Region:Middle East
Author(s):Rebecca
Product Code:KRAC2538
Pages:82
Published On:October 2025
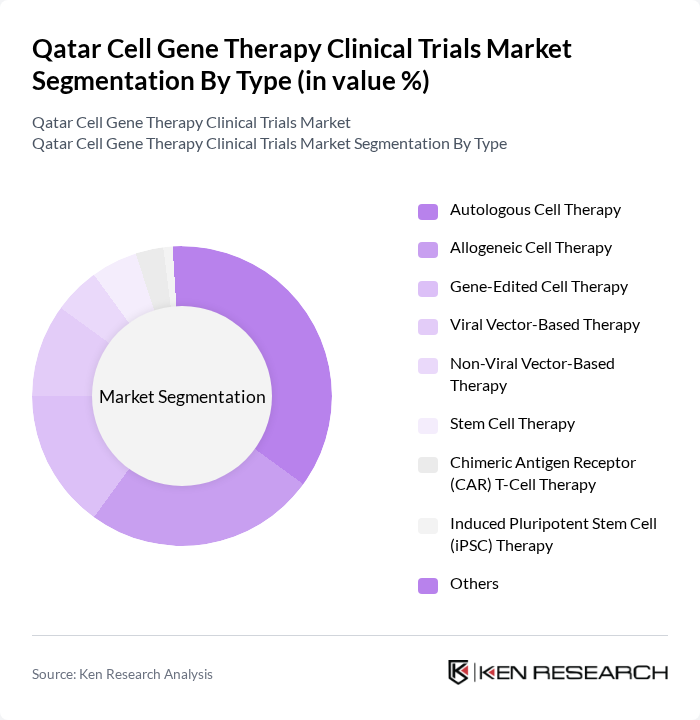
By Type:The market is segmented into various types of therapies, including Autologous Cell Therapy, Allogeneic Cell Therapy, Gene-Edited Cell Therapy, Viral Vector-Based Therapy, Non-Viral Vector-Based Therapy, Stem Cell Therapy, Chimeric Antigen Receptor (CAR) T-Cell Therapy, Induced Pluripotent Stem Cell (iPSC) Therapy, and Others. Among these, Autologous Cell Therapy is currently leading the market due to its personalized approach, which has shown promising results in treating various cancers and genetic disorders. The increasing acceptance of personalized medicine, growing demand for regenerative therapies, and advancements in cell processing technologies are driving the growth of this segment .
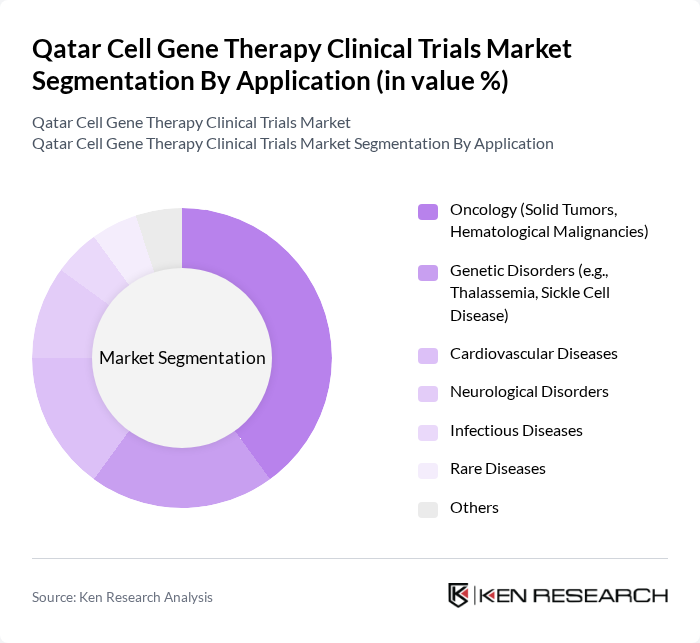
By Application:The applications of cell gene therapy include Oncology (Solid Tumors, Hematological Malignancies), Genetic Disorders (e.g., Thalassemia, Sickle Cell Disease), Cardiovascular Diseases, Neurological Disorders, Infectious Diseases, Rare Diseases, and Others. The Oncology segment is the most significant contributor to the market, driven by the increasing incidence of cancer, high demand for innovative treatment options, and the success of CAR T-cell therapies in hematological malignancies. The segment’s growth is further propelled by ongoing clinical trials targeting solid tumors and blood cancers, as healthcare providers seek effective solutions for challenging cancer cases .
