Region:Global
Author(s):Dev
Product Code:KRAD1596
Pages:94
Published On:November 2025
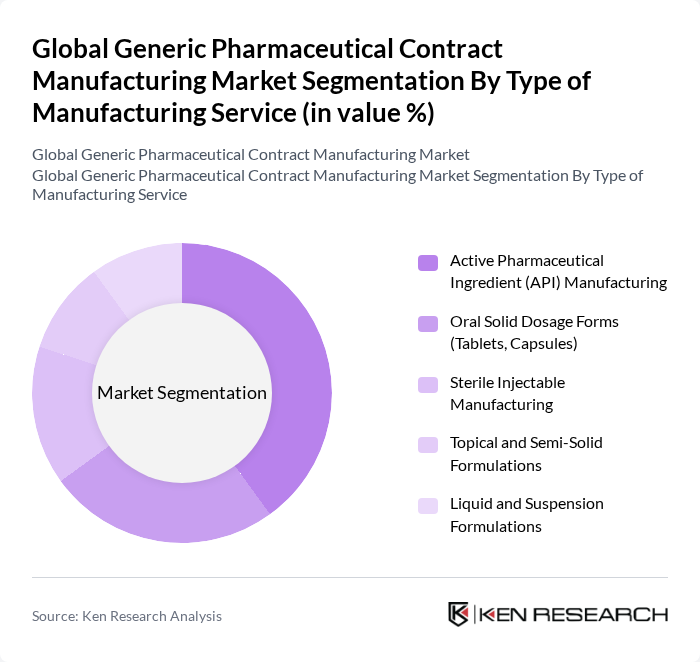
By Type of Manufacturing Service:The market is segmented into various types of manufacturing services, including Active Pharmaceutical Ingredient (API) Manufacturing, Oral Solid Dosage Forms, Sterile Injectable Manufacturing, Topical and Semi-Solid Formulations, and Liquid and Suspension Formulations. Among these, API manufacturing is currently the leading segment due to the increasing demand for raw materials in drug production. The trend towards complex generics, personalized medicine, and biologics is also driving growth in this segment as pharmaceutical companies seek specialized manufacturing expertise and regulatory compliance.
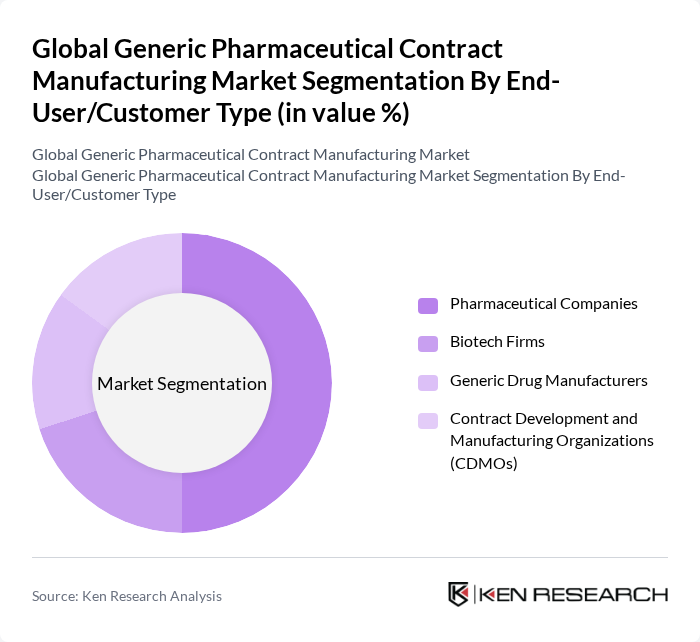
By End-User/Customer Type:The market is categorized into Pharmaceutical Companies, Biotech Firms, Generic Drug Manufacturers, and Contract Development and Manufacturing Organizations (CDMOs). Pharmaceutical companies dominate this segment as they increasingly outsource manufacturing to focus on core competencies such as research and development. This trend is further fueled by the need for cost efficiency, the ability to scale production without significant capital investment, and the growing complexity of drug formulations requiring specialized manufacturing capabilities.
