About the Report
Base Year 2024Global Oncology Clinical Trials Market Overview
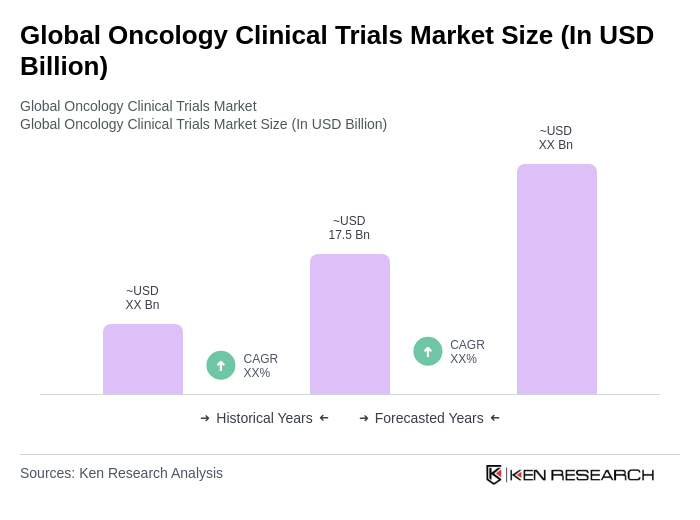
- The Global Oncology Clinical Trials Market is valued at USD 17.5 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of cancer worldwide, rapid advancements in precision medicine and biomarker-driven therapies, and the rising demand for personalized treatment approaches. The market is further supported by significant investments in research and development from pharmaceutical and biotechnology companies, as well as government initiatives focused on improving cancer treatment outcomes. Additional drivers include the expansion of trial sites in emerging markets and the integration of real-world data into hybrid evidence protocols.
- Key players in this market include the United States, Germany, and Japan, which dominate due to their robust healthcare infrastructure, substantial investment in oncology research and development, and large patient populations. North America, particularly the United States, holds the largest market share, attributed to advanced healthcare systems, a mature pharmaceutical industry, and a high prevalence of cancer cases. The presence of leading pharmaceutical and biotechnology companies in these countries further strengthens their market position, enabling them to conduct a higher volume of clinical trials and bring innovative therapies to market.
- In 2023, the U.S. Food and Drug Administration (FDA) implemented new regulations to streamline the clinical trial process for oncology drugs. This includes the Real-Time Oncology Review (RTOR) program, which enables expedited review of clinical trial data and accelerates the approval process for promising cancer therapies. The RTOR initiative, as outlined in the FDA's Real-Time Oncology Review Program Guidance for Industry (U.S. Food and Drug Administration, 2023), aims to enhance patient access to innovative treatments while maintaining rigorous safety and efficacy standards .
Global Oncology Clinical Trials Market Segmentation
By Trial Phase:
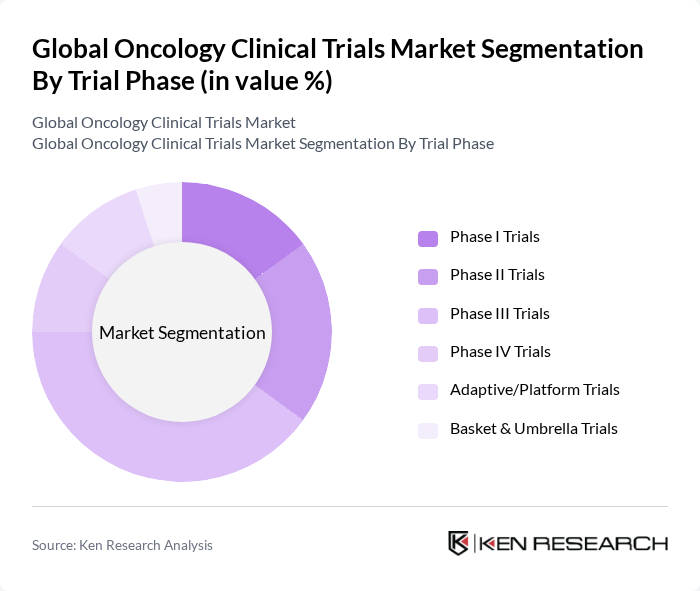
The trial phase segmentation includes Phase I Trials, Phase II Trials, Phase III Trials, Phase IV Trials, Adaptive/Platform Trials, and Basket & Umbrella Trials. Among these, Phase III Trials dominate the market due to their pivotal role in determining the efficacy and safety of new treatments prior to regulatory approval. These trials typically involve larger patient populations and are essential for generating comprehensive data on long-term outcomes and adverse effects, making them a focal point for pharmaceutical companies seeking market authorization for new oncology therapies.
