Region:Asia
Author(s):Geetanshi
Product Code:KRAC9474
Pages:85
Published On:November 2025
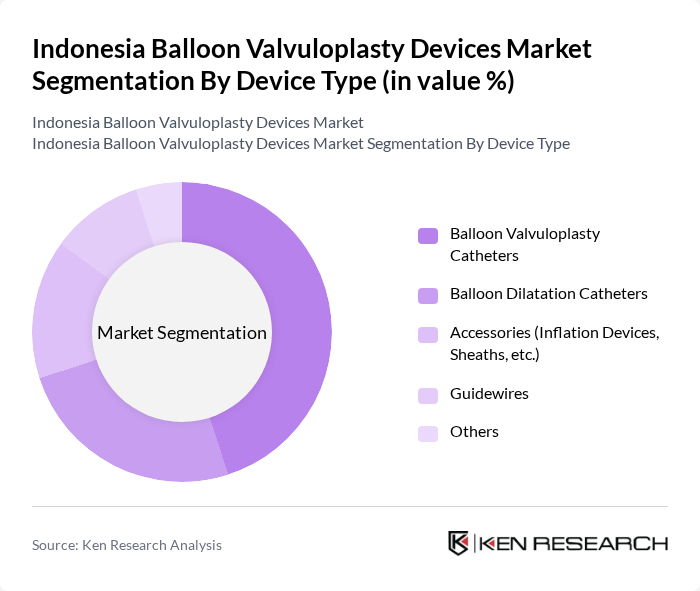
By Device Type:The device type segmentation includes Balloon Valvuloplasty Catheters, Balloon Dilatation Catheters, Accessories (Inflation Devices, Sheaths, etc.), Guidewires, and Others. Balloon Valvuloplasty Catheters remain the most dominant segment due to their essential role in the procedure and high utilization rates in hospitals and clinics. The rising number of cardiac interventions, driven by increasing disease prevalence and improved access to advanced treatment options, has led to greater consumption of these catheters, making them a critical component in the market .
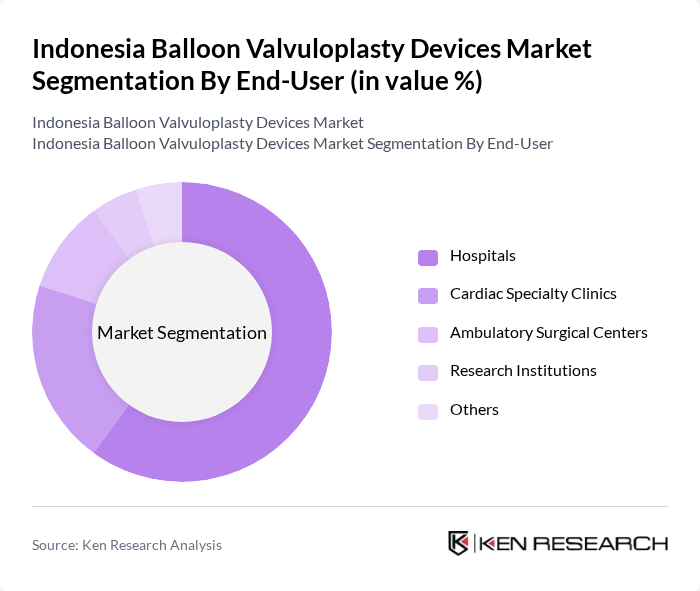
By End-User:The end-user segmentation comprises Hospitals, Cardiac Specialty Clinics, Ambulatory Surgical Centers, Research Institutions, and Others. Hospitals are the leading end-users, accounting for the majority of market share. This dominance is attributed to the high volume of cardiac procedures performed in hospitals, the availability of advanced medical technologies, and the presence of skilled healthcare professionals. Cardiac specialty clinics and ambulatory surgical centers are also expanding their roles, driven by increased demand for specialized and outpatient cardiac care .
The Indonesia Balloon Valvuloplasty Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Boston Scientific Corporation, Medtronic plc, Abbott Laboratories, Edwards Lifesciences Corporation, Terumo Corporation, B. Braun Melsungen AG, Cook Medical, Johnson & Johnson (Ethicon/Cardiovascular), St. Jude Medical, LLC (now part of Abbott), Cardiovascular Systems, Inc., Merit Medical Systems, Inc., Stryker Corporation, Philips Healthcare, GE HealthCare, Siemens Healthineers, PT Enseval Medika Prima, PT Kimia Farma Tbk, PT Prasasti Mitra Medika contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Indonesia balloon valvuloplasty devices market appears promising, driven by ongoing healthcare reforms and technological advancements. As the government continues to invest in healthcare infrastructure, the availability of advanced medical devices is expected to increase. Additionally, the growing geriatric population, projected to reach 25 million in future, will further fuel demand for cardiovascular treatments, including balloon valvuloplasty, enhancing patient care and outcomes across the nation.
| Segment | Sub-Segments |
|---|---|
| By Device Type | Balloon Valvuloplasty Catheters Balloon Dilatation Catheters Accessories (Inflation Devices, Sheaths, etc.) Guidewires Others |
| By End-User | Hospitals Cardiac Specialty Clinics Ambulatory Surgical Centers Research Institutions Others |
| By Procedure Type | Aortic Balloon Valvuloplasty Mitral Balloon Valvuloplasty Pulmonary Balloon Valvuloplasty Others |
| By Distribution Channel | Direct Sales Medical Device Distributors Online Sales Others |
| By Region | Java Sumatra Bali Kalimantan Sulawesi |
| By Patient Demographics | Age Group (Pediatric, Adult, Geriatric) Gender (Male, Female) Others |
| By Clinical Application | Rheumatic Heart Disease Congenital Heart Disease Degenerative Valve Disease Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiology Departments in Major Hospitals | 45 | Cardiologists, Interventional Cardiologists |
| Medical Device Distributors | 35 | Procurement Managers, Sales Directors |
| Healthcare Administrators | 30 | Hospital Administrators, Clinical Managers |
| Patient Advocacy Groups | 20 | Patient Representatives, Health Advocates |
| Regulatory Bodies | 15 | Regulatory Affairs Specialists, Compliance Officers |
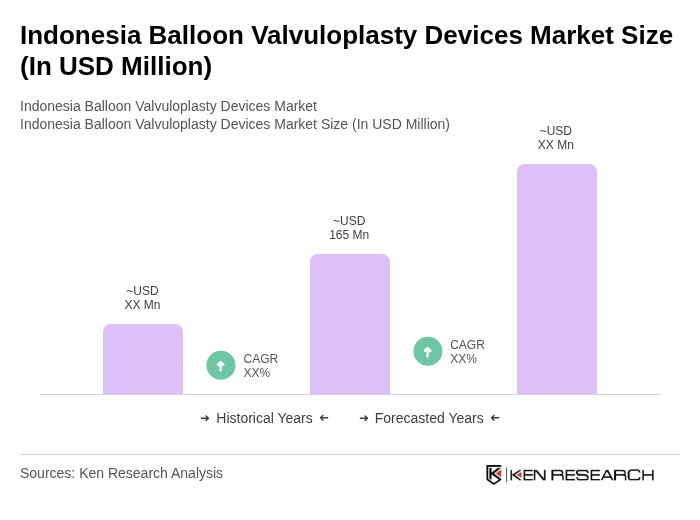
The Indonesia Balloon Valvuloplasty Devices Market is valued at approximately USD 165 million, reflecting a significant increase driven by the rising prevalence of heart diseases and advancements in medical technology.