The Global Generic Pharmaceutical Contract Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Teva Pharmaceutical Industries Ltd., Viatris Inc. (formerly Mylan N.V.), Sandoz International GmbH, Sun Pharmaceutical Industries Ltd., Aurobindo Pharma Ltd., Cipla Ltd., Dr. Reddy's Laboratories Ltd., Fresenius Kabi AG, Hikma Pharmaceuticals PLC, Zydus Lifesciences Ltd., Lupin Limited, Perrigo Company plc, Catalent, Inc., Recro Pharma, Inc., and Evonik Industries AG contribute to innovation, geographic expansion, and service delivery in this space. These companies are expanding their service portfolios, investing in advanced manufacturing technologies, and strengthening regulatory compliance to meet the evolving needs of the global generic pharmaceutical sector.
The future of the generic pharmaceutical contract manufacturing market appears promising, driven by the increasing demand for affordable medications and the rise of chronic diseases. As manufacturers adopt advanced technologies, operational efficiencies will improve, enabling quicker responses to market needs. Additionally, the expansion into emerging markets will provide new growth avenues. Companies that prioritize strategic partnerships and invest in research and development will likely lead the way in innovation and sustainability, positioning themselves favorably in the evolving landscape.
| Segment | Sub-Segments |
|---|---|
| By Type of Manufacturing Service | Active Pharmaceutical Ingredient (API) Manufacturing Oral Solid Dosage Forms (Tablets, Capsules) Sterile Injectable Manufacturing Topical and Semi-Solid Formulations Liquid and Suspension Formulations |
| By End-User/Customer Type | Pharmaceutical Companies Biotech Firms Generic Drug Manufacturers Contract Development and Manufacturing Organizations (CDMOs) |
| By Therapeutic Area | Cardiovascular Diseases Oncology Infectious Diseases Respiratory Disorders Gastrointestinal Disorders |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Manufacturing Process Technology | Batch Manufacturing Continuous Manufacturing Automated Manufacturing Systems |
| By Scale of Operations | Small-Scale Manufacturing Mid-Scale Manufacturing Large-Scale Manufacturing |
| By Regulatory Compliance Level | FDA-Compliant Facilities EMA-Compliant Facilities WHO-GMP Certified Facilities |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Generic Drug Manufacturing | 100 | Operations Managers, Production Supervisors |
| Contract Manufacturing Services | 60 | Business Development Managers, Sales Directors |
| Quality Control in Generics | 50 | Quality Assurance Managers, Compliance Officers |
| Regulatory Affairs in Pharmaceuticals | 40 | Regulatory Affairs Specialists, Legal Advisors |
| Market Access Strategies | 45 | Market Access Managers, Health Economists |
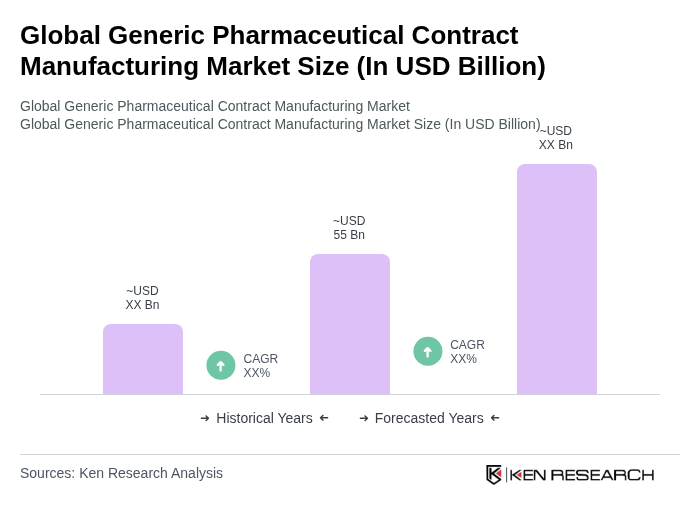
The Global Generic Pharmaceutical Contract Manufacturing Market is valued at approximately USD 55 billion, driven by the increasing demand for affordable medications and the rise of chronic diseases, alongside the trend of outsourcing manufacturing processes by pharmaceutical companies.