By Cancer Type (Indication):
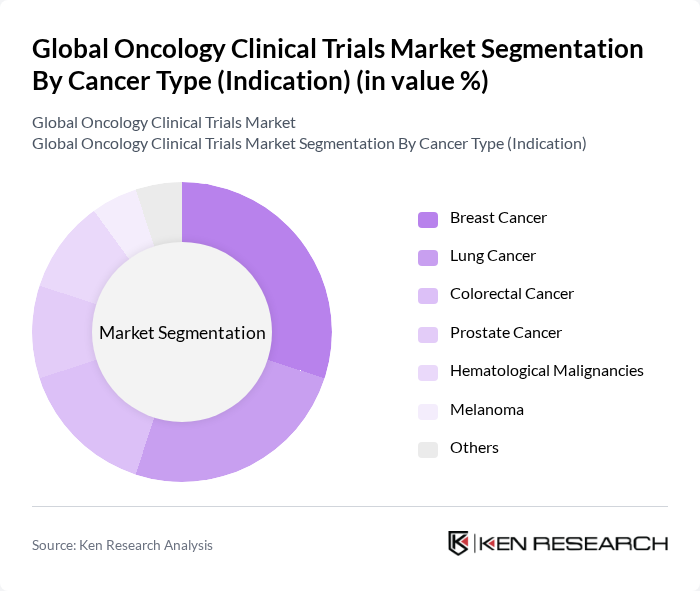
This segmentation includes Breast Cancer, Lung Cancer, Colorectal Cancer, Prostate Cancer, Hematological Malignancies (Leukemia, Lymphoma, Myeloma), Melanoma, and Others. Breast Cancer trials are the most prevalent, driven by high global incidence rates and ongoing research into targeted and personalized therapies. The focus on precision medicine and the development of innovative treatment options for breast cancer patients have led to a significant number of clinical trials in this area, making it a key segment in the oncology clinical trials market. Lung cancer also represents a major segment due to its high mortality and incidence, followed by colorectal and prostate cancers.
Global Oncology Clinical Trials Market Competitive Landscape
The Global Oncology Clinical Trials Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pfizer Inc., Merck & Co., Inc., F. Hoffmann-La Roche AG, Novartis AG, Bristol-Myers Squibb Company, AstraZeneca PLC, Johnson & Johnson (Janssen Pharmaceuticals), Eli Lilly and Company, Amgen Inc., Gilead Sciences, Inc., AbbVie Inc., Sanofi S.A., Takeda Pharmaceutical Company Limited, Bayer AG, ICON plc, Syneos Health, Inc., IQVIA Holdings Inc., Parexel International Corporation contribute to innovation, geographic expansion, and service delivery in this space.
Global Oncology Clinical Trials Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Cancer:The global cancer burden is projected to reach approximately 29.5 million new cases in future, according to the World Health Organization. This rising incidence drives demand for innovative treatments and clinical trials. An estimated 1.9 million new cancer cases are expected in the United States in future, highlighting the urgent need for effective therapies. This growing patient population fuels the oncology clinical trials market, as pharmaceutical companies seek to develop targeted therapies to address this escalating health crisis.
- Advancements in Biotechnology:The biotechnology sector is experiencing rapid growth, with global investments reaching approximately $500 billion in future. Innovations in gene therapy, monoclonal antibodies, and immunotherapy are revolutionizing cancer treatment. For instance, CAR-T cell therapy has shown remarkable efficacy in treating certain blood cancers, leading to increased clinical trial activity. These advancements not only enhance treatment options but also attract significant funding, further propelling the oncology clinical trials market forward as companies strive to bring novel therapies to market.
