About the Report
Base Year 2024Listen to the audio summary
Indonesia Endoscopy Operative Devices Market Overview
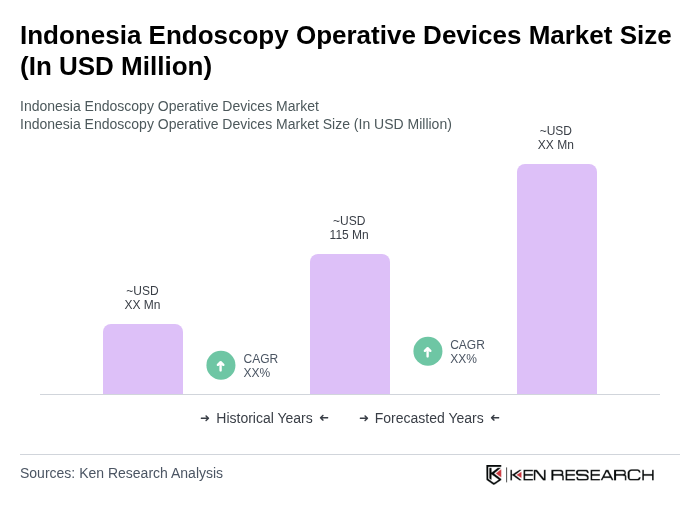
- The Indonesia Endoscopy Operative Devices Market is valued at USD 115 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of gastrointestinal and other chronic diseases, rising healthcare expenditure, and continuous advancements in endoscopic imaging, therapeutic accessories, and minimally invasive surgical platforms. The demand for minimally invasive procedures has surged, leading to higher adoption of flexible endoscopes, disposable endoscopes, and advanced visualization systems across gastroenterology, pulmonology, urology, and laparoscopic surgery.
- Key cities such as Jakarta, Surabaya, and Bandung dominate the market due to their advanced healthcare infrastructure and concentration of tertiary and referral hospitals, specialty clinics, and teaching institutions. These urban centers are equipped with state-of-the-art public and private hospitals that perform high volumes of gastrointestinal, pulmonary, and laparoscopic procedures using modern endoscopic systems, thereby driving market growth in the region.
- In 2023, the Indonesian government strengthened the regulatory framework for medical devices, including endoscopy equipment, through the implementation and enforcement of the Regulation of the Minister of Health of the Republic of Indonesia Number 14 of 2021 concerning the Organization and Work Procedure of the Directorate General of Pharmaceuticals and Medical Devices and related technical regulations under the Ministry of Health’s medical device registration system (Alat Kesehatan/Alkes), which mandate that all endoscopic devices marketed in Indonesia must be registered, tested, and certified to meet safety, quality, and performance standards aligned with international benchmarks. These requirements cover conformity assessment, post?market surveillance, and import licensing, thereby improving patient safety, clinical outcomes, and trust in endoscopic technologies among healthcare providers.
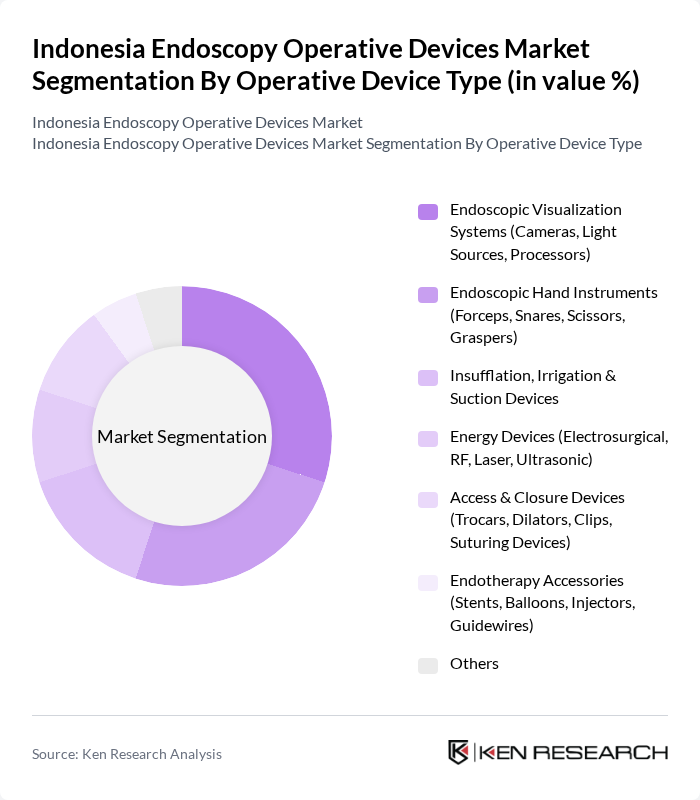
Indonesia Endoscopy Operative Devices Market Segmentation
By Product Type:The product type segmentation includes various categories of endoscopic devices that cater to different medical needs. The market is characterized by a diverse range of products, including rigid endoscopes, flexible endoscopes, capsule endoscopes, disposable endoscopes, and robot-assisted platforms, reflecting the structure used in recent Indonesia endoscopy market studies. Among these, flexible endoscopes are particularly popular due to their versatility and ease of use in gastrointestinal, respiratory, and urological procedures, while disposable endoscopes are gaining traction to reduce infection risk and reprocessing burden.

