Region:Global
Author(s):Dev
Product Code:KRAE0069
Pages:94
Published On:December 2025
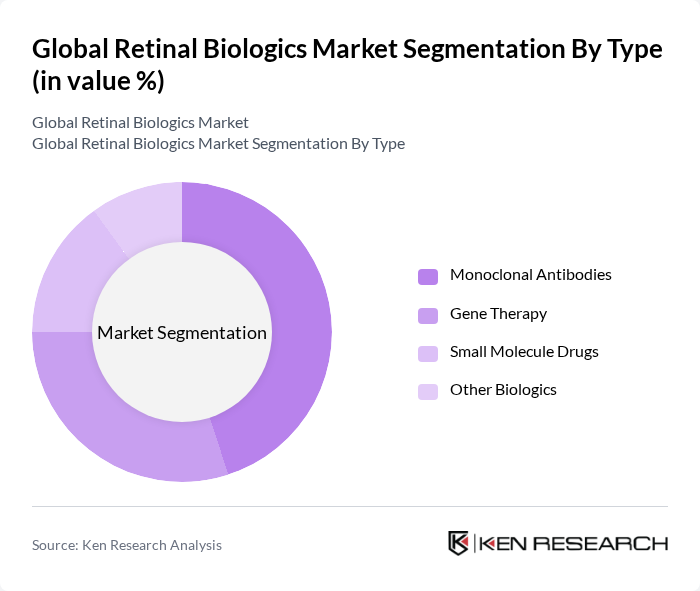
By Type:The market is segmented into four main types: Monoclonal Antibodies, Gene Therapy, Small Molecule Drugs, and Other Biologics. Monoclonal antibodies are currently the leading subsegment due to their targeted action and effectiveness in treating various retinal diseases. Gene therapy is gaining traction as a revolutionary approach, while small molecule drugs continue to play a significant role in treatment regimens.
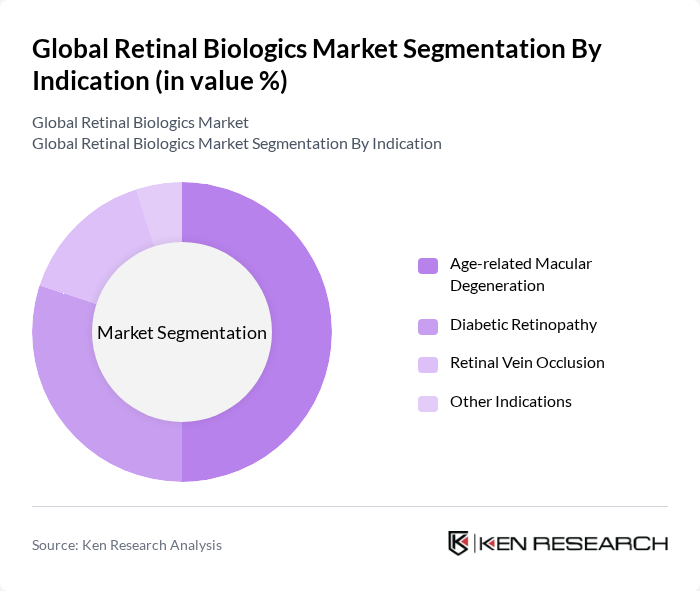
By Indication:The market is categorized by indications including Age-related Macular Degeneration, Diabetic Retinopathy, Retinal Vein Occlusion, and Other Indications. Age-related Macular Degeneration is the dominant indication, driven by the high prevalence of this condition among the elderly population. Diabetic Retinopathy follows closely, reflecting the rising incidence of diabetes globally.
The Global Retinal Biologics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Regeneron Pharmaceuticals, Inc., Novartis AG, Roche Holding AG, Bayer AG, AbbVie Inc., Amgen Inc., Santen Pharmaceutical Co., Ltd., Alcon Inc., EyePoint Pharmaceuticals, Inc., Spark Therapeutics, Inc., Apellis Pharmaceuticals, Inc., Iveric Bio, Inc., Genentech, Inc., Ocular Therapeutix, Inc., Eyenovia, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the retinal biologics market appears promising, driven by ongoing advancements in personalized medicine and the integration of digital health solutions. As healthcare providers increasingly adopt telemedicine for retinal care, patient access to specialized treatments will improve. Additionally, the focus on patient-centric approaches will enhance treatment adherence and outcomes. The market is expected to evolve with innovative therapies and technologies, addressing the growing demand for effective retinal disease management while overcoming existing challenges in accessibility and affordability.
| Segment | Sub-Segments |
|---|---|
| By Type | Monoclonal Antibodies Gene Therapy Small Molecule Drugs Other Biologics |
| By Indication | Age-related Macular Degeneration Diabetic Retinopathy Retinal Vein Occlusion Other Indications |
| By Route of Administration | Intravitreal Injection Subretinal Injection Systemic Administration Other Routes |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Other Channels |
| By Geography | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients Other Demographics |
| By Others | Combination Therapies Emerging Biologics Innovative Drug Delivery Systems |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Ophthalmology Clinics | 150 | Ophthalmologists, Clinic Managers |
| Pharmaceutical Companies | 100 | Product Managers, R&D Directors |
| Patient Advocacy Groups | 80 | Patient Representatives, Healthcare Advocates |
| Healthcare Payers | 70 | Insurance Analysts, Policy Makers |
| Clinical Research Organizations | 60 | Clinical Trial Managers, Regulatory Affairs Specialists |
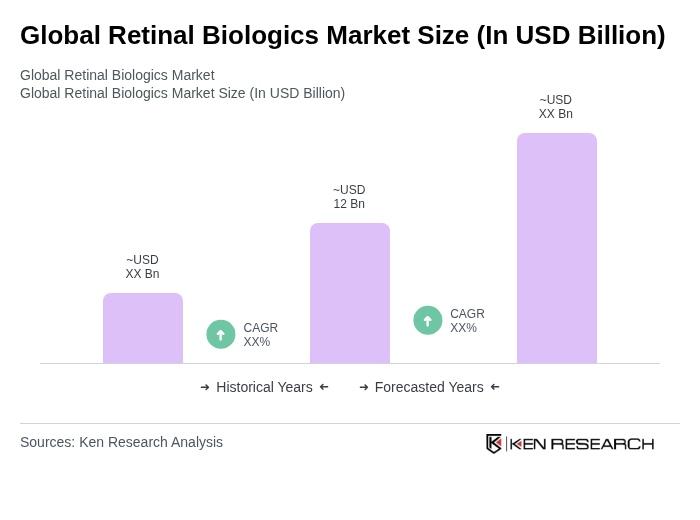
The Global Retinal Biologics Market is valued at approximately USD 12 billion, reflecting significant growth driven by the increasing prevalence of retinal diseases, advancements in biologic therapies, and a growing aging population.