Region:Global
Author(s):Dev
Product Code:KRAA2527
Pages:99
Published On:August 2025
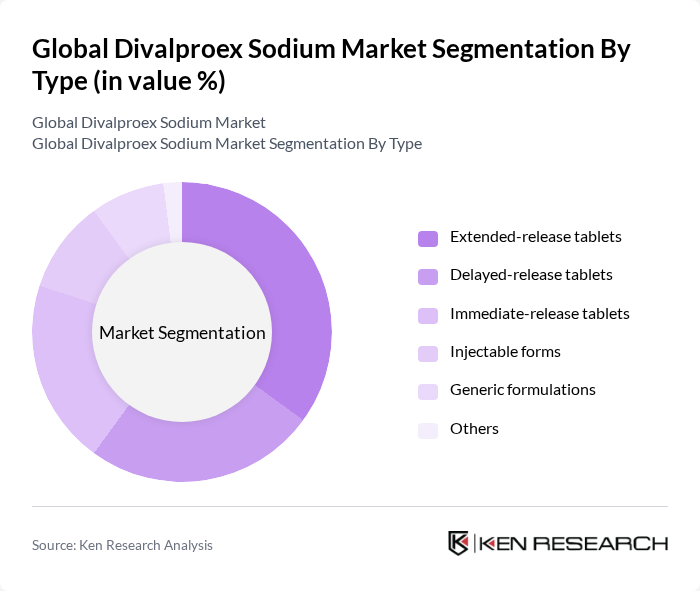
By Type:The market is segmented into various types of Divalproex Sodium formulations, including Extended-release tablets, Delayed-release tablets, Immediate-release tablets, Injectable forms, Generic formulations, and Others. Among these, Extended-release tablets are gaining traction due to their convenience and improved patient compliance. The demand for Delayed-release and Immediate-release tablets remains strong, particularly in acute treatment scenarios. The introduction of generic formulations has increased accessibility and affordability, while injectable forms are primarily utilized in hospital settings for patients unable to take oral medications .
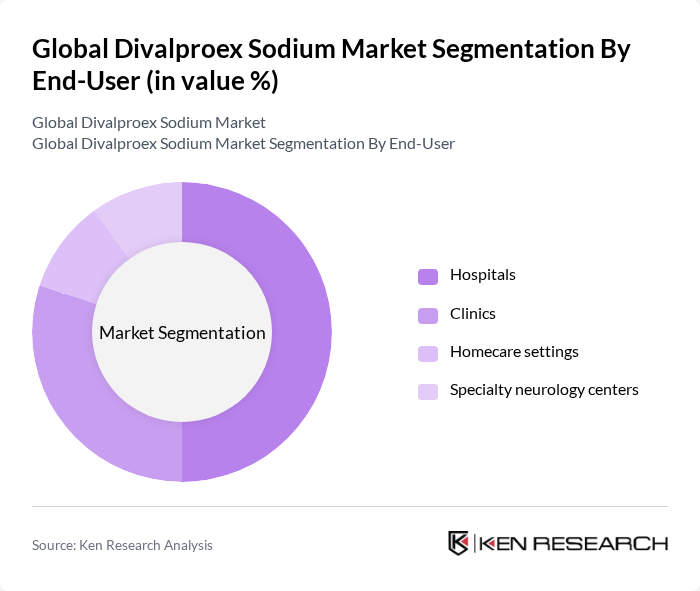
By End-User:The end-user segmentation includes Hospitals, Clinics, Homecare settings, and Specialty neurology centers. Hospitals are the leading end-users due to their capacity to provide comprehensive care and access to advanced treatment options. Clinics and specialty centers also play a significant role, particularly in outpatient settings where ongoing management of conditions like epilepsy is required. The expansion of homecare settings is supported by the availability of user-friendly formulations, while specialty neurology centers focus on complex and refractory cases .
The Global Divalproex Sodium Market is characterized by a dynamic mix of regional and international players. Leading participants such as AbbVie Inc., Teva Pharmaceutical Industries Ltd., Mylan N.V., Sanofi S.A., Aurobindo Pharma Ltd., H. Lundbeck A/S, Pfizer Inc., Novartis AG, Hikma Pharmaceuticals PLC, Amneal Pharmaceuticals Inc., Sandoz International GmbH, Sun Pharmaceutical Industries Ltd., Dr. Reddy's Laboratories Ltd., Zydus Lifesciences Ltd., Cipla Ltd., Lupin Limited, Torrent Pharmaceuticals Ltd., Alkem Laboratories Ltd., Apotex Inc., Glenmark Pharmaceuticals Ltd. contribute to innovation, geographic expansion, and service delivery in this space .
The future of the divalproex sodium market appears promising, driven by ongoing advancements in drug development and a growing focus on personalized medicine. As healthcare systems increasingly adopt telemedicine and remote monitoring, patient access to treatment is expected to improve significantly. Furthermore, the rising demand for combination therapies will likely lead to innovative treatment regimens that enhance efficacy and safety, ultimately benefiting patients and healthcare providers alike. The market is poised for growth as these trends continue to evolve.
| Segment | Sub-Segments |
|---|---|
| By Type | Extended-release tablets Delayed-release tablets Immediate-release tablets Injectable forms Generic formulations Others |
| By End-User | Hospitals Clinics Homecare settings Specialty neurology centers |
| By Distribution Channel | Retail pharmacies Online pharmacies Hospital pharmacies Wholesalers & distributors |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Application | Epilepsy treatment Bipolar disorder management Migraine prevention Off-label uses |
| By Patient Demographics | Pediatric patients Adult patients Geriatric patients |
| By Pricing Tier | Premium Mid-range Economy |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurology Clinics | 60 | Neurologists, Nurse Practitioners |
| Psychiatric Hospitals | 50 | Psychiatrists, Clinical Psychologists |
| Pharmacies | 70 | Pharmacists, Pharmacy Technicians |
| Healthcare Providers | 40 | General Practitioners, Family Medicine Doctors |
| Patient Advocacy Groups | 40 | Patient Representatives, Care Coordinators |
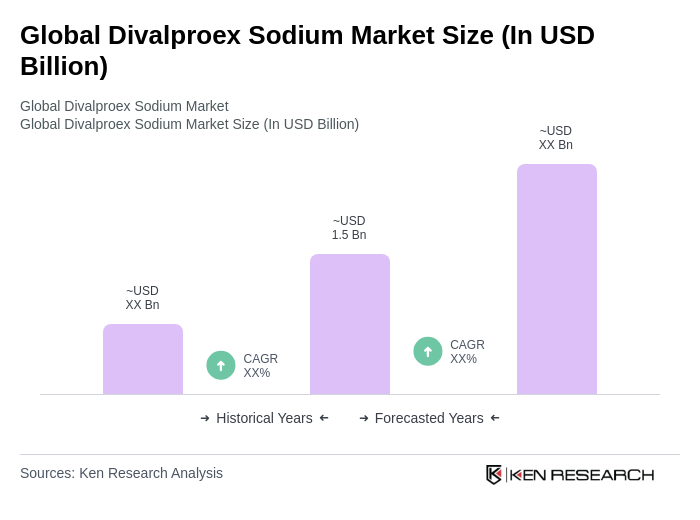
The Global Divalproex Sodium Market is valued at approximately USD 1.5 billion, reflecting a five-year historical analysis. This valuation highlights the market's growth driven by increasing neurological disorder prevalence and rising mental health awareness.