By Operative Device Type:This segmentation focuses on the various operative devices used in endoscopic procedures. It includes visualization systems, hand instruments, insufflation devices, energy devices, access and closure systems, and endotherapy accessories, which aligns with the major hardware and accessory categories tracked in Indonesia and global endoscopy device reports. The demand for endoscopic visualization systems is particularly high, as high?definition and 3D imaging, enhanced light sources, and advanced processors are essential for providing clear images during procedures, thus enhancing surgical precision and enabling complex therapeutic interventions.
Indonesia Endoscopy Operative Devices Market Competitive Landscape
The Indonesia Endoscopy Operative Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT. Medika Sarana Trijaya, PT Indofarma Tbk, PT Itama Ranoraya Tbk, PT Enseval Putera Megatrading Tbk, PT Dharma Kencana Utama, Olympus Corporation, Karl Storz SE & Co. KG, Fujifilm Holdings Corporation, Stryker Corporation, Boston Scientific Corporation, Medtronic plc, Johnson & Johnson (Ethicon, DePuy Synthes), CONMED Corporation, HOYA Corporation (PENTAX Medical), Smith & Nephew plc, B. Braun Melsungen AG, Teleflex Incorporated, Terumo Corporation contribute to innovation, geographic expansion, and service delivery in this space.
Indonesia Endoscopy Operative Devices Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Gastrointestinal Diseases:The rise in gastrointestinal diseases in Indonesia is significant, with over 10 million cases reported annually. According to the World Health Organization, the incidence of colorectal cancer has increased by 25% in the last decade. This growing health concern drives the demand for endoscopic procedures, as they are essential for diagnosis and treatment. The increasing awareness of gastrointestinal health among the population further fuels the need for advanced endoscopic devices.
- Advancements in Endoscopic Technology:Technological innovations in endoscopy, such as high-definition imaging and minimally invasive techniques, have transformed the landscape of gastrointestinal diagnostics. The Indonesian market has seen a 35% increase in the adoption of advanced endoscopic devices over the past five years. These advancements not only enhance diagnostic accuracy but also improve patient outcomes, leading to a higher demand for these devices in hospitals and clinics across the country.
- Rising Demand for Minimally Invasive Surgeries:The shift towards minimally invasive surgical procedures is evident in Indonesia, with a reported 30% increase in such surgeries from 2019 to 2023. Patients prefer these procedures due to shorter recovery times and reduced hospital stays. This trend is supported by the Indonesian Ministry of Health, which promotes minimally invasive techniques as part of its healthcare improvement initiatives, further driving the demand for endoscopic devices.
Market Challenges
- High Cost of Advanced Endoscopic Devices:The cost of advanced endoscopic devices remains a significant barrier in Indonesia, with prices ranging from IDR 50 million to IDR 200 million (approximately USD 3,500 to USD 14,000). This high cost limits accessibility for many healthcare facilities, particularly in rural areas. As a result, hospitals may struggle to invest in the latest technology, hindering the overall growth of the endoscopy market in the region.
