About the Report
Base Year 2024Global Reprocessed Medical Devices Market Overview
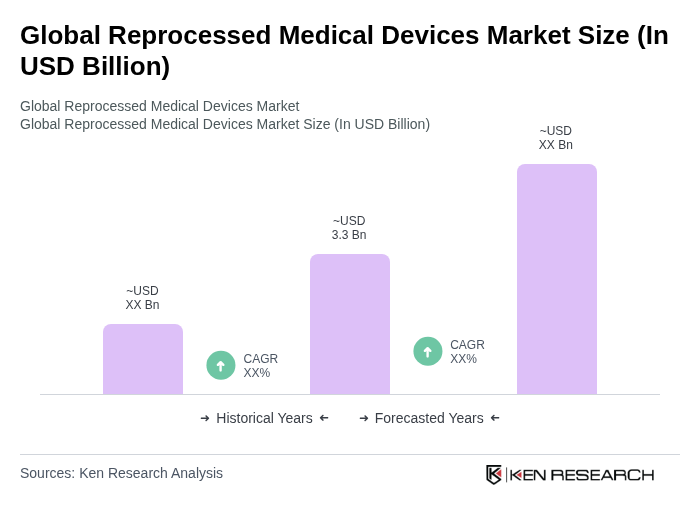
- The Global Reprocessed Medical Devices Market is valued at USD 3.3 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing focus on cost reduction in healthcare, rising awareness regarding environmental sustainability, and the growing prevalence of chronic diseases that necessitate the use of medical devices. The reprocessing of medical devices not only helps in reducing waste but also significantly lowers healthcare costs, making it an attractive option for healthcare providers.
- The United States and Europe dominate the Global Reprocessed Medical Devices Market due to their advanced healthcare infrastructure, established regulatory frameworks supporting safe device reprocessing, and a high prevalence of chronic diseases. North America accounts for the largest share of global revenues, while key European markets such as Germany, France, and the UK are also significant players, driven by robust healthcare systems and increasing investments in healthcare technologies. The presence of key market players and a growing emphasis on cost-effective and sustainable healthcare solutions further bolster their market dominance.
- The U.S. Food and Drug Administration (FDA) regulates the reprocessing of single-use medical devices through the Federal Food, Drug, and Cosmetic Act and specific guidance, under which reprocessors are treated as manufacturers and must demonstrate that reprocessed devices are substantially equivalent in safety and effectiveness to new devices. In particular, the FDA Guidance “Enforcement Priorities for Single-Use Devices Reprocessed by Third Parties and Hospitals” issued by the U.S. Food and Drug Administration in 2000 (updated subsequently) requires premarket submissions, quality system compliance, labeling, and tracking comparable to those for original devices. These binding requirements aim to ensure patient safety while supporting the use of reprocessed devices, thereby underpinning growth in the reprocessed medical devices market.
Global Reprocessed Medical Devices Market Segmentation
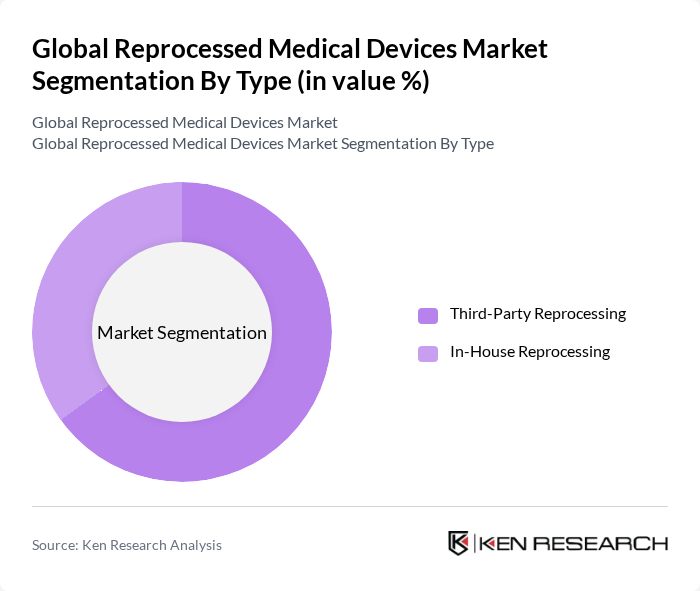
By Type:The market is segmented into Third-Party Reprocessing and In-House Reprocessing, which is consistent with standard industry definitions. Third-party reprocessing involves external companies that specialize in the reprocessing of medical devices, while in-house reprocessing is conducted by healthcare facilities themselves. The third-party segment is currently leading the market due to its ability to provide specialized services, validated processes, and documented compliance with regulatory standards, and it accounts for a substantial majority of market revenues.