- Rising Investment in R&D:Global spending on pharmaceutical R&D is projected to exceed $200 billion in future, with oncology research receiving a substantial share. This investment is driven by the need for innovative cancer therapies and the potential for high returns on successful drug development. Companies are increasingly allocating resources to oncology trials, recognizing the lucrative market opportunities. As a result, the influx of capital into oncology research is expected to accelerate the pace of clinical trials, fostering a more dynamic and competitive landscape.
Market Challenges
- High Costs of Clinical Trials:The average cost of conducting a clinical trial can exceed $2.6 billion, according to the Tufts Center for the Study of Drug Development. These exorbitant costs pose significant barriers for many companies, particularly smaller firms with limited resources. The financial burden can lead to reduced trial initiation and delays in bringing new therapies to market. Consequently, the high costs associated with oncology clinical trials remain a critical challenge that must be addressed to ensure continued innovation in cancer treatment.
- Regulatory Hurdles:Navigating the complex regulatory landscape is a significant challenge for oncology clinical trials. In future, the FDA and EMA are expected to implement stricter guidelines, increasing the time and resources required for compliance. Delays in obtaining necessary approvals can hinder trial timelines and increase costs. Furthermore, the evolving nature of regulations can create uncertainty for sponsors, making it essential for companies to stay informed and adaptable to maintain their competitive edge in the oncology market.
Global Oncology Clinical Trials Market Future Outlook
The oncology clinical trials market is poised for transformative growth, driven by technological advancements and an increasing focus on patient-centric approaches. As decentralized trials gain traction, they promise to enhance patient recruitment and retention, making participation more accessible. Additionally, the integration of artificial intelligence and machine learning is expected to streamline data management and analysis, improving trial efficiency. These trends indicate a shift towards more innovative and efficient trial methodologies, positioning the market for significant advancements in cancer treatment and research.
Market Opportunities
- Growth in Personalized Medicine:The personalized medicine market is projected to reach $2.5 trillion in future, creating substantial opportunities for oncology trials focused on tailored therapies. This trend emphasizes the need for clinical trials that explore genetic and molecular profiling, allowing for more effective and targeted treatment options for patients, thereby enhancing trial success rates and patient outcomes.
- Emerging Markets Expansion:Emerging markets, particularly in Asia-Pacific and Latin America, are witnessing a surge in cancer cases, with an estimated 10 million new cases expected in future. This growth presents significant opportunities for oncology clinical trials, as companies seek to tap into these regions for patient recruitment and collaboration. Expanding into these markets can enhance trial diversity and accelerate the development of new therapies.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Trial Phase | Phase I Trials Phase II Trials Phase III Trials Phase IV Trials Adaptive/Platform Trials Basket & Umbrella Trials |
| By Cancer Type (Indication) | Breast Cancer Lung Cancer Colorectal Cancer Prostate Cancer Hematological Malignancies (Leukemia, Lymphoma, Myeloma) Melanoma Others |
| By Study Design | Interventional Trials Observational Trials Expanded Access Trials |
| By Geography | North America (U.S., Canada, Mexico) Europe (Germany, U.K., France, Italy, Spain, Rest of Europe) Asia-Pacific (China, Japan, India, Rest of APAC) Latin America (Brazil, Rest of Latin America) Middle East & Africa (UAE, South Africa, Rest of MEA) |
| By Sponsor Type | Pharmaceutical Companies Biotechnology Firms Academic & Research Institutions Contract Research Organizations (CROs) |
| By Patient Demographics | Adult Patients Pediatric Patients Geriatric Patients |
| By Funding Source | Government Funding Private Funding Non-Profit Organizations Industry Funding Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Pharmaceutical and Biotechnology Companies
Clinical Research Organizations (CROs)
Healthcare Providers and Hospitals
Health Insurance Companies
Patient Advocacy Groups
Biomarker and Diagnostic Test Developers
Players Mentioned in the Report:
Pfizer Inc.
Merck & Co., Inc.
F. Hoffmann-La Roche AG
Novartis AG
Bristol-Myers Squibb Company
AstraZeneca PLC
Johnson & Johnson (Janssen Pharmaceuticals)
Eli Lilly and Company
Amgen Inc.
Gilead Sciences, Inc.
AbbVie Inc.
Sanofi S.A.
