About the Report
Base Year 2024GCC Reprocessed Medical Devices Market Overview
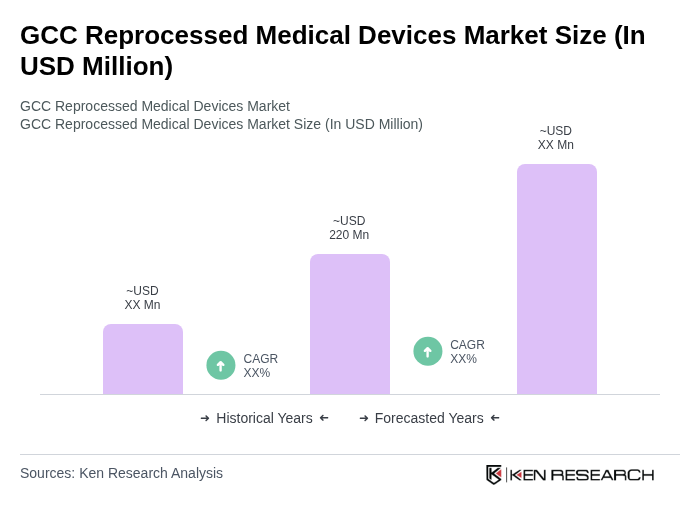
- The GCC Reprocessed Medical Devices Market is valued at USD 220 million, based on a five-year historical analysis and benchmarked against the global medical device reprocessing market and the GCC share in overall medical devices spending. This growth is primarily driven by the increasing demand for cost-effective healthcare solutions, rising awareness about sustainability, and the need to reduce medical waste, as reprocessed devices can offer significant cost savings and waste reduction for hospitals. The market is also supported by advancements in reprocessing technologies, such as validated cleaning, sterilization, and functional testing protocols, and by regulatory frameworks that promote the safe reuse of medical devices under strict quality and documentation requirements.
- Key players in this market include Saudi Arabia and the United Arab Emirates, which dominate due to their advanced healthcare infrastructure, high healthcare expenditure, and a growing population, resulting in higher per-capita medical device consumption compared with other GCC states. These countries have developed regulatory and institutional frameworks for medical devices that incorporate requirements for labeling, quality management systems, and conformity assessment, which support the controlled adoption of reprocessed medical devices in accredited hospitals and large healthcare groups.
- In 2023, the Saudi Food and Drug Authority (SFDA) further strengthened the oversight of reprocessed medical devices through the Medical Devices Interim Regulation and associated implementing rules, which mandate that all medical devices, including reprocessed devices placed on the Saudi market, comply with essential safety and performance requirements, undergo conformity assessment, and be registered in the SFDA system before use. These provisions require manufacturers, importers, and reprocessors to implement quality management systems, maintain traceability, and provide evidence of biocompatibility, cleaning, sterilization validation, and functional testing, thereby enhancing patient safety and promoting the responsible use of reprocessed devices in healthcare settings across the GCC region.