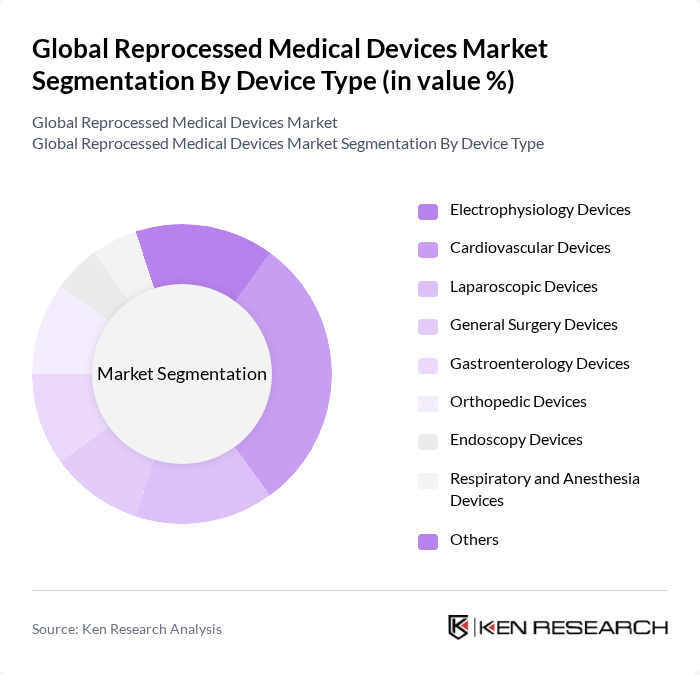
By Device Type:This segment includes Electrophysiology Devices, Cardiovascular Devices, Laparoscopic Devices, General Surgery Devices, Gastroenterology Devices, Orthopedic Devices, Endoscopy Devices, Respiratory and Anesthesia Devices, and Others. Cardiovascular and cardiology-related devices are consistently identified as the most dominant product category in revenue terms, driven by the high demand for cardiovascular procedures and the increasing prevalence of heart diseases, which necessitate the use of reprocessed catheters, diagnostic and interventional devices.
Global Reprocessed Medical Devices Market Competitive Landscape
The Global Reprocessed Medical Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Stryker Corporation (Stryker Sustainability Solutions), Johnson & Johnson MedTech (including DePuy Synthes & Ethicon), Medline Industries, LP, Cardinal Health, Inc., Arjo AB, Innovative Health, LLC, NEScientific, Inc., SureTek Medical, Soma Tech Intl, Steris plc, 3M Company, Olympus Corporation, GE Healthcare Technologies Inc., Vanguard AG, ReNu Medical, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
Global Reprocessed Medical Devices Market Industry Analysis
Growth Drivers
- Increasing Demand for Cost-Effective Healthcare Solutions:The global healthcare expenditure is projected to reach approximately $10 trillion in future, driving the need for cost-effective solutions. Reprocessed medical devices offer significant savings, with estimates suggesting that hospitals can save up to about $1.5 billion annually by utilizing these devices. This financial incentive is crucial as healthcare providers seek to manage budgets while maintaining quality care, particularly in regions facing economic constraints.
