About the Report
Base Year 2024Global Craniomaxillofacial CMF Devices System Market Overview
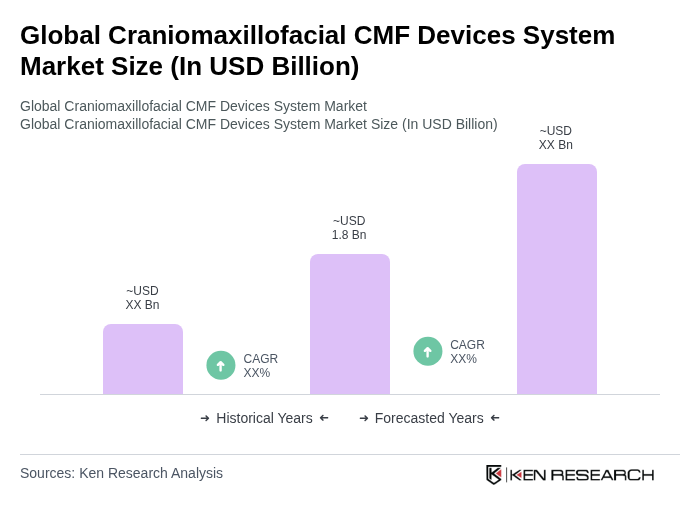
- The Global Craniomaxillofacial CMF Devices System Market is valued at USD 1.8 billion, based on a five-year historical analysis, aligning with recent global craniomaxillofacial devices revenue estimates. This growth is primarily driven by the increasing prevalence of facial trauma from road traffic accidents and sports injuries, a higher incidence of congenital craniofacial deformities, and the rising demand for elective and reconstructive cosmetic surgeries. Additionally, advancements in surgical techniques such as computer-assisted and image-guided surgery, 3D planning, and patient-specific/3D-printed implants, along with the use of next?generation materials including titanium and bioresorbable polymers, have further propelled market expansion as healthcare providers seek innovative solutions to enhance functional and aesthetic outcomes.
- Key players in this market are predominantly located in North America and Europe, with the United States and Germany leading due to their advanced healthcare infrastructure, high investment in medical technology, and a strong focus on research and development. These regions benefit from a well-established network of hospitals, trauma centers, and specialty clinics, widespread reimbursement coverage, and strong presence of global CMF device manufacturers, which together facilitate rapid adoption of technologically advanced fixation systems, distraction devices, and joint replacement solutions.
- Craniomaxillofacial devices marketed in the United States are regulated as medical devices by the U.S. Food and Drug Administration (FDA) under the Federal Food, Drug, and Cosmetic Act and its implementing regulations in Title 21 of the Code of Federal Regulations, including device classification, premarket notification or approval, and post?market requirements. For example, craniofacial bone plates and screws are classified under 21 CFR 872.4760, and manufacturers must comply with the Quality System Regulation in 21 CFR Part 820, Medical Device Reporting requirements in 21 CFR Part 803, and, where applicable, Premarket Approval provisions in 21 CFR Part 814, which together mandate robust clinical evidence, risk management, labeling, and post?market surveillance to ensure safety and effectiveness.