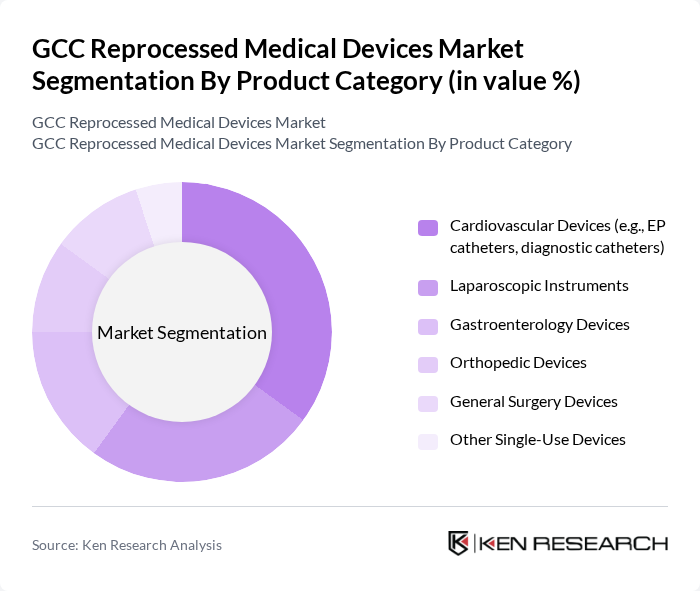
GCC Reprocessed Medical Devices Market Segmentation
By Product Category:The product category segmentation includes various types of medical devices that are commonly reprocessed. Among these, cardiovascular devices, such as electrophysiology (EP) catheters and diagnostic catheters, are leading the market due to their high usage rates in hospitals and the significant cost savings associated with their reprocessing, consistent with global trends where cardiovascular products form the largest reprocessed category. Laparoscopic instruments and gastroenterology devices also hold substantial market shares, driven by the increasing number of minimally invasive surgeries and the presence of robust decontamination and sterilization workflows in large GCC hospitals. Orthopedic devices and general surgery devices are gaining traction as healthcare providers seek to optimize costs while maintaining quality care, particularly in high-volume procedure areas where reprocessing can generate measurable savings per case. Other single-use devices, including certain accessories and pressure monitoring lines, are also being reprocessed, although they represent a smaller segment of the market due to more limited clinical indications and reprocessing protocols.
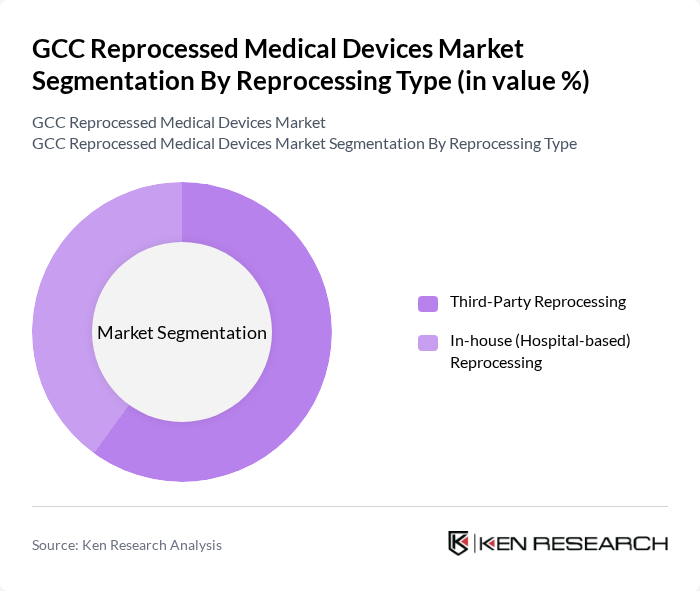
By Reprocessing Type:The reprocessing type segmentation is divided into third-party reprocessing and in-house (hospital-based) reprocessing. Third-party reprocessing is gaining popularity due to its cost-effectiveness and the ability to meet stringent regulatory standards, as specialized reprocessors typically operate under certified quality management systems and validated cleaning, sterilization, and performance testing protocols. Hospitals are increasingly outsourcing reprocessing to specialized companies that ensure compliance with safety regulations and provide documentation to support audits and accreditation requirements. In-house reprocessing is also prevalent, particularly in larger healthcare facilities that have the resources to manage their own reprocessing programs and central sterile services departments. This segment is driven by the desire for control over quality, integration with existing sterilization workflows, and the potential for significant cost savings in high-volume device categories.
GCC Reprocessed Medical Devices Market Competitive Landscape
The GCC Reprocessed Medical Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Stryker Corporation (Stryker Sustainability Solutions), Johnson & Johnson MedTech, Medline Industries, LP, Innovative Health, Inc., NEScientific, Inc., Arjo AB, Cardinal Health, Inc., SureTek Medical, Soma Tech Intl, GE Healthcare, Terumo Corporation, Olympus Corporation, Zimmer Biomet, Smith & Nephew, Local / Regional GCC Reprocessors (Representative Profiles) contribute to innovation, geographic expansion, and service delivery in this space.
