Region:Asia
Author(s):Dev
Product Code:KRAD1777
Pages:96
Published On:November 2025
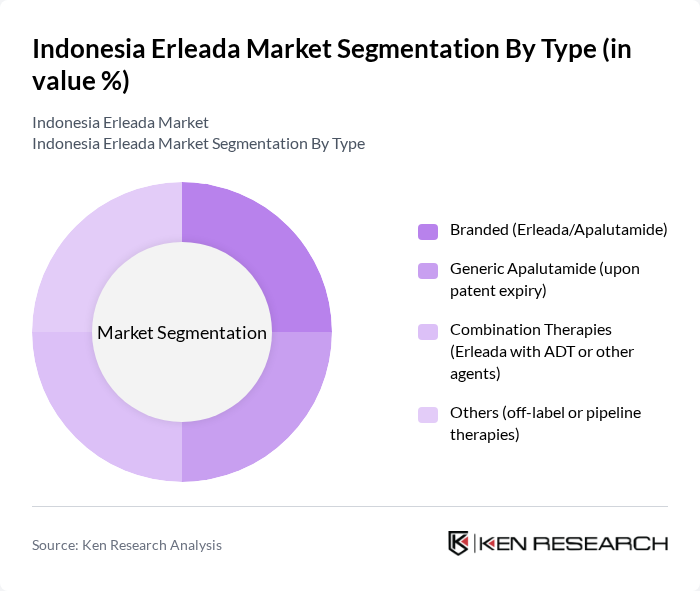
By Type:The market is segmented into branded therapies, generics, combination therapies, and others. Thebranded segment, particularly Erleada, currently leads the market due to its established efficacy, strong patent protection, and brand recognition among healthcare providers.Combination therapies(e.g., Erleada with androgen deprivation therapy) are gaining traction, reflecting updated clinical guidelines and the pursuit of improved patient outcomes. The generic segment is expected to expand following future patent expiries, in line with regional trends.
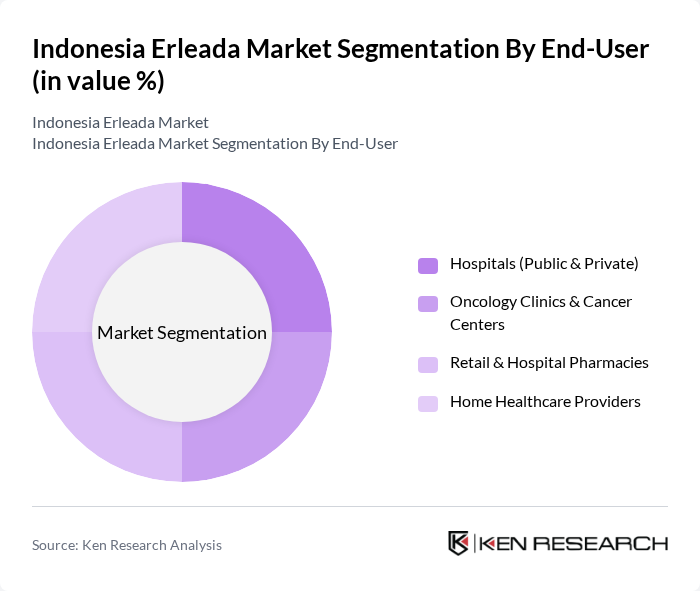
By End-User:The end-user segmentation includes hospitals, oncology clinics, retail pharmacies, and home healthcare providers.Hospitals, both public and private, dominate the market as the primary treatment centers for prostate cancer patients.Oncology clinicsand cancer centers are significant contributors, providing specialized care and driving adoption of advanced therapies. Retail and hospital pharmacies, as well as home healthcare providers, play a supporting role in medication access and patient management.
The Indonesia Erleada Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT Johnson & Johnson Indonesia (Janssen), PT Kalbe Farma Tbk, PT Kimia Farma Tbk, PT Dexa Medica, PT Pharos Indonesia, PT Enseval Putera Megatrading Tbk, PT Soho Global Health Tbk, PT Tempo Scan Pacific Tbk, PT Guardian Pharmatama, PT Mensa Bina Sukses, PT Otto Pharmaceutical Industries, PT Hexpharm Jaya Laboratories, PT Ferron