Takeda Pharmaceutical Company Limited
Bayer AG
ICON plc
Syneos Health, Inc.
IQVIA Holdings Inc.
Parexel International Corporation
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Oncology Clinical Trials Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Oncology Clinical Trials Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Oncology Clinical Trials Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of cancer
3.1.2 Advancements in biotechnology
3.1.3 Rising investment in R&D
3.1.4 Expanding patient population
3.2 Market Challenges
3.2.1 High costs of clinical trials
3.2.2 Regulatory hurdles
3.2.3 Patient recruitment difficulties
3.2.4 Data management complexities
3.3 Market Opportunities
3.3.1 Growth in personalized medicine
3.3.2 Emerging markets expansion
3.3.3 Collaborations with technology firms
3.3.4 Increased focus on rare cancers
3.4 Market Trends
3.4.1 Shift towards decentralized trials
3.4.2 Integration of AI and machine learning
3.4.3 Rise of patient-centric approaches
3.4.4 Enhanced data analytics capabilities
3.5 Government Regulation
3.5.1 FDA guidelines for oncology trials
3.5.2 EMA regulations on clinical trials
3.5.3 ICH GCP compliance
3.5.4 Data protection laws impacting trials
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Oncology Clinical Trials Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Oncology Clinical Trials Market Segmentation
8.1 By Trial Phase
8.1.1 Phase I Trials
8.1.2 Phase II Trials
8.1.3 Phase III Trials
8.1.4 Phase IV Trials
8.1.5 Adaptive/Platform Trials
8.1.6 Basket & Umbrella Trials
8.2 By Cancer Type (Indication)
8.2.1 Breast Cancer
8.2.2 Lung Cancer
8.2.3 Colorectal Cancer
8.2.4 Prostate Cancer
8.2.5 Hematological Malignancies (Leukemia, Lymphoma, Myeloma)
8.2.6 Melanoma
8.2.7 Others
8.3 By Study Design
8.3.1 Interventional Trials
8.3.2 Observational Trials
8.3.3 Expanded Access Trials
8.4 By Geography
8.4.1 North America (U.S., Canada, Mexico)
8.4.2 Europe (Germany, U.K., France, Italy, Spain, Rest of Europe)
8.4.3 Asia-Pacific (China, Japan, India, Rest of APAC)
8.4.4 Latin America (Brazil, Rest of Latin America)
8.4.5 Middle East & Africa (UAE, South Africa, Rest of MEA)
8.5 By Sponsor Type
8.5.1 Pharmaceutical Companies
8.5.2 Biotechnology Firms
8.5.3 Academic & Research Institutions
8.5.4 Contract Research Organizations (CROs)
8.6 By Patient Demographics
8.6.1 Adult Patients
8.6.2 Pediatric Patients
8.6.3 Geriatric Patients
8.7 By Funding Source
8.7.1 Government Funding
8.7.2 Private Funding
8.7.3 Non-Profit Organizations
8.7.4 Industry Funding
8.7.5 Others
9. Global Oncology Clinical Trials Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Size (Large, Medium, Small by global trial volume)