GCC Reprocessed Medical Devices Market Industry Analysis
Growth Drivers
- Increasing Demand for Cost-Effective Healthcare Solutions:The GCC region is experiencing a significant rise in healthcare expenditure, projected to reach $120 billion by 2025. This surge is driven by a growing population and increasing prevalence of chronic diseases. Reprocessed medical devices offer a cost-effective alternative, reducing expenses for healthcare providers while maintaining quality. The demand for affordable healthcare solutions is further supported by government initiatives aimed at enhancing healthcare accessibility, making reprocessed devices an attractive option for hospitals and clinics.
- Rising Awareness of Environmental Sustainability:The GCC countries are increasingly prioritizing sustainability, with initiatives like Saudi Arabia's Vision 2030 aiming to reduce waste and promote recycling. In future, the region is expected to generate over 2 million tons of medical waste, highlighting the urgent need for sustainable practices. Reprocessed medical devices align with these goals, as they significantly reduce waste and resource consumption. This growing awareness among healthcare providers and patients is driving the adoption of reprocessed devices, contributing to a more sustainable healthcare system.
- Technological Advancements in Reprocessing Techniques:Innovations in reprocessing technologies are enhancing the safety and efficacy of reprocessed medical devices. In future, the market is expected to see a 25% increase in the adoption of advanced sterilization methods, such as ethylene oxide and hydrogen peroxide plasma. These advancements not only improve the quality of reprocessed devices but also address safety concerns, making them more appealing to healthcare providers. As technology continues to evolve, the reprocessed medical devices market is likely to expand, driven by improved reprocessing standards and practices.
Market Challenges
- Concerns Over Safety and Efficacy:Despite advancements, safety and efficacy concerns remain significant barriers to the adoption of reprocessed medical devices. In future, approximately 35% of healthcare providers in the GCC express skepticism regarding the reliability of these devices. High-profile recalls and adverse events associated with reprocessed products have heightened these concerns. Addressing these issues through rigorous testing and transparent communication is essential for building trust and encouraging wider acceptance among healthcare professionals.
- Limited Awareness Among Healthcare Providers:A substantial knowledge gap exists among healthcare providers regarding the benefits and applications of reprocessed medical devices. In future, surveys indicate that only 45% of healthcare professionals are familiar with the advantages of reprocessing. This lack of awareness can hinder the adoption of reprocessed devices, as many providers may default to using new devices due to perceived reliability. Educational initiatives and outreach programs are crucial to bridging this gap and promoting the benefits of reprocessed options.
GCC Reprocessed Medical Devices Market Future Outlook
The future of the GCC reprocessed medical devices market appears promising, driven by increasing healthcare costs and a growing emphasis on sustainability. As healthcare providers seek cost-effective solutions, the demand for reprocessed devices is expected to rise. Additionally, advancements in technology will likely enhance the safety and efficacy of these products, further encouraging adoption. Collaborative efforts between manufacturers and healthcare institutions will play a pivotal role in overcoming challenges and expanding market reach, ultimately contributing to a more sustainable healthcare landscape in the region.
Market Opportunities
- Expansion into Emerging Markets:The GCC region presents significant opportunities for reprocessed medical device manufacturers to expand into emerging markets. With healthcare spending projected to grow by 12% annually in countries like Oman and Bahrain, there is a rising demand for cost-effective medical solutions. Targeting these markets can enhance revenue streams and promote sustainable practices in healthcare delivery.
- Collaborations with Healthcare Institutions:Forming strategic partnerships with healthcare institutions can create substantial opportunities for reprocessed medical device companies. Collaborations can facilitate access to larger patient populations and enhance product visibility. By working together, manufacturers and healthcare providers can develop tailored solutions that meet specific needs, driving innovation and improving patient outcomes in the GCC region.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Category | Cardiovascular Devices (e.g., EP catheters, diagnostic catheters) Laparoscopic Instruments Gastroenterology Devices Orthopedic Devices General Surgery Devices Other Single-Use Devices |
| By Reprocessing Type | Third-Party Reprocessing In-house (Hospital-based) Reprocessing |
| By Risk / Device Classification | Low-Risk / Non-Critical Devices Semi-Critical Devices High-Risk / Critical Devices |
| By End-User | Hospitals Ambulatory Surgical Centers (ASCs) Specialty Clinics Others |
| By Country | Saudi Arabia United Arab Emirates Qatar Kuwait Oman Bahrain |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Gulf Cooperation Council, Saudi Food and Drug Authority, Ministry of Health - UAE)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Reprocessing Companies
Industry Associations (e.g., Gulf Medical Devices Association)
Financial Institutions
Players Mentioned in the Report:
Stryker Corporation (Stryker Sustainability Solutions)
Johnson & Johnson MedTech
Medline Industries, LP
Innovative Health, Inc.
