Region:Global
Author(s):Dev
Product Code:KRAC3401
Pages:80
Published On:October 2025
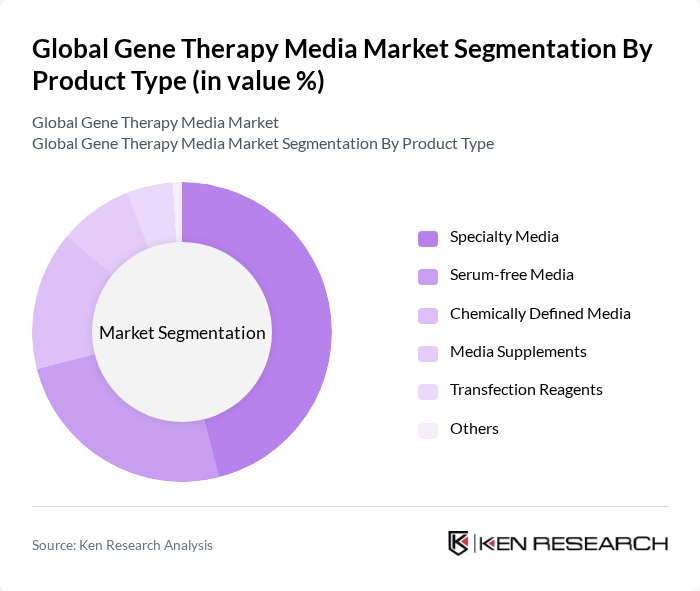
By Product Type:The product type segmentation includes various categories such as Specialty Media, Serum-free Media, Chemically Defined Media, Media Supplements, Transfection Reagents, and Others. Among these, Specialty Media is currently dominating the market due to its tailored formulations that enhance cell growth and productivity in gene therapy applications. The increasing demand for customized solutions in research and clinical settings is driving the growth of this sub-segment .
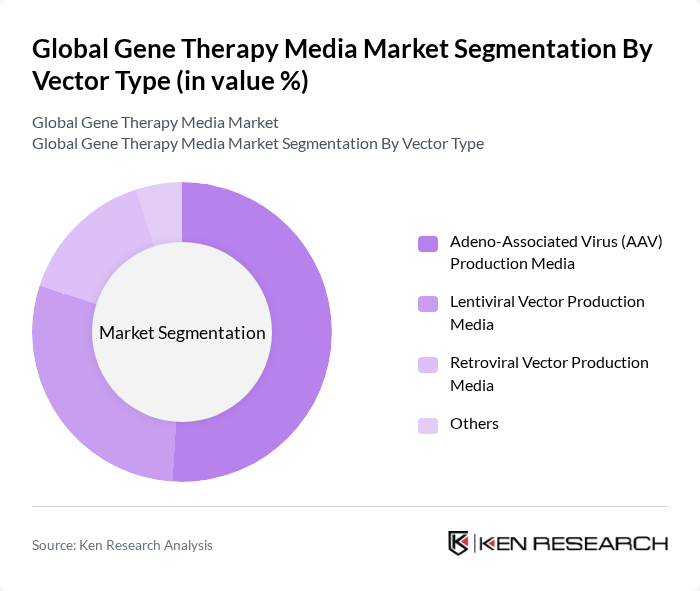
By Vector Type:The vector type segmentation encompasses Adeno-Associated Virus (AAV) Production Media, Lentiviral Vector Production Media, Retroviral Vector Production Media, and Others. The Adeno-Associated Virus (AAV) Production Media segment is leading the market, primarily due to its effectiveness in delivering genetic material into host cells with minimal immune response. This has made AAV a preferred choice for many gene therapy applications, driving its market share significantly .
The Global Gene Therapy Media Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific Inc., Sartorius AG, Merck KGaA (MilliporeSigma), Lonza Group AG, Danaher Corporation (Cytiva), FUJIFILM Holdings Corporation (Irvine Scientific), MP Biomedicals, Corning Incorporated, PAN-Biotech GmbH, Bio-Techne Corporation (R&D Systems, Inc.), InVitria Inc., Florabio AS, Revvity, Inc., Virica Biotech Inc., CellGenix GmbH contribute to innovation, geographic expansion, and service delivery in this space .
The future of the gene therapy media market appears promising, driven by technological advancements and increasing public awareness. As gene editing technologies continue to evolve, the integration of artificial intelligence in research is expected to enhance efficiency and precision. Furthermore, the growing acceptance of personalized medicine will likely lead to increased demand for tailored therapies. These trends indicate a dynamic market landscape, with significant potential for growth and innovation in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Specialty Media Serum-free Media Chemically Defined Media Media Supplements Transfection Reagents Others |
| By Vector Type | Adeno-Associated Virus (AAV) Production Media Lentiviral Vector Production Media Retroviral Vector Production Media Others |
| By Application | Clinical Manufacturing Research & Development Process Development Others |
| By Cell Type | HEK293 Cells CHO Cells Primary T Cells Stem Cells Others |
| By End-User | Pharmaceutical & Biotechnology Companies Contract Research Organizations (CROs) Academic & Research Institutions Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Product Formulation | Liquid Formulations Lyophilized Formulations Others |
| By Pricing Model | Premium Pricing Competitive Pricing Value-Based Pricing Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Gene Therapy | 100 | Oncologists, Clinical Researchers |
| Rare Genetic Disorders | 60 | Geneticists, Patient Advocacy Representatives |
| Regulatory Affairs in Gene Therapy | 50 | Regulatory Affairs Specialists, Compliance Officers |
| Market Access Strategies | 40 | Market Access Managers, Health Economists |
| Clinical Trial Management | 50 | Clinical Trial Managers, Biostatisticians |
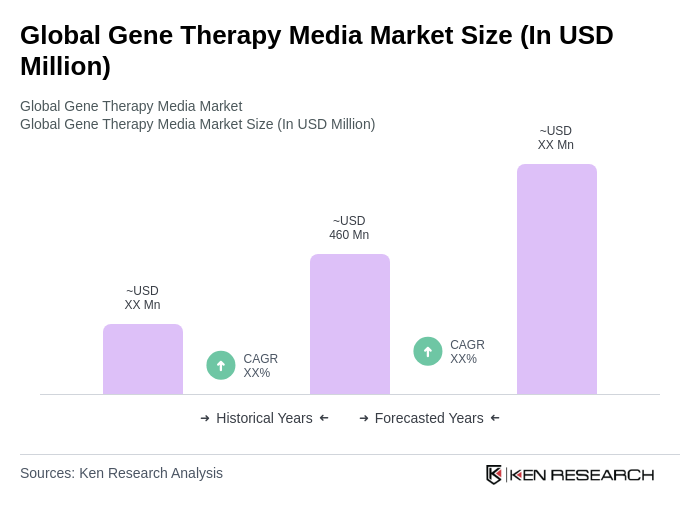
The Global Gene Therapy Media Market is valued at approximately USD 460 million, driven by advancements in gene therapy technologies, increased R&D investments, and a rising prevalence of genetic disorders.