Region:Global
Author(s):Geetanshi
Product Code:KRAC8225
Pages:81
Published On:November 2025
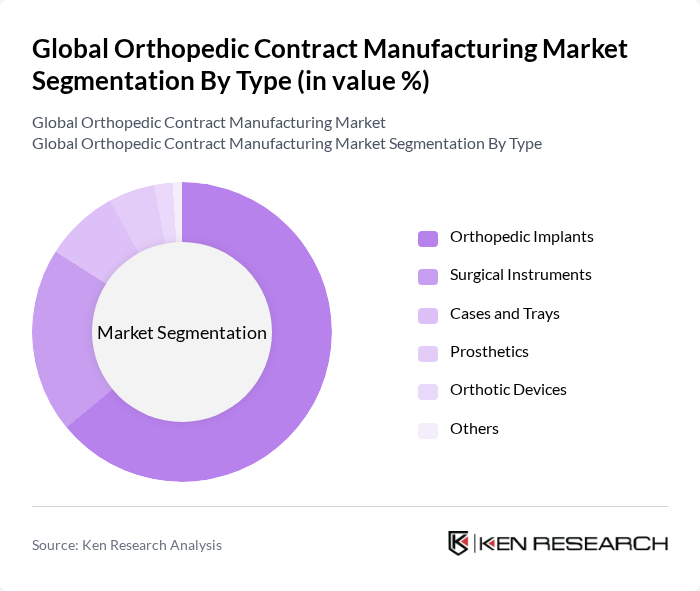
By Type:The orthopedic contract manufacturing market is segmented into various types, including orthopedic implants, surgical instruments, cases and trays, prosthetics, orthotic devices, and others. Among these, orthopedic implants are the most significant segment, driven by the increasing number of joint replacement surgeries and advancements in implant technology. Surgical instruments also hold a substantial share due to their essential role in orthopedic procedures.
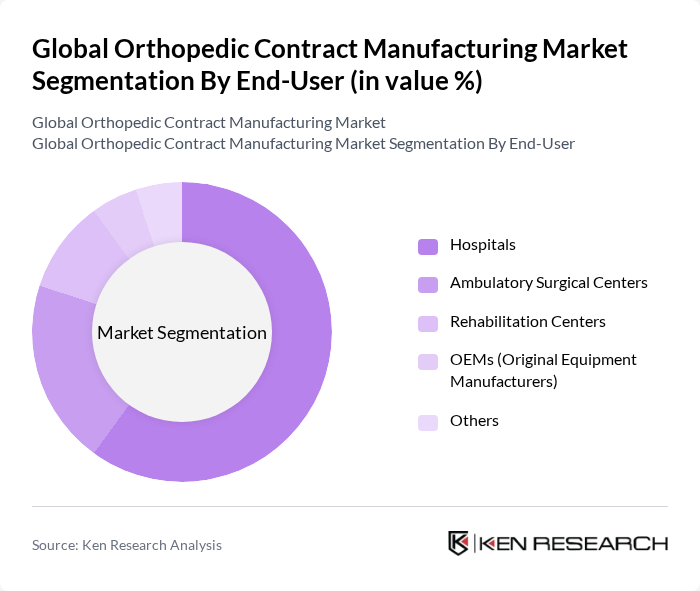
By End-User:The market is also segmented by end-user, which includes hospitals, ambulatory surgical centers, rehabilitation centers, OEMs (Original Equipment Manufacturers), and others. Hospitals are the leading end-user segment, primarily due to the high volume of orthopedic surgeries performed in these facilities. Ambulatory surgical centers are gaining traction as they offer cost-effective and efficient surgical options, contributing to their growing market share.
The Global Orthopedic Contract Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Orchid Orthopedic Solutions, Tecomet Inc., Viant Medical, Paragon Medical, Avalign Technologies, Medtronic, Stryker Corporation, Zimmer Biomet, Smith & Nephew, DePuy Synthes (Johnson & Johnson), NuVasive, Orthofix Medical, Aesculap Implant Systems (B. Braun Melsungen AG), Conmed Corporation, Arthrex, Medacta International, RTI Surgical, Exactech, Marle Group, LISI Medical contribute to innovation, geographic expansion, and service delivery in this space.
The orthopedic contract manufacturing market is poised for significant growth, driven by technological advancements and demographic shifts. The integration of digital technologies, such as AI and IoT, is expected to enhance manufacturing processes and improve product quality. Furthermore, the trend towards personalized medicine will likely lead to increased demand for customized orthopedic solutions, allowing manufacturers to cater to specific patient needs while optimizing production efficiency and reducing costs.
| Segment | Sub-Segments |
|---|---|
| By Type | Orthopedic Implants Surgical Instruments Cases and Trays Prosthetics Orthotic Devices Others |
| By End-User | Hospitals Ambulatory Surgical Centers Rehabilitation Centers OEMs (Original Equipment Manufacturers) Others |
| By Region | North America Europe Asia Pacific Latin America Middle East & Africa |
| By Material | Metal Polymer Ceramic Composite Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Application | Joint Replacement Spine Surgery Trauma Surgery Sports Medicine Others |
| By Technology | Traditional Manufacturing Additive Manufacturing (3D Printing) Computer Numerical Control (CNC) Machining Injection Molding Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Orthopedic Implants | 120 | Orthopedic Surgeons, Hospital Administrators |
| Orthopedic Instruments | 90 | Procurement Managers, Operating Room Coordinators |
| Biologics and Regenerative Medicine | 60 | Clinical Researchers, Product Development Managers |
| Orthopedic Rehabilitation Devices | 50 | Physical Therapists, Rehabilitation Center Directors |
| Market Trends and Innovations | 70 | Industry Analysts, Medical Device Consultants |
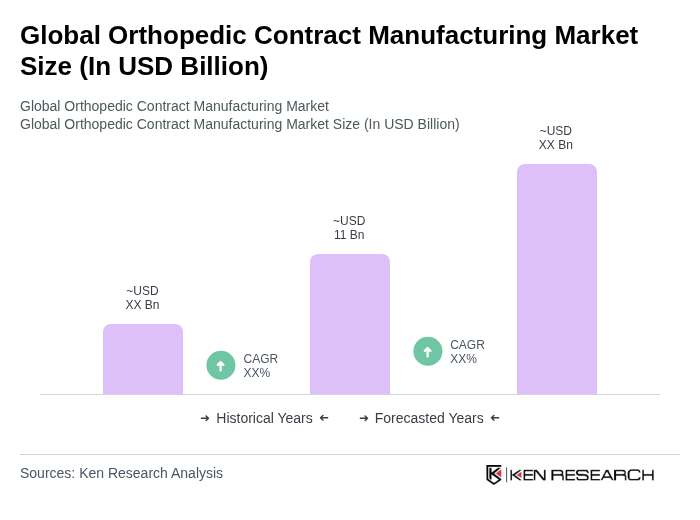
The Global Orthopedic Contract Manufacturing Market is valued at approximately USD 11 billion, driven by factors such as the increasing prevalence of orthopedic disorders, advancements in medical technology, and a growing geriatric population.