- Limited Access to Healthcare Facilities in Rural Areas:Approximately 60% of Indonesia's population resides in rural areas, where access to healthcare facilities is severely limited. According to the Indonesian Health Ministry, only 30% of rural health centers are equipped with endoscopic devices. This disparity creates a significant challenge in providing adequate healthcare services, as patients in these regions often face delays in diagnosis and treatment, impacting the overall market growth.
Indonesia Endoscopy Operative Devices Market Future Outlook
The future of the Indonesia endoscopy operative devices market appears promising, driven by ongoing technological advancements and increasing healthcare investments. The government’s commitment to enhancing healthcare infrastructure is expected to facilitate greater access to endoscopic services. Additionally, the rising trend of medical tourism, with over 2 million international patients visiting Indonesia annually, will likely boost demand for advanced endoscopic procedures, further propelling market growth in the coming years.
Market Opportunities
- Expansion of Healthcare Facilities:The Indonesian government plans to invest IDR 100 trillion (approximately USD 7 billion) in healthcare infrastructure in future. This investment will lead to the establishment of new hospitals and clinics, increasing the availability of endoscopic services and devices, thereby creating significant market opportunities for manufacturers and suppliers.
- Growing Medical Tourism in Indonesia:Indonesia's medical tourism sector is projected to grow by 20% annually, attracting patients seeking affordable and high-quality healthcare services. This trend presents a lucrative opportunity for endoscopy device manufacturers, as the influx of international patients will drive demand for advanced diagnostic and therapeutic procedures, enhancing market potential.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Rigid Endoscopes Flexible Endoscopes Capsule Endoscopes Disposable / Single-use Endoscopes Endoscopic Ultrasound (EUS) Devices Robot-assisted Endoscopy Systems Others |
| By Operative Device Type | Endoscopic Visualization Systems (Cameras, Light Sources, Processors) Endoscopic Hand Instruments (Forceps, Snares, Scissors, Graspers) Insufflation, Irrigation & Suction Devices Energy Devices (Electrosurgical, RF, Laser, Ultrasonic) Access & Closure Devices (Trocars, Dilators, Clips, Suturing Devices) Endotherapy Accessories (Stents, Balloons, Injectors, Guidewires) Others |
| By Application | Gastrointestinal Endoscopy Pulmonology / Bronchoscopy Urology Endoscopy (Cystoscopy) Gynecology & Obstetrics Endoscopy Laparoscopy ENT Endoscopy Orthopedic / Arthroscopy Others |
| By End-User | Public Hospitals Private Hospitals Ambulatory Surgical Centers Specialty Clinics & Diagnostic Centers Academic & Research Institutions Others |
| By Hygiene / Usage Type | Reusable Devices Single-use / Disposable Devices Reprocessed Devices |
| By Distribution Channel | Direct Sales (Subsidiaries & Local Offices) Authorized Distributors / Dealers Group Purchasing Organizations (GPOs) & Tenders Online / E-procurement Platforms Others |
| By Region | Java Sumatra Bali & Nusa Tenggara Kalimantan Sulawesi Papua & Maluku Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, National Agency of Drug and Food Control)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Importers
Industry Associations
Financial Institutions
Players Mentioned in the Report:
PT. Medika Sarana Trijaya
PT Indofarma Tbk
PT Itama Ranoraya Tbk
PT Enseval Putera Megatrading Tbk
PT Dharma Kencana Utama
Olympus Corporation
Karl Storz SE & Co. KG
Fujifilm Holdings Corporation
Stryker Corporation
Boston Scientific Corporation
Medtronic plc
Johnson & Johnson (Ethicon, DePuy Synthes)
CONMED Corporation
HOYA Corporation (PENTAX Medical)
Smith & Nephew plc
B. Braun Melsungen AG
Teleflex Incorporated
Terumo Corporation
