Region:Global
Author(s):Shubham
Product Code:KRAC2830
Pages:92
Published On:October 2025
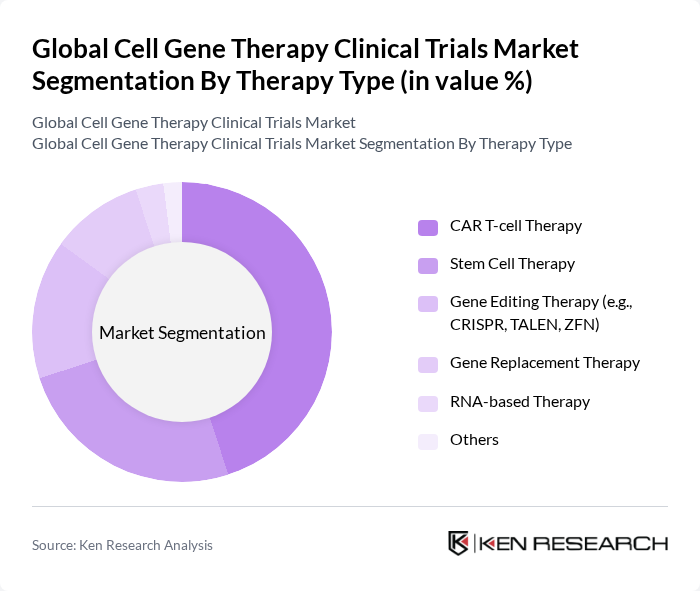
By Therapy Type:The therapy type segmentation includes a diverse range of innovative approaches to gene therapy, each with distinct mechanisms and clinical applications. The dominant sub-segment is CAR T-cell Therapy, which has gained significant traction due to its effectiveness in treating hematologic cancers and select solid tumors. Stem Cell Therapy is also prominent, particularly for regenerative medicine and rare disease applications. Gene Editing Therapy, including CRISPR, TALEN, and ZFN, is rapidly evolving, enabling precise genomic modifications for inherited disorders. Gene Replacement and RNA-based Therapies are gaining attention for their potential in treating monogenic diseases and rare conditions. The "Others" category encompasses emerging modalities such as in vivo gene transfer and next-generation cell therapies that remain in early-stage development .
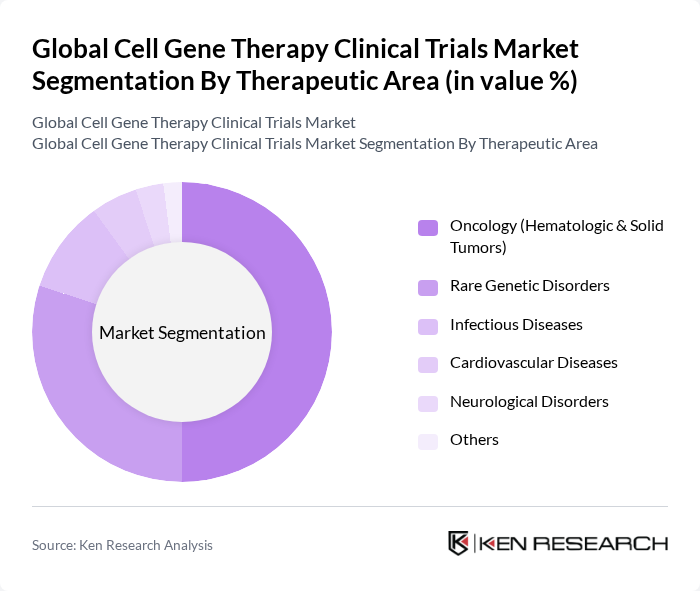
By Therapeutic Area:The therapeutic area segmentation highlights the broad applications of cell and gene therapies across multiple medical conditions. Oncology, especially hematologic and solid tumors, remains the leading area due to the high demand for advanced cancer treatments. Rare Genetic Disorders are a significant market segment, driven by the need for targeted therapies for conditions such as sickle cell anemia and inherited retinal diseases. Infectious Diseases and Cardiovascular Diseases are emerging areas of focus, supported by ongoing clinical trials for viral infections and heart failure. Neurological Disorders are gaining attention due to the increasing prevalence of conditions like Alzheimer's and Parkinson's disease. The "Others" category includes less common indications such as ophthalmology and autoimmune disorders .