9.2.3 Oncology Clinical Trial Revenue
9.2.4 Number of Oncology Trials Conducted (Annual/Total)
9.2.5 Global Geographic Footprint (Number of Countries/Regions Active)
9.2.6 Clinical Trial Success Rate (Oncology-specific)
9.2.7 Average Patient Recruitment Time
9.2.8 R&D Spend on Oncology Trials
9.2.9 Pipeline Size (Number of Oncology Assets in Trials)
9.2.10 Strategic Partnerships/Collaborations in Oncology
9.2.11 Digital/Decentralized Trial Capabilities
9.2.12 Regulatory Approval Rate (Oncology)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Pfizer Inc.
9.5.2 Merck & Co., Inc.
9.5.3 F. Hoffmann-La Roche AG
9.5.4 Novartis AG
9.5.5 Bristol-Myers Squibb Company
9.5.6 AstraZeneca PLC
9.5.7 Johnson & Johnson (Janssen Pharmaceuticals)
9.5.8 Eli Lilly and Company
9.5.9 Amgen Inc.
9.5.10 Gilead Sciences, Inc.
9.5.11 AbbVie Inc.
9.5.12 Sanofi S.A.
9.5.13 Takeda Pharmaceutical Company Limited
9.5.14 Bayer AG
9.5.15 ICON plc
9.5.16 Syneos Health, Inc.
9.5.17 IQVIA Holdings Inc.
9.5.18 Parexel International Corporation
10. Global Oncology Clinical Trials Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Government health departments
10.1.2 Research institutions
10.1.3 Non-profit organizations
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in clinical trial infrastructure
10.2.2 Funding for research initiatives
10.2.3 Expenditure on technology integration
10.3 Pain Point Analysis by End-User Category
10.3.1 Delays in trial approvals
10.3.2 High operational costs
10.3.3 Difficulty in patient recruitment
10.4 User Readiness for Adoption
10.4.1 Awareness of clinical trial benefits
10.4.2 Accessibility of trial information
10.4.3 Support from healthcare providers
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Evaluation of trial outcomes
10.5.2 Long-term patient follow-up
10.5.3 Expansion into new therapeutic areas
11. Global Oncology Clinical Trials Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Key partnerships and alliances
1.5 Customer segments targeting
1.6 Cost structure analysis
1.7 Competitive landscape overview
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience engagement
2.4 Digital marketing initiatives
2.5 Content marketing strategies
3. Distribution Plan
3.1 Urban retail vs rural NGO tie-ups
3.2 Direct sales strategies
3.3 Online distribution channels
3.4 Partnerships with healthcare providers
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands
4.3 Competitive pricing analysis
5. Unmet Demand & Latent Needs
5.1 Category gaps
5.2 Consumer segments
5.3 Emerging trends identification
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer feedback mechanisms
7. Value Proposition
7.1 Sustainability
7.2 Integrated supply chains
7.3 Unique selling points
8. Key Activities
8.1 Regulatory compliance
8.2 Branding
8.3 Distribution setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix
9.1.2 Pricing band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target countries
9.2.2 Compliance roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone tracking
15.2.2 Performance metrics
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published reports from global health organizations and oncology research institutions
- Review of clinical trial registries such as ClinicalTrials.gov and WHO International Clinical Trials Registry Platform
- Examination of market reports and white papers from industry analysts focusing on oncology therapeutics
Primary Research
- Interviews with oncologists and clinical trial coordinators to gather insights on trial designs and patient recruitment
- Surveys with pharmaceutical companies involved in oncology to understand their pipeline and investment strategies
- Field interviews with regulatory affairs specialists to discuss compliance and approval processes for oncology trials
Validation & Triangulation
- Cross-validation of data through multiple sources including academic publications and industry reports
- Triangulation of findings from primary interviews with secondary data to ensure consistency
- Sanity checks conducted through expert panel reviews comprising oncologists and clinical researchers
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the global oncology clinical trials market size based on total healthcare expenditure and oncology drug sales
- Segmentation of market data by trial phase (Phase I, II, III) and therapeutic area (e.g., breast cancer, lung cancer)
- Incorporation of trends in patient demographics and disease prevalence rates in oncology
Bottom-up Modeling
- Collection of data on the number of active clinical trials from major pharmaceutical companies and CROs
- Estimation of average costs associated with conducting clinical trials, including patient recruitment and site management
- Volume x cost analysis based on trial phases and therapeutic areas to derive market size
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as technological advancements and regulatory changes
- Scenario modeling based on potential shifts in healthcare policies and funding for oncology research
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Phase I Clinical Trials | 60 | Clinical Trial Managers, Oncologists |
| Phase II Clinical Trials | 50 | Research Scientists, Regulatory Affairs Specialists |
| Phase III Clinical Trials | 70 | Clinical Research Coordinators, Medical Directors |
| Patient Recruitment Strategies | 40 | Patient Advocacy Group Leaders, Oncologists |
| Oncology Drug Development Trends | 45 | Pharmaceutical Executives, Market Access Managers |
Frequently Asked Questions
What is the current value of the Global Oncology Clinical Trials Market?
The Global Oncology Clinical Trials Market is valued at approximately USD 17.5 billion, driven by the increasing prevalence of cancer, advancements in precision medicine, and significant investments in research and development from pharmaceutical and biotechnology companies.