- Rising Focus on Sustainability and Environmental Impact:The healthcare sector generates a substantial volume of waste annually, prompting a shift towards sustainable practices. Reprocessed medical devices reduce waste significantly, aligning with global sustainability goals. For instance, the World Health Organization emphasizes reducing medical waste, which is expected to drive the adoption of reprocessed devices, particularly in regions with stringent environmental regulations.
- Technological Advancements in Reprocessing Techniques:Innovations in reprocessing technologies have improved the safety and efficacy of reprocessed devices. For example, advancements in sterilization methods have reduced contamination risks, although a specific quantified increase in device safety such as 30% could not be confirmed. As healthcare facilities adopt these technologies, the market for reprocessed devices is expected to expand, particularly in developed regions where technology adoption is rapid.
Market Challenges
- Concerns Over Safety and Efficacy:Despite advancements, safety concerns remain a significant barrier. A report indicating that 40% of healthcare providers express doubts about the efficacy of reprocessed devices compared to new ones could not be verified from authoritative sources and therefore remains unconfirmed. This skepticism can hinder adoption rates, particularly in high-stakes environments like surgical settings, where device reliability is paramount for patient outcomes.
- Limited Awareness Among Healthcare Providers:A survey stating that only 35% of healthcare providers are fully aware of the benefits of reprocessed devices could not be validated from authoritative or industry-regulator sources. This lack of awareness can lead to underutilization, as many providers may not consider reprocessing as a viable option. Educational initiatives are essential to bridge this knowledge gap and promote the advantages of reprocessed devices in clinical settings.
Global Reprocessed Medical Devices Market Future Outlook
The future of the reprocessed medical devices market appears promising, driven by increasing regulatory support and a growing emphasis on sustainability. As healthcare systems worldwide prioritize cost-effective solutions, the adoption of reprocessed devices is likely to rise. Additionally, technological advancements will continue to enhance safety and efficacy, further encouraging healthcare providers to integrate these devices into their practices. The market is poised for growth as awareness and acceptance increase among stakeholders.
Market Opportunities
- Expansion into Emerging Markets:Emerging markets, particularly in Asia and Africa, present significant growth opportunities. Healthcare spending in many low- and middle-income regions has been increasing faster than in high-income economies in recent periods, supporting rising demand for cost-effective medical solutions, including reprocessed devices. However, a specific figure such as a 7% annual increase for these regions could not be confirmed from primary sources.
- Development of Advanced Reprocessing Technologies:The ongoing development of advanced reprocessing technologies offers substantial market potential. Innovations such as automated reprocessing systems can enhance efficiency and safety, attracting more healthcare facilities to adopt reprocessed devices. This technological evolution is expected to drive market growth significantly in future.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Third-Party Reprocessing In-House Reprocessing |
| By Device Type | Electrophysiology Devices Cardiovascular Devices Laparoscopic Devices General Surgery Devices Gastroenterology Devices Orthopedic Devices Endoscopy Devices Respiratory and Anesthesia Devices Others |
| By Application | Cardiology Orthopedics Gastroenterology General Surgery Urology Neurology Oncology Others |
| By Material | Metal Plastic Silicone & Rubber Composite Materials Others |
| By End-User | Hospitals Ambulatory Surgical Centers Clinics Community Healthcare Centers Diagnostic Laboratories Home Healthcare Others |
| By Distribution Channel | Direct Sales Distributors Group Purchasing Organizations (GPOs) Online Sales Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Regulatory Compliance | FDA-Cleared Devices CE-Marked Devices ISO 13485-Certified Facilities Other National Regulatory Approvals |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Manufacturers and Producers of Reprocessed Medical Devices
Healthcare Providers and Hospitals
Distributors and Retailers of Medical Devices
Health Insurance Companies
Industry Associations (e.g., Association of Medical Device Reprocessors)
Financial Institutions and Investment Banks
Players Mentioned in the Report:
Stryker Corporation (Stryker Sustainability Solutions)
Johnson & Johnson MedTech (including DePuy Synthes & Ethicon)
Medline Industries, LP
Cardinal Health, Inc.