The Global Cell Gene Therapy Clinical Trials Market is characterized by a dynamic mix of regional and international players. Leading participants such as Novartis AG, Gilead Sciences, Inc., Bristol-Myers Squibb Company, Spark Therapeutics, Inc., Bluebird Bio, Inc., Regeneron Pharmaceuticals, Inc., Amgen Inc., Roche Holding AG, Pfizer Inc., CRISPR Therapeutics AG, Editas Medicine, Inc., Intellia Therapeutics, Inc., Sangamo Therapeutics, Inc., Cellectis S.A., Orchard Therapeutics plc, Vertex Pharmaceuticals Incorporated, Beam Therapeutics Inc., Fate Therapeutics, Inc., Legend Biotech Corporation, Poseida Therapeutics, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the cell gene therapy clinical trials market appears promising, driven by technological advancements and increasing collaboration among stakeholders. As regulatory frameworks evolve to accommodate innovative therapies, the approval processes are expected to become more efficient. Additionally, the integration of artificial intelligence in trial design will enhance patient recruitment and data analysis, leading to faster and more effective clinical outcomes. This dynamic environment will likely foster a surge in novel therapies entering the market.
| Segment | Sub-Segments |
|---|---|
| By Therapy Type | CAR T-cell Therapy Stem Cell Therapy Gene Editing Therapy (e.g., CRISPR, TALEN, ZFN) Gene Replacement Therapy RNA-based Therapy Others |
| By Therapeutic Area | Oncology (Hematologic & Solid Tumors) Rare Genetic Disorders Infectious Diseases Cardiovascular Diseases Neurological Disorders Others |
| By Sponsor Type | Industry-Sponsored Trials (Biotech/Pharma) Academic/Research Institution Sponsored Trials Collaborative Trials (Industry-Academic Partnerships) Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Funding Source | Government Grants Private Investments Venture Capital Public Offerings Philanthropic Foundations Others |
| By Clinical Phase | Phase I Phase II Phase III Phase IV Preclinical Others |
| By Delivery Method | Intravenous Intramuscular Subcutaneous Local Administration In Vivo vs Ex Vivo Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Gene Therapy Trials | 120 | Clinical Researchers, Oncologists |
| Rare Disease Gene Therapy Studies | 100 | Geneticists, Rare Disease Specialists |
| Regulatory Affairs in Gene Therapy | 80 | Regulatory Managers, Compliance Officers |
| Patient Recruitment Strategies | 70 | Clinical Trial Managers, Patient Advocacy Leaders |
| Market Access and Reimbursement | 90 | Health Economists, Payer Relations Managers |
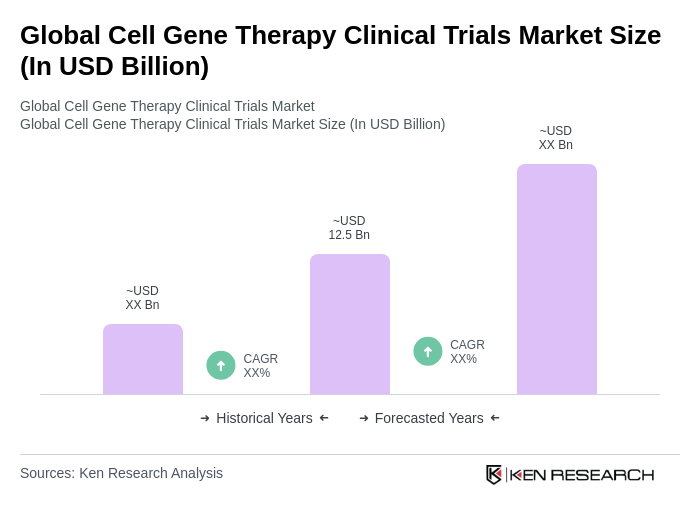
The Global Cell Gene Therapy Clinical Trials Market is valued at approximately USD 12.5 billion, reflecting significant growth driven by advancements in biotechnology and increasing prevalence of genetic disorders, alongside rising investments in research and development.