests:The average cost of liquid biopsy tests can range from $1,000 to $3,000, which poses a significant barrier to widespread adoption. Many healthcare systems face budget constraints, limiting their ability to implement these advanced diagnostic tools. As a result, the high costs can deter patients from opting for liquid biopsies, thereby hindering market growth despite the technology's potential benefits.
- Regulatory Hurdles and Approval Processes:The regulatory landscape for liquid biopsies is complex, with the FDA requiring extensive clinical validation before approval. As of future, only 10 liquid biopsy tests have received FDA approval, creating a bottleneck in market entry for new products. This lengthy approval process can delay the introduction of innovative solutions, limiting the market's growth potential and discouraging investment in research and development.
Global Breast Cancer Liquid Biopsy Market Future Outlook
The future of the breast cancer liquid biopsy market appears promising, driven by technological advancements and increasing awareness of non-invasive testing methods. As healthcare providers continue to prioritize early detection and personalized treatment plans, the integration of artificial intelligence and machine learning into diagnostic processes is expected to enhance accuracy and efficiency. Furthermore, the expansion into emerging markets will provide new avenues for growth, as these regions increasingly adopt advanced healthcare technologies to combat rising cancer rates.
Market Opportunities
- Expansion into Emerging Markets:Emerging markets, particularly in Asia and Africa, are witnessing a surge in breast cancer cases, with an estimated 1 million new cases expected in future. This presents a significant opportunity for liquid biopsy companies to establish a foothold in these regions, where demand for innovative diagnostic solutions is rapidly increasing, driven by improving healthcare infrastructure and rising awareness.
- Development of Personalized Medicine Approaches:The shift towards personalized medicine is creating opportunities for liquid biopsy technologies to play a pivotal role in tailoring treatment plans. With over 60% of oncologists expressing interest in using liquid biopsies for personalized treatment strategies in future, companies that focus on developing targeted therapies based on liquid biopsy results are likely to gain a competitive edge in the market.