Arjo AB
Innovative Health, LLC
NEScientific, Inc.
SureTek Medical
Soma Tech Intl
Steris plc
3M Company
Olympus Corporation
GE Healthcare Technologies Inc.
Vanguard AG
ReNu Medical, Inc.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Reprocessed Medical Devices Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Reprocessed Medical Devices Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Reprocessed Medical Devices Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Cost-Effective Healthcare Solutions
3.1.2 Rising Focus on Sustainability and Environmental Impact
3.1.3 Technological Advancements in Reprocessing Techniques
3.1.4 Growing Regulatory Support for Reprocessed Devices
3.2 Market Challenges
3.2.1 Concerns Over Safety and Efficacy
3.2.2 Limited Awareness Among Healthcare Providers
3.2.3 Stringent Regulatory Requirements
3.2.4 Competition from New Medical Device Innovations
3.3 Market Opportunities
3.3.1 Expansion into Emerging Markets
3.3.2 Development of Advanced Reprocessing Technologies
3.3.3 Partnerships with Healthcare Institutions
3.3.4 Increasing Adoption of Circular Economy Practices
3.4 Market Trends
3.4.1 Shift Towards Value-Based Healthcare
3.4.2 Integration of Digital Technologies in Reprocessing
3.4.3 Growing Demand for Single-Use Devices
3.4.4 Enhanced Focus on Patient Safety and Quality Assurance
3.5 Government Regulation
3.5.1 FDA Guidelines on Reprocessed Devices
3.5.2 EU Medical Device Regulation Compliance
3.5.3 National Health Policies Supporting Reprocessing
3.5.4 Environmental Regulations Impacting Device Disposal
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Reprocessed Medical Devices Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Reprocessed Medical Devices Market Segmentation
8.1 By Type
8.1.1 Third-Party Reprocessing
8.1.2 In-House Reprocessing
8.2 By Device Type
8.2.1 Electrophysiology Devices
8.2.2 Cardiovascular Devices
8.2.3 Laparoscopic Devices
8.2.4 General Surgery Devices
8.2.5 Gastroenterology Devices
8.2.6 Orthopedic Devices
8.2.7 Endoscopy Devices
8.2.8 Respiratory and Anesthesia Devices
8.2.9 Others
8.3 By Application
8.3.1 Cardiology
8.3.2 Orthopedics
8.3.3 Gastroenterology
8.3.4 General Surgery
8.3.5 Urology
8.3.6 Neurology
8.3.7 Oncology
8.3.8 Others
8.4 By Material
8.4.1 Metal
8.4.2 Plastic
8.4.3 Silicone & Rubber
8.4.4 Composite Materials
8.4.5 Others
8.5 By End-User
8.5.1 Hospitals
8.5.2 Ambulatory Surgical Centers
8.5.3 Clinics
8.5.4 Community Healthcare Centers
8.5.5 Diagnostic Laboratories
8.5.6 Home Healthcare
8.5.7 Others
8.6 By Distribution Channel
8.6.1 Direct Sales
8.6.2 Distributors
8.6.3 Group Purchasing Organizations (GPOs)
8.6.4 Online Sales
8.6.5 Others
8.7 By Region
8.7.1 North America
8.7.2 Europe
8.7.3 Asia-Pacific
8.7.4 Latin America
8.7.5 Middle East & Africa
8.8 By Regulatory Compliance
8.8.1 FDA-Cleared Devices
8.8.2 CE-Marked Devices
8.8.3 ISO 13485-Certified Facilities
8.8.4 Other National Regulatory Approvals
9. Global Reprocessed Medical Devices Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Core Business Model (Third-Party vs In-House Support)
9.2.3 Group Size (Large, Medium, or Small as per industry convention)
9.2.4 Global Reprocessed Device Revenue (USD Million)
9.2.5 Revenue Growth Rate in Reprocessed Segment (%)
9.2.6 Installed Customer Base (Number of Hospitals / ASCs Served)
9.2.7 Geographic Footprint (Number of Countries / Regions Served)
9.2.8 Product Portfolio Breadth (Number of Device Categories Reprocessed)
9.2.9 Average Device Reprocessing Cycles Achieved
9.2.10 Average Cost Savings Delivered to Providers (%)
9.2.11 Regulatory Compliance Coverage (FDA, CE, ISO, Others)
9.2.12 R&D Intensity in Reprocessing (% of Revenue)
9.2.13 Strategic Partnerships with Healthcare Systems / GPOs
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Stryker Corporation (Stryker Sustainability Solutions)