The Qatar Cell Gene Therapy Clinical Trials Market is characterized by a dynamic mix of regional and international players. Leading participants such as Novartis AG, Gilead Sciences, Inc. (Kite Pharma), Spark Therapeutics, Inc. (Roche Group), bluebird bio, Inc., CRISPR Therapeutics AG, Editas Medicine, Inc., Amgen Inc., Regeneron Pharmaceuticals, Inc., Celgene Corporation (Bristol Myers Squibb), Roche Holding AG, Bayer AG, AstraZeneca PLC, Merck & Co., Inc., Pfizer Inc., Sanofi S.A., Lonza Group AG, Catalent, Inc., Thermo Fisher Scientific Inc., Charles River Laboratories International, Inc., Avernus Pharma, Opal Biopharma contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Qatar cell gene therapy clinical trials market appears promising, driven by increasing collaboration between local and international research institutions. As the government continues to support biotechnology initiatives, the establishment of more clinical trial networks is anticipated. Additionally, the integration of artificial intelligence in trial designs is expected to enhance efficiency and patient recruitment, paving the way for innovative therapies that address unmet medical needs in the region.
| Segment | Sub-Segments |
|---|---|
| By Type | Autologous Cell Therapy Allogeneic Cell Therapy Gene-Edited Cell Therapy Viral Vector-Based Therapy Non-Viral Vector-Based Therapy Stem Cell Therapy Chimeric Antigen Receptor (CAR) T-Cell Therapy Induced Pluripotent Stem Cell (iPSC) Therapy Others |
| By Application | Oncology (Solid Tumors, Hematological Malignancies) Genetic Disorders (e.g., Thalassemia, Sickle Cell Disease) Cardiovascular Diseases Neurological Disorders Infectious Diseases Rare Diseases Others |
| By End-User | Hospitals & Specialty Clinics Academic & Research Institutions Biotechnology & Pharmaceutical Companies Contract Research Organizations (CROs) Others |
| By Funding Source | Government Grants Private Investments Public-Private Partnerships Venture Capital International Funding Agencies Others |
| By Clinical Phase | Pre-Clinical Trials Phase I Trials Phase II Trials Phase III Trials Phase IV Trials Others |
| By Region | Doha Al Rayyan Al Wakrah Al Khor Others |
| By Regulatory Status | Approved Therapies Therapies Under Review Investigational Therapies Compassionate Use Programs Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Trial Coordinators | 60 | Clinical Research Associates, Trial Managers |
| Gene Therapy Researchers | 50 | Academic Researchers, Laboratory Directors |
| Healthcare Providers | 40 | Oncologists, Genetic Counselors |
| Patient Advocacy Groups | 40 | Patient Representatives, Advocacy Leaders |
| Regulatory Experts | 40 | Regulatory Affairs Specialists, Compliance Officers |
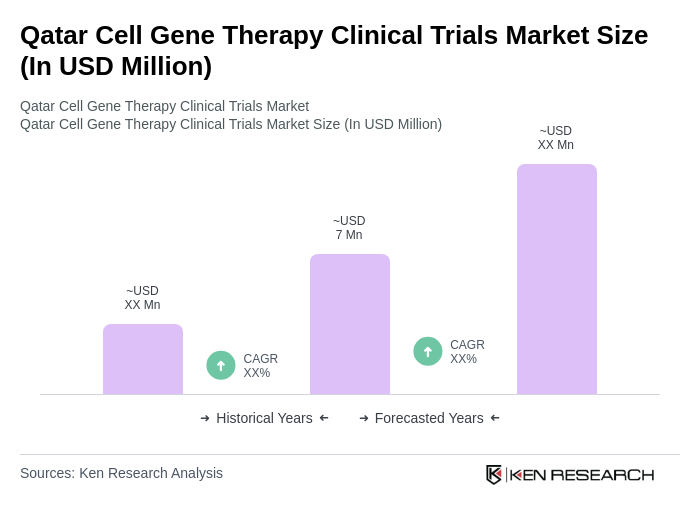
The Qatar Cell Gene Therapy Clinical Trials Market is valued at approximately USD 7 million, driven by increasing investments in biotechnology, advancements in gene editing technologies, and a rising prevalence of genetic disorders and cancers.