Region:Global
Author(s):Dev
Product Code:KRAC3604
Pages:82
Published On:January 2026
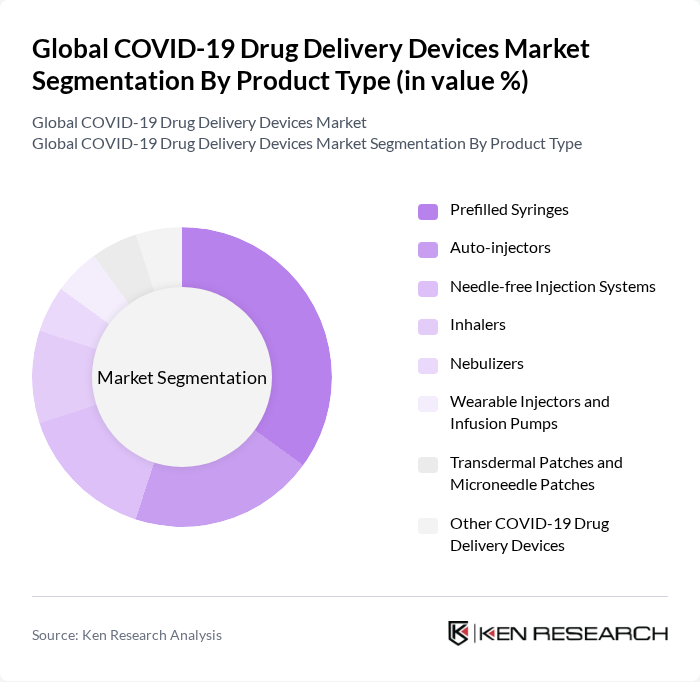
By Product Type:The product type segmentation includes various devices designed for the effective delivery of COVID-19 vaccines and therapeutics. The subsegments are Prefilled Syringes, Auto-injectors, Needle-free Injection Systems, Inhalers, Nebulizers, Wearable Injectors and Infusion Pumps, Transdermal Patches and Microneedle Patches, and Other COVID-19 Drug Delivery Devices. Among these, Prefilled Syringes have emerged as one of the leading subsegments for mass vaccination due to their ease of use, reduced risk of dosing errors and contamination, and ability to streamline the vaccination process in large-scale campaigns. The demand for prefilled syringes and related injection systems has increased as they facilitate rapid vaccination efforts and support high-throughput immunization in hospital, clinic, and community settings.
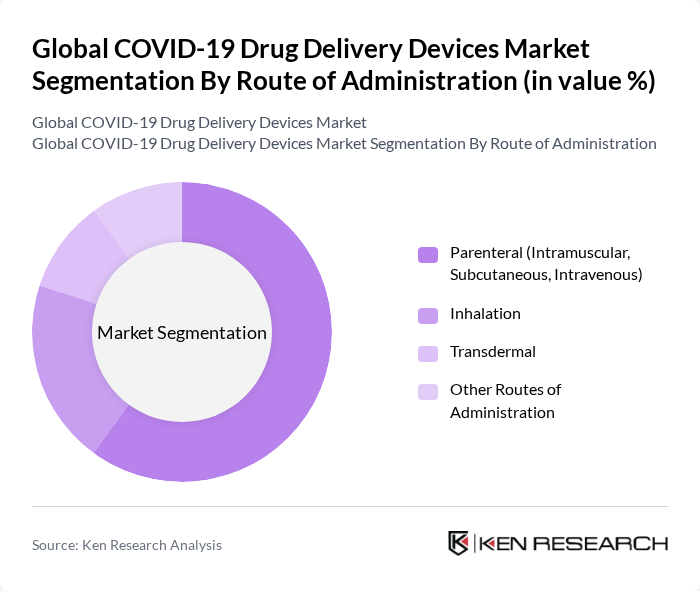
By Route of Administration:The route of administration segmentation encompasses various methods through which COVID-19 vaccines and therapeutics are delivered. The subsegments include Parenteral (Intramuscular, Subcutaneous, Intravenous), Inhalation, Transdermal, and Other Routes of Administration. Parenteral administration is the dominant route for COVID?19 vaccines, which are primarily delivered via intramuscular injection, reflecting widespread acceptance of injectable vaccines and established clinical protocols. This route ensures efficient induction of immune responses and is supported by well-developed infrastructure in hospitals, clinics, and mass vaccination sites for administering injections at scale.