NEScientific, Inc.
Arjo AB
Cardinal Health, Inc.
SureTek Medical
Soma Tech Intl
GE Healthcare
Terumo Corporation
Olympus Corporation
Zimmer Biomet
Smith & Nephew
Local / Regional GCC Reprocessors (Representative Profiles)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. GCC Reprocessed Medical Devices Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 GCC Reprocessed Medical Devices Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. GCC Reprocessed Medical Devices Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Cost-Effective Healthcare Solutions
3.1.2 Rising Awareness of Environmental Sustainability
3.1.3 Technological Advancements in Reprocessing Techniques
3.1.4 Supportive Government Policies and Regulations
3.2 Market Challenges
3.2.1 Concerns Over Safety and Efficacy
3.2.2 Limited Awareness Among Healthcare Providers
3.2.3 Stringent Regulatory Requirements
3.2.4 Competition from New Medical Devices
3.3 Market Opportunities
3.3.1 Expansion into Emerging Markets
3.3.2 Collaborations with Healthcare Institutions
3.3.3 Development of Innovative Reprocessing Technologies
3.3.4 Increasing Focus on Preventive Healthcare
3.4 Market Trends
3.4.1 Growing Adoption of Circular Economy Practices
3.4.2 Integration of IoT in Medical Device Reprocessing
3.4.3 Shift Towards Value-Based Healthcare Models
3.4.4 Rising Investment in Healthcare Infrastructure
3.5 Government Regulation
3.5.1 Implementation of Reprocessing Standards
3.5.2 Guidelines for Safety and Efficacy Testing
3.5.3 Regulatory Framework for Market Entry
3.5.4 Incentives for Sustainable Practices
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. GCC Reprocessed Medical Devices Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. GCC Reprocessed Medical Devices Market Segmentation
8.1 By Product Category
8.1.1 Cardiovascular Devices (e.g., EP catheters, diagnostic catheters)
8.1.2 Laparoscopic Instruments
8.1.3 Gastroenterology Devices
8.1.4 Orthopedic Devices
8.1.5 General Surgery Devices
8.1.6 Other Single-Use Devices
8.2 By Reprocessing Type
8.2.1 Third-Party Reprocessing
8.2.2 In-house (Hospital-based) Reprocessing
8.3 By Risk / Device Classification
8.3.1 Low-Risk / Non-Critical Devices
8.3.2 Semi-Critical Devices
8.3.3 High-Risk / Critical Devices
8.4 By End-User
8.4.1 Hospitals
8.4.2 Ambulatory Surgical Centers (ASCs)
8.4.3 Specialty Clinics
8.4.4 Others
8.5 By Country
8.5.1 Saudi Arabia
8.5.2 United Arab Emirates
8.5.3 Qatar
8.5.4 Kuwait
8.5.5 Oman
8.5.6 Bahrain
9. GCC Reprocessed Medical Devices Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 GCC Reprocessed Devices Revenue (USD, latest FY)
9.2.4 GCC Reprocessed Devices Revenue CAGR (3–5 Years)
9.2.5 EBITDA Margin from Reprocessed Portfolio (%)
9.2.6 Share of Reprocessed Devices in Total GCC Device Revenue (%)
9.2.7 Average Selling Price vs New Devices (Discount %)
9.2.8 Contracted Hospitals / ASCs in GCC (Number)
9.2.9 Devices Reprocessed per Year in GCC (Units)
9.2.10 Regulatory / Quality Certifications Held (e.g., SFDA, DHA, ISO)
9.2.11 R&D or Innovation Spend on Reprocessing (% of Segment Revenue)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Stryker Corporation (Stryker Sustainability Solutions)