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Indonesia Endoscopy Operative Devices Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Indonesia Endoscopy Operative Devices Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Indonesia Endoscopy Operative Devices Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of gastrointestinal diseases
3.1.2 Advancements in endoscopic technology
3.1.3 Rising demand for minimally invasive surgeries
3.1.4 Government initiatives to improve healthcare infrastructure
3.2 Market Challenges
3.2.1 High cost of advanced endoscopic devices
3.2.2 Limited access to healthcare facilities in rural areas
3.2.3 Shortage of trained medical professionals
3.2.4 Stringent regulatory requirements
3.3 Market Opportunities
3.3.1 Expansion of healthcare facilities
3.3.2 Growing medical tourism in Indonesia
3.3.3 Increasing awareness about preventive healthcare
3.3.4 Development of innovative endoscopic solutions
3.4 Market Trends
3.4.1 Shift towards outpatient procedures
3.4.2 Integration of AI and robotics in endoscopy
3.4.3 Rising adoption of single-use endoscopic devices
3.4.4 Focus on patient-centric healthcare solutions
3.5 Government Regulation
3.5.1 Medical Device Regulation No. 16 of 2018
3.5.2 Health Minister Regulation No. 24 of 2016
3.5.3 Import Regulation for Medical Devices
3.5.4 Certification requirements for medical devices
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Indonesia Endoscopy Operative Devices Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Indonesia Endoscopy Operative Devices Market Segmentation
8.1 By Product Type
8.1.1 Rigid Endoscopes
8.1.2 Flexible Endoscopes
8.1.3 Capsule Endoscopes
8.1.4 Disposable / Single-use Endoscopes
8.1.5 Endoscopic Ultrasound (EUS) Devices
8.1.6 Robot-assisted Endoscopy Systems
8.1.7 Others
8.2 By Operative Device Type
8.2.1 Endoscopic Visualization Systems (Cameras, Light Sources, Processors)
8.2.2 Endoscopic Hand Instruments (Forceps, Snares, Scissors, Graspers)
8.2.3 Insufflation, Irrigation & Suction Devices
8.2.4 Energy Devices (Electrosurgical, RF, Laser, Ultrasonic)
8.2.5 Access & Closure Devices (Trocars, Dilators, Clips, Suturing Devices)
8.2.6 Endotherapy Accessories (Stents, Balloons, Injectors, Guidewires)
8.2.7 Others
8.3 By Application
8.3.1 Gastrointestinal Endoscopy
8.3.2 Pulmonology / Bronchoscopy
8.3.3 Urology Endoscopy (Cystoscopy)
8.3.4 Gynecology & Obstetrics Endoscopy
8.3.5 Laparoscopy
8.3.6 ENT Endoscopy
8.3.7 Orthopedic / Arthroscopy
8.3.8 Others
8.4 By End-User
8.4.1 Public Hospitals
8.4.2 Private Hospitals
8.4.3 Ambulatory Surgical Centers
8.4.4 Specialty Clinics & Diagnostic Centers
8.4.5 Academic & Research Institutions
8.4.6 Others
8.5 By Hygiene / Usage Type
8.5.1 Reusable Devices
8.5.2 Single-use / Disposable Devices
8.5.3 Reprocessed Devices
8.6 By Distribution Channel
8.6.1 Direct Sales (Subsidiaries & Local Offices)
8.6.2 Authorized Distributors / Dealers
8.6.3 Group Purchasing Organizations (GPOs) & Tenders
8.6.4 Online / E-procurement Platforms
8.6.5 Others
8.7 By Region
8.7.1 Java
8.7.2 Sumatra
8.7.3 Bali & Nusa Tenggara
8.7.4 Kalimantan
8.7.5 Sulawesi
8.7.6 Papua & Maluku
8.7.7 Others
9. Indonesia Endoscopy Operative Devices Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Size (Global / Regional / Local Player)
9.2.3 Indonesia Endoscopy Operative Devices Revenue (Latest Year, USD Mn)
9.2.4 3-year CAGR in Indonesia Endoscopy Operative Devices Revenue
9.2.5 Indonesia Market Share in Endoscopy Operative Devices (%)
9.2.6 Product Portfolio Breadth (No. of Endoscopy Operative SKUs / Categories)
9.2.7 R&D Intensity (% of Revenue Spent on R&D)
9.2.8 New Product Launches in Last 3 Years (Number)
9.2.9 Installed Base of Endoscopy Systems in Indonesia (Units)
9.2.10 Average Selling Price Positioning (vs. Market Index)
9.2.11 EBITDA Margin from Endoscopy Operative Devices Business (%)
9.2.12 Revenue Mix: Capital Equipment vs. Consumables & Accessories (%)
9.2.13 Channel Mix: Direct vs. Distributor-led Sales (%)
9.2.14 Geographic Coverage within Indonesia (No. of Provinces / Cities Served)
9.2.15 Key Hospital & ASC Accounts (Number of Strategic Accounts)
9.2.16 Average Tender Win Rate in Public Sector (%)