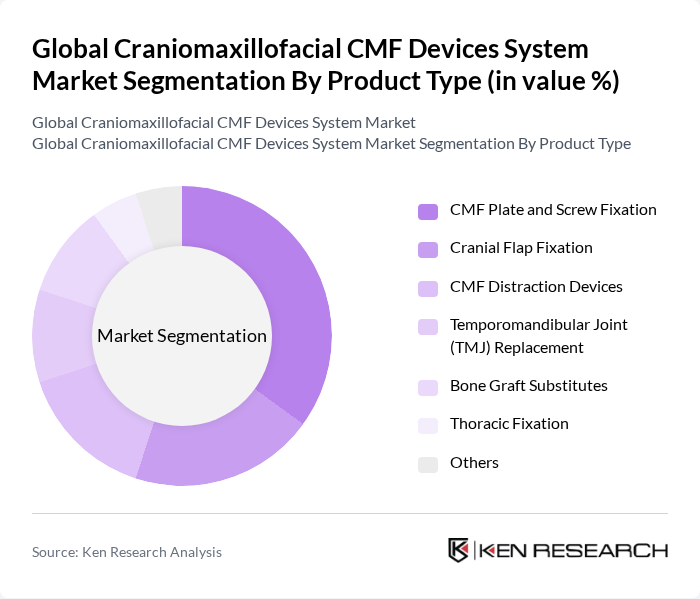
Global Craniomaxillofacial CMF Devices System Market Segmentation
By Product Type:The product type segmentation includes various categories such as CMF Plate and Screw Fixation, Cranial Flap Fixation, CMF Distraction Devices, Temporomandibular Joint (TMJ) Replacement, Bone Graft Substitutes, Thoracic Fixation, and Others. This structure is consistent with standard segmentation used in recent CMF market analyses. Among these, CMF Plate and Screw Fixation is the leading sub-segment due to its widespread application in fracture stabilization, reconstructive surgeries, and trauma cases, accounting for the largest revenue contribution within the global CMF devices portfolio. The increasing number of craniofacial trauma procedures, neurosurgical interventions, and orthognathic surgeries, coupled with the demand for rigid and reliable internal fixation solutions, advanced low?profile plating systems, and patient?specific plates, contributes to its dominance in the market.
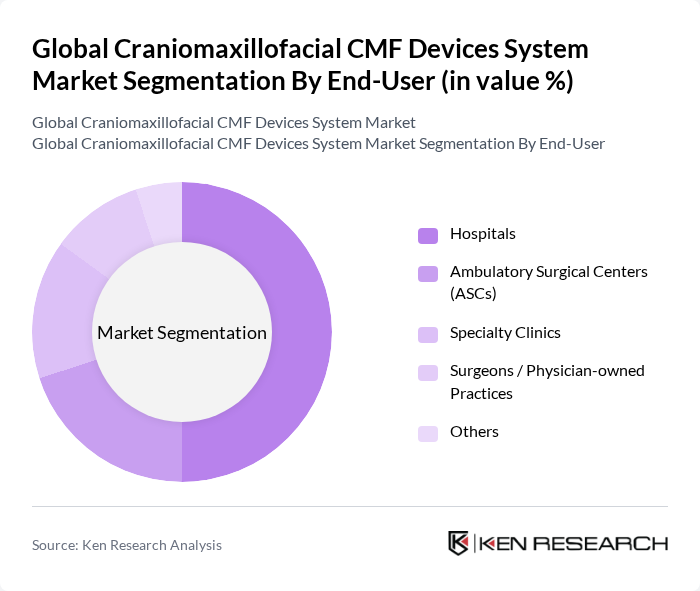
By End-User:The end-user segmentation encompasses Hospitals, Ambulatory Surgical Centers (ASCs), Specialty Clinics, Surgeons/Physician-owned Practices, and Others, reflecting the main care settings where CMF procedures are performed. Hospitals are the dominant end-user segment, primarily due to their capacity to handle complex trauma, neurosurgical, oncologic, and reconstructive surgeries, and the availability of advanced operating room infrastructure, imaging, and intensive care facilities. The increasing number of road?traffic and sports?related injuries treated in hospital trauma centers, alongside a growing patient population undergoing elective orthognathic and craniofacial corrective procedures in tertiary hospitals, drives the demand for CMF devices in this segment, while ASCs and specialty clinics are gaining importance in select reconstructive and cosmetic indications.
Global Craniomaxillofacial CMF Devices System Market Competitive Landscape
The Global Craniomaxillofacial CMF Devices System Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Stryker Corporation, DePuy Synthes (Johnson & Johnson), Zimmer Biomet Holdings, Inc., KLS Martin Group, Integra LifeSciences Holdings Corporation, Aesculap Implant Systems (B. Braun), OsteoMed L.P., Biomet Microfixation (Zimmer Biomet), CONMED Corporation, B. Braun Melsungen AG, Medartis AG, KLS Martin LP / subsidiaries (regional profiles as relevant), Other Emerging CMF-focused Players, Regional / Local Manufacturers (Representative Sample) contribute to innovation, geographic expansion, and service delivery in this space.
