Region:Global
Author(s):Rebecca
Product Code:KRAC2890
Pages:84
Published On:January 2026
By Type:The market is segmented into various types of clinical trials, including preclinical trials, Phase I, Phase II, Phase III, Phase IV, and others. Each type plays a crucial role in the development and approval of veterinary products, with specific methodologies and objectives tailored to different stages of research.
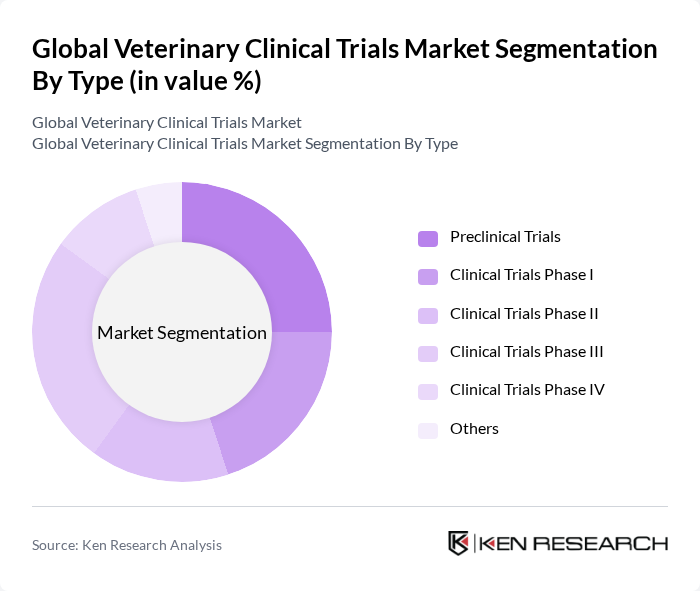
The Phase III Trials segment dominates the market due to its critical role in assessing the efficacy and safety of veterinary products before they receive regulatory approval. This phase involves a larger population and is essential for gathering comprehensive data on the product's performance in real-world settings. The increasing complexity of veterinary diseases and the demand for effective treatments further drive the need for extensive Phase III trials.
By Animal Type:The market is segmented based on the type of animals involved in clinical trials, including companion animals, livestock, exotic animals, and others. Each segment has unique requirements and considerations that influence the design and execution of clinical trials.
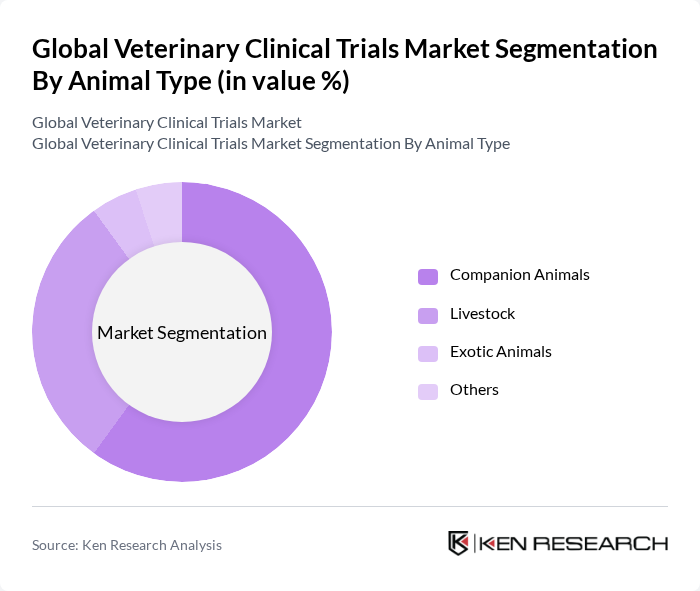
The Companion Animals segment leads the market, driven by the growing trend of pet ownership and the increasing willingness of pet owners to invest in their pets' health. This segment encompasses a wide range of animals, including dogs and cats, which are often the focus of innovative treatments and therapies, leading to a higher volume of clinical trials.
The Global Veterinary Clinical Trials Market is characterized by a dynamic mix of regional and international players. Leading participants such as Covance, Charles River Laboratories, Elanco Animal Health, Zoetis, Merck Animal Health, Boehringer Ingelheim, IDEXX Laboratories, Neogen Corporation, VetMedTeam, Animalytix, VetCT, VetPartners, Vetoquinol, PetDx, Trupanion contribute to innovation, geographic expansion, and service delivery in this space.
The future of the veterinary clinical trials market appears promising, driven by increasing investments in animal health research and the growing demand for innovative treatment options. As the industry embraces digital technologies and telemedicine, the efficiency of clinical trials is expected to improve significantly. Furthermore, the collaboration between academia and industry is likely to foster the development of personalized veterinary medicine, enhancing treatment efficacy and animal welfare in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Preclinical Trials Phase I Trials Phase II Trials Phase III Trials Phase IV Trials Others |
| By Animal Type | Companion Animals Livestock Exotic Animals Others |
| By Therapeutic Area | Infectious Diseases Oncology Orthopedic Disorders Dermatology Others |
| By Study Design | Randomized Controlled Trials Observational Studies Longitudinal Studies Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Funding Source | Government Grants Private Investments Non-Profit Organizations Others |
| By Duration | Short-term Trials Long-term Trials Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Veterinary Pharmaceutical Trials | 120 | Veterinary Researchers, Clinical Trial Managers |
| Animal Health Product Development | 100 | Product Development Scientists, Regulatory Affairs Managers |
| Livestock Clinical Trials | 80 | Livestock Veterinarians, Farm Managers |
| Pet Care Clinical Studies | 100 | Pet Veterinarians, Animal Behaviorists |
| Veterinary CRO Insights | 90 | CRO Executives, Clinical Operations Directors |
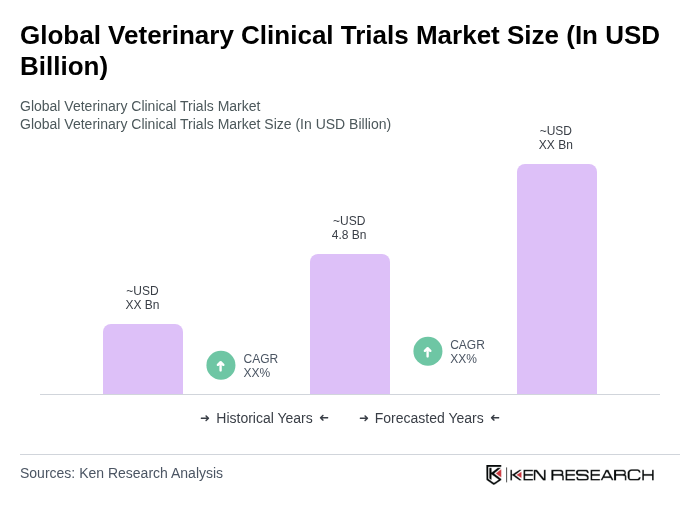
The Global Veterinary Clinical Trials Market is valued at approximately USD 4.8 billion, reflecting significant growth driven by factors such as increasing pet ownership, the prevalence of zoonotic diseases, and advancements in veterinary medicine and technology.