9.2.17 Average Contract Length with Key Accounts (Years)
9.2.18 Service & Maintenance Coverage (No. of Field Service Engineers)
9.2.19 Customer Satisfaction / NPS Score
9.2.20 Regulatory & Compliance Track Record (No. of Recalls / Warnings in Last 5 Years)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 PT. Medika Sarana Trijaya
9.5.2 PT Indofarma Tbk
9.5.3 PT Itama Ranoraya Tbk
9.5.4 PT Enseval Putera Megatrading Tbk
9.5.5 PT Dharma Kencana Utama
9.5.6 Olympus Corporation
9.5.7 Karl Storz SE & Co. KG
9.5.8 Fujifilm Holdings Corporation
9.5.9 Stryker Corporation
9.5.10 Boston Scientific Corporation
9.5.11 Medtronic plc
9.5.12 Johnson & Johnson (Ethicon, DePuy Synthes)
9.5.13 CONMED Corporation
9.5.14 HOYA Corporation (PENTAX Medical)
9.5.15 Smith & Nephew plc
9.5.16 B. Braun Melsungen AG
9.5.17 Teleflex Incorporated
9.5.18 Terumo Corporation
10. Indonesia Endoscopy Operative Devices Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Finance
10.1.3 Ministry of Education
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Hospital Infrastructure Development
10.2.2 Medical Equipment Procurement
10.2.3 Training and Development
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Research Institutions
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training Programs
10.4.2 Technology Familiarity
10.4.3 Infrastructure Availability
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings Analysis
10.5.2 Patient Outcome Improvement
10.5.3 Scalability of Solutions
10.5.4 Others
11. Indonesia Endoscopy Operative Devices Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from healthcare associations and medical device regulatory bodies in Indonesia
- Review of published market studies and white papers on endoscopy devices and trends
- Examination of government health expenditure reports and hospital procurement guidelines
Primary Research
- Interviews with key opinion leaders in gastroenterology and surgical departments
- Surveys with procurement managers at major hospitals and clinics across Indonesia
- Field interviews with endoscopy device manufacturers and distributors
Validation & Triangulation
- Cross-validation of findings through multiple data sources including sales data and expert insights
- Triangulation of market size estimates using both top-down and bottom-up approaches
- Sanity checks through expert panel reviews and feedback from industry stakeholders
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on national healthcare spending and endoscopy device penetration rates
- Segmentation of the market by device type, including flexible endoscopes, rigid endoscopes, and accessories
- Incorporation of demographic data and disease prevalence rates to refine market estimates
Bottom-up Modeling
- Collection of sales data from leading endoscopy device manufacturers operating in Indonesia
- Estimation of market share based on unit sales and revenue figures from key players
- Analysis of pricing strategies and average selling prices for various endoscopy devices
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as population growth, healthcare access, and technological advancements
- Scenario modeling based on potential regulatory changes and shifts in healthcare policy
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gastroenterology Departments | 100 | Gastroenterologists, Department Heads |
| Hospital Procurement Teams | 90 | Procurement Managers, Supply Chain Directors |
| Endoscopy Device Manufacturers | 60 | Product Managers, Sales Directors |
| Healthcare Policy Makers | 50 | Health Ministry Officials, Regulatory Experts |
| Clinical Research Organizations | 70 | Clinical Researchers, Project Managers |
Frequently Asked Questions
What is the current value of the Indonesia Endoscopy Operative Devices Market?
The Indonesia Endoscopy Operative Devices Market is valued at approximately USD 115 million, reflecting a significant growth driven by the rising prevalence of gastrointestinal diseases, increased healthcare expenditure, and advancements in endoscopic technologies.