Global Craniomaxillofacial CMF Devices System Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Facial Trauma:The World Health Organization reported that approximately 1.19 million people die each year from road traffic accidents, with many survivors suffering facial injuries. In the U.S. alone, over 3 million cases of facial trauma are estimated to be treated annually in hospital emergency departments, driving demand for CMF devices. This rising incidence of facial trauma necessitates advanced surgical interventions, thereby propelling the growth of the CMF devices market significantly.
- Advancements in Surgical Techniques:The integration of innovative surgical techniques, such as minimally invasive procedures, has transformed CMF surgeries. For instance, the use of endoscopic techniques has increased in recent years, enhancing recovery times and patient outcomes. These advancements not only improve surgical precision but also expand the range of treatable conditions, thus stimulating market growth for CMF devices.
- Rising Demand for Cosmetic Surgeries:The American Society of Plastic Surgeons reported that over 26 million cosmetic procedures (surgical and minimally invasive) were performed in the U.S. in future, reflecting an increase from previous periods. This growing interest in aesthetic enhancements is driving the demand for CMF devices, as patients seek advanced solutions for facial reconstruction and enhancement, further boosting market dynamics.
Market Challenges
- High Cost of CMF Devices:The average cost of CMF devices can range from USD 1,000 to USD 10,000, depending on the complexity and technology involved. This high price point can limit accessibility for many patients, particularly in developing regions. Consequently, the financial burden associated with these devices poses a significant challenge to market penetration and growth.
- Stringent Regulatory Requirements:Regulatory bodies, such as the FDA and EMA, impose rigorous standards for the approval of medical devices, including CMF products. The approval process can take several years and requires extensive clinical data, which can delay product launches. This regulatory landscape creates barriers for new entrants and can hinder innovation within the CMF devices market.
Global Craniomaxillofacial CMF Devices System Market Future Outlook
The future of the CMF devices market appears promising, driven by technological advancements and increasing healthcare investments. The integration of 3D printing technology is expected to revolutionize device customization, enhancing patient outcomes. Additionally, the growing focus on personalized medicine will likely lead to tailored surgical solutions, improving recovery rates. As healthcare infrastructure expands, particularly in emerging markets, the demand for CMF devices is anticipated to rise, fostering innovation and collaboration among industry players.
Market Opportunities
- Expansion in Emerging Markets:Emerging economies, particularly in Asia-Pacific and Latin America, are witnessing rapid urbanization and increased healthcare spending. For instance, healthcare expenditure in India is projected to reach around USD 372 billion in future. This growth presents significant opportunities for CMF device manufacturers to penetrate these markets and cater to the rising demand for advanced surgical solutions.
- Development of Innovative Materials:The ongoing research into biocompatible materials and smart technologies is creating new avenues for CMF device development. Innovations such as bioactive glass and titanium alloys are gaining traction, enhancing the performance and longevity of devices. This trend not only improves patient outcomes but also positions companies to capitalize on the growing demand for advanced CMF solutions.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | CMF Plate and Screw Fixation Cranial Flap Fixation CMF Distraction Devices Temporomandibular Joint (TMJ) Replacement Bone Graft Substitutes Thoracic Fixation Others |
| By End-User | Hospitals Ambulatory Surgical Centers (ASCs) Specialty Clinics Surgeons / Physician-owned Practices Others |
| By Material | Metal-based Implants (e.g., Titanium, Stainless Steel) Bioabsorbable Materials Ceramic-based Materials Composite / Hybrid Materials Others |
| By Application | Trauma Reconstruction Orthognathic and Dental Surgery Neurosurgery & ENT Cosmetic / Plastic Surgery Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Distribution Channel | Direct Tenders / Institutional Sales Distributor / Dealer Network Group Purchasing Organizations (GPOs) Online / E-Procurement Platforms Others |
| By Technology / Surgical Approach | Conventional / Open Surgical Techniques Image-Guided and Navigation-assisted Surgery D Printing and Patient-specific Implants Robotic-Assisted Surgery Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., FDA, EMA)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Regulatory Consultants
Industry Associations (e.g., American Association of Oral and Maxillofacial Surgeons)
Health Insurance Companies
Players Mentioned in the Report:
Medtronic plc
Stryker Corporation
DePuy Synthes (Johnson & Johnson)
Zimmer Biomet Holdings, Inc.