The Global COVID-19 Drug Delivery Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pfizer Inc., Moderna, Inc., Johnson & Johnson Services, Inc., AstraZeneca plc, Novavax, Inc., GlaxoSmithKline plc (GSK), Sanofi S.A., Merck & Co., Inc., F. Hoffmann-La Roche Ltd, Regeneron Pharmaceuticals, Inc., Eli Lilly and Company, Serum Institute of India Pvt. Ltd., Becton, Dickinson and Company (BD), Terumo Corporation, West Pharmaceutical Services, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the COVID-19 drug delivery devices market is poised for significant transformation, driven by ongoing technological advancements and an increasing focus on patient-centric solutions. As healthcare systems adapt to the evolving landscape of infectious diseases, the integration of artificial intelligence and digital health solutions will enhance the efficiency of drug delivery. Furthermore, the emphasis on personalized medicine will lead to the development of tailored delivery systems, ensuring that treatments are more effective and accessible to diverse patient populations.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Prefilled Syringes Auto-injectors Needle-free Injection Systems Inhalers Nebulizers Wearable Injectors and Infusion Pumps Transdermal Patches and Microneedle Patches Other COVID-19 Drug Delivery Devices |
| By Route of Administration | Parenteral (Intramuscular, Subcutaneous, Intravenous) Inhalation Transdermal Other Routes of Administration |
| By Application | Vaccine Delivery Antiviral and Therapeutic Delivery Supportive and Adjunctive Therapy Delivery Other COVID-19-related Applications |
| By End-User | Hospitals Hospital Pharmacies Retail Pharmacies Ambulatory Care and Clinics Home Care Settings Other End-Users |
| By Distribution Channel | Direct Tenders and Institutional Sales Retail Pharmacies Online Pharmacies and E-commerce Platforms Distributor and Wholesale Channels Other Distribution Channels |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Technology | Conventional Mechanical Delivery Devices Connected and Smart Drug Delivery Devices Needle-free and Microneedle Technologies Other Enabling Technologies |
| By Regulatory Approval Status | Commercially Approved Devices Emergency Use Authorization (EUA) Devices Devices in Clinical Trials Devices in Development and Preclinical Stage |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 120 | Product Managers, R&D Directors |
| Healthcare Providers | 110 | Doctors, Nurses, Pharmacists |
| Patients Using COVID-19 Treatments | 90 | COVID-19 Patients, Caregivers |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Market Analysts | 60 | Healthcare Analysts, Market Researchers |
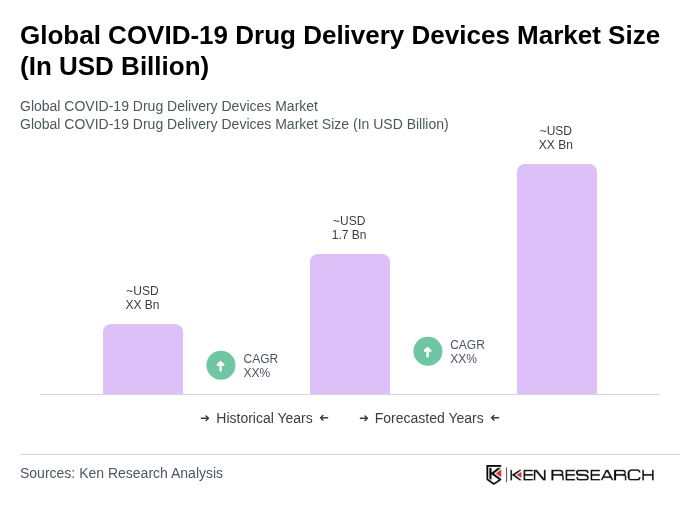
The Global COVID-19 Drug Delivery Devices Market is valued at approximately USD 1.7 billion, reflecting significant growth driven by the urgent need for effective vaccine distribution and advancements in drug delivery technologies during the pandemic.