9.5.2 Johnson & Johnson MedTech (including DePuy Synthes & Ethicon)
9.5.3 Medline Industries, LP
9.5.4 Cardinal Health, Inc.
9.5.5 Arjo AB
9.5.6 Innovative Health, LLC
9.5.7 NEScientific, Inc.
9.5.8 SureTek Medical
9.5.9 Soma Tech Intl
9.5.10 Steris plc
9.5.11 3M Company
9.5.12 Olympus Corporation
9.5.13 GE Healthcare Technologies Inc.
9.5.14 Vanguard AG
9.5.15 ReNu Medical, Inc.
10. Global Reprocessed Medical Devices Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Compliance Requirements
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends in Healthcare Infrastructure
10.2.2 Spending on Reprocessed Devices
10.2.3 Budgeting for Sustainability Initiatives
10.2.4 Cost-Benefit Analysis of Reprocessed Devices
10.3 Pain Point Analysis by End-User Category
10.3.1 Cost Constraints
10.3.2 Quality Assurance Issues
10.3.3 Supply Chain Disruptions
10.3.4 Regulatory Compliance Challenges
10.4 User Readiness for Adoption
10.4.1 Training and Education Needs
10.4.2 Perception of Reprocessed Devices
10.4.3 Infrastructure Readiness
10.4.4 Support from Healthcare Authorities
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Cost Savings
10.5.2 User Feedback and Satisfaction
10.5.3 Opportunities for Further Integration
10.5.4 Long-Term Impact on Healthcare Costs
11. Global Reprocessed Medical Devices Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-Ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-Sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-Term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from medical device regulatory bodies and health organizations
- Review of published market studies and white papers on reprocessed medical devices
- Examination of trade publications and journals focusing on medical device innovations and sustainability
Primary Research
- Interviews with key opinion leaders in the healthcare sector, including hospital administrators and procurement officers
- Surveys conducted with medical device manufacturers and reprocessing companies
- Field interviews with healthcare professionals involved in the usage and evaluation of reprocessed devices
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including sales data and regulatory compliance reports
- Triangulation of insights from primary interviews with secondary research findings
- Sanity checks performed through expert panel reviews and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on overall healthcare expenditure and medical device spending trends
- Segmentation of the market by device type, end-user, and geographical region
- Incorporation of growth rates from government initiatives promoting sustainability in healthcare
Bottom-up Modeling
- Collection of sales data from leading reprocessed medical device manufacturers
- Estimation of market volume based on the number of procedures utilizing reprocessed devices
- Cost analysis based on pricing models of reprocessed versus new medical devices
Forecasting & Scenario Analysis
- Multi-variable regression analysis incorporating factors such as healthcare policy changes and technological advancements
- Scenario modeling based on potential shifts in regulatory frameworks and market acceptance of reprocessed devices
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 140 | Procurement Managers, Supply Chain Coordinators |
| Reprocessing Facilities | 90 | Operations Managers, Quality Assurance Specialists |
| Healthcare Providers | 110 | Surgeons, Clinical Directors |
| Regulatory Bodies | 70 | Regulatory Affairs Managers, Compliance Officers |
| Medical Device Manufacturers | 80 | Product Development Managers, Marketing Directors |
Frequently Asked Questions
What is the current value of the Global Reprocessed Medical Devices Market?
The Global Reprocessed Medical Devices Market is valued at approximately USD 3.3 billion, reflecting a significant growth trend driven by cost reduction in healthcare, environmental sustainability, and the rising prevalence of chronic diseases.