KLS Martin Group
Integra LifeSciences Holdings Corporation
Aesculap Implant Systems (B. Braun)
OsteoMed L.P.
Biomet Microfixation (Zimmer Biomet)
CONMED Corporation
B. Braun Melsungen AG
Medartis AG
KLS Martin LP / subsidiaries (regional profiles as relevant)
Other Emerging CMF-focused Players
Regional / Local Manufacturers (Representative Sample)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Craniomaxillofacial CMF Devices System Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Craniomaxillofacial CMF Devices System Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Craniomaxillofacial CMF Devices System Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of facial trauma and congenital deformities
3.1.2 Advancements in surgical techniques and technologies
3.1.3 Rising demand for cosmetic surgeries
3.1.4 Growing geriatric population requiring reconstructive surgeries
3.2 Market Challenges
3.2.1 High cost of CMF devices
3.2.2 Stringent regulatory requirements
3.2.3 Limited reimbursement policies
3.2.4 Competition from alternative treatment options
3.3 Market Opportunities
3.3.1 Expansion in emerging markets
3.3.2 Development of innovative materials and technologies
3.3.3 Increasing investment in healthcare infrastructure
3.3.4 Collaborations and partnerships among key players
3.4 Market Trends
3.4.1 Growing adoption of minimally invasive procedures
3.4.2 Integration of 3D printing technology in CMF devices
3.4.3 Rise in patient-centric approaches
3.4.4 Focus on personalized medicine in CMF surgeries
3.5 Government Regulation
3.5.1 FDA regulations on medical devices
3.5.2 CE marking requirements in Europe
3.5.3 Guidelines for clinical trials and approvals
3.5.4 Compliance with international standards (ISO)
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Craniomaxillofacial CMF Devices System Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Craniomaxillofacial CMF Devices System Market Segmentation
8.1 By Product Type
8.1.1 CMF Plate and Screw Fixation
8.1.2 Cranial Flap Fixation
8.1.3 CMF Distraction Devices
8.1.4 Temporomandibular Joint (TMJ) Replacement
8.1.5 Bone Graft Substitutes
8.1.6 Thoracic Fixation
8.1.7 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Ambulatory Surgical Centers (ASCs)
8.2.3 Specialty Clinics
8.2.4 Surgeons / Physician-owned Practices
8.2.5 Others
8.3 By Material
8.3.1 Metal-based Implants (e.g., Titanium, Stainless Steel)
8.3.2 Bioabsorbable Materials
8.3.3 Ceramic-based Materials
8.3.4 Composite / Hybrid Materials
8.3.5 Others
8.4 By Application
8.4.1 Trauma Reconstruction
8.4.2 Orthognathic and Dental Surgery
8.4.3 Neurosurgery & ENT
8.4.4 Cosmetic / Plastic Surgery
8.4.5 Others
8.5 By Region
8.5.1 North America
8.5.2 Europe
8.5.3 Asia-Pacific
8.5.4 Latin America
8.5.5 Middle East & Africa
8.6 By Distribution Channel
8.6.1 Direct Tenders / Institutional Sales
8.6.2 Distributor / Dealer Network
8.6.3 Group Purchasing Organizations (GPOs)
8.6.4 Online / E-Procurement Platforms
8.6.5 Others
8.7 By Technology / Surgical Approach
8.7.1 Conventional / Open Surgical Techniques
8.7.2 Image-Guided and Navigation-assisted Surgery
8.7.3 3D Printing and Patient-specific Implants
8.7.4 Robotic-Assisted Surgery
8.7.5 Others
9. Global Craniomaxillofacial CMF Devices System Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players (KPIs)
9.2.1 Company Name
9.2.2 Global CMF Revenue (US$ Mn)
9.2.3 CMF Revenue CAGR (Historical & Forecast)
9.2.4 CMF Market Share (%) by Region
9.2.5 EBITDA Margin (%) – CMF Segment
9.2.6 R&D Intensity (% of CMF Revenue)
9.2.7 Number of CMF SKUs / Product Lines
9.2.8 New Product Approvals / Launches (3–5 Years)
9.2.9 Geographic Footprint (Countries / Regions Served)
9.2.10 Key Distribution / Hospital Accounts Won (Last 3 Years)
9.2.11 Average Selling Price Positioning (vs. Market Average)
9.2.12 CMF-focused M&A / Strategic Alliances (Last 5 Years)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medtronic plc