9.5.2 Johnson & Johnson MedTech
9.5.3 Medline Industries, LP
9.5.4 Innovative Health, Inc.
9.5.5 NEScientific, Inc.
9.5.6 Arjo AB
9.5.7 Cardinal Health, Inc.
9.5.8 SureTek Medical
9.5.9 Soma Tech Intl
9.5.10 GE Healthcare
9.5.11 Terumo Corporation
9.5.12 Olympus Corporation
9.5.13 Zimmer Biomet
9.5.14 Smith & Nephew
9.5.15 Local / Regional GCC Reprocessors (Representative Profiles)
10. GCC Reprocessed Medical Devices Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Compliance Requirements
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends
10.2.2 Key Projects
10.2.3 Funding Sources
10.2.4 Strategic Partnerships
10.3 Pain Point Analysis by End-User Category
10.3.1 Cost Constraints
10.3.2 Quality Concerns
10.3.3 Supply Chain Issues
10.3.4 Regulatory Compliance Challenges
10.4 User Readiness for Adoption
10.4.1 Training Needs
10.4.2 Technology Acceptance
10.4.3 Infrastructure Readiness
10.4.4 Support Services
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 Case Studies
10.5.3 User Feedback
10.5.4 Future Expansion Plans
11. GCC Reprocessed Medical Devices Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams
1.4 Cost Structure Analysis
1.5 Key Partnerships
1.6 Customer Segments
1.7 Channels
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Audience Identification
2.4 Communication Strategy
2.5 Digital Marketing Approaches
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 E-commerce Integration
3.4 Logistics and Supply Chain Management
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
5.3 Emerging Trends
5.4 Future Needs Assessment
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Feedback Mechanisms
6.4 Community Engagement
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Customer-Centric Innovations
7.4 Competitive Differentiation
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Training and Development
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Performance Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from health authorities and medical device regulatory bodies in the GCC region
- Review of published articles and white papers on reprocessed medical devices from industry journals
- Examination of trade publications and market analysis reports focusing on medical device recycling and sustainability trends
Primary Research
- Interviews with key opinion leaders in the healthcare sector, including hospital administrators and procurement officers
- Surveys conducted with medical device manufacturers and reprocessing companies to gather insights on market dynamics
- Field interviews with healthcare professionals to understand the acceptance and usage of reprocessed devices
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including government health statistics and industry reports
- Triangulation of qualitative insights from interviews with quantitative data from surveys
- Sanity checks performed through expert panel reviews to ensure data accuracy and relevance
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market for reprocessed medical devices based on healthcare expenditure in the GCC
- Segmentation of the market by device type, including surgical instruments, diagnostic devices, and consumables
- Incorporation of regional healthcare policies promoting the use of sustainable medical practices
Bottom-up Modeling
- Collection of sales data from leading reprocessing firms to establish baseline market volumes
- Operational cost analysis of reprocessing facilities to determine pricing structures
- Volume estimates based on device usage rates in hospitals and clinics across the GCC
Forecasting & Scenario Analysis
- Multi-variable forecasting models incorporating trends in healthcare spending and environmental regulations
- Scenario analysis based on potential shifts in consumer attitudes towards sustainability and reprocessed devices
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospitals and Healthcare Facilities | 120 | Procurement Managers, Clinical Directors |
| Medical Device Manufacturers | 90 | Product Managers, R&D Heads |
| Reprocessing Companies | 70 | Operations Managers, Quality Assurance Officers |
| Regulatory Bodies | 40 | Policy Makers, Compliance Officers |
| Healthcare Professionals | 110 | Surgeons, Nurses, Medical Technologists |
Frequently Asked Questions
What is the current value of the GCC Reprocessed Medical Devices Market?
The GCC Reprocessed Medical Devices Market is valued at approximately USD 220 million, reflecting a significant growth trend driven by the demand for cost-effective healthcare solutions and sustainability initiatives in the region.