9.5.2 Stryker Corporation
9.5.3 DePuy Synthes (Johnson & Johnson)
9.5.4 Zimmer Biomet Holdings, Inc.
9.5.5 KLS Martin Group
9.5.6 Integra LifeSciences Holdings Corporation
9.5.7 Aesculap Implant Systems (B. Braun)
9.5.8 OsteoMed L.P.
9.5.9 Biomet Microfixation (Zimmer Biomet)
9.5.10 CONMED Corporation
9.5.11 B. Braun Melsungen AG
9.5.12 Medartis AG
9.5.13 KLS Martin LP / subsidiaries (regional profiles as relevant)
9.5.14 Other Emerging CMF-focused Players
9.5.15 Regional / Local Manufacturers (Representative Sample)
10. Global Craniomaxillofacial CMF Devices System Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Supplier Selection Criteria
10.1.4 Contracting Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends in Healthcare Facilities
10.2.2 Spending on Advanced Medical Equipment
10.2.3 Budgeting for Surgical Procedures
10.2.4 Financial Planning for CMF Devices
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges Faced by Hospitals
10.3.2 Issues in Ambulatory Surgical Centers
10.3.3 Concerns of Specialty Clinics
10.3.4 Needs of Research Institutions
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training and Support Needs
10.4.3 Technology Acceptance
10.4.4 Feedback Mechanisms
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Success Metrics
10.5.2 Case Studies of Successful Implementations
10.5.3 Future Use Cases
10.5.4 Long-term Value Assessment
11. Global Craniomaxillofacial CMF Devices System Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Solutions
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships Evaluation
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from medical device associations and healthcare market research firms
- Review of published articles in peer-reviewed journals focusing on craniomaxillofacial surgery and device innovations
- Examination of regulatory documents and guidelines from health authorities such as the FDA and EMA
Primary Research
- Interviews with leading surgeons and specialists in craniomaxillofacial procedures
- Surveys conducted with hospital procurement managers and medical device distributors
- Focus groups with patients who have undergone CMF procedures to gather insights on device effectiveness and satisfaction
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and industry surveys
- Triangulation of data from clinical studies, market reports, and expert opinions
- Sanity checks through feedback from a panel of healthcare professionals and market analysts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the global CMF devices market size based on healthcare expenditure and surgical procedure volumes
- Segmentation analysis by product type, including plates, screws, and distraction devices
- Incorporation of demographic trends and population health statistics influencing CMF surgery rates
Bottom-up Modeling
- Collection of sales data from key manufacturers and distributors of CMF devices
- Estimation of market share based on product sales and regional distribution
- Volume x price analysis to derive revenue estimates for each product category
Forecasting & Scenario Analysis
- Utilization of time-series analysis to project market growth based on historical data
- Scenario modeling considering factors such as technological advancements and regulatory changes
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Surgeon Insights on CMF Devices | 110 | Maxillofacial Surgeons, Plastic Surgeons |
| Hospital Procurement Strategies | 90 | Procurement Managers, Supply Chain Directors |
| Patient Experience and Satisfaction | 70 | Patients who have undergone CMF procedures |
| Market Trends and Innovations | 60 | Medical Device R&D Managers, Product Development Leads |
| Regulatory Compliance and Challenges | 50 | Regulatory Affairs Specialists, Quality Assurance Managers |
Frequently Asked Questions
What is the current value of the Global Craniomaxillofacial CMF Devices System Market?
The Global Craniomaxillofacial CMF Devices System Market is valued at approximately USD 1.8 billion, reflecting a significant growth trend driven by factors such as increasing facial trauma incidents and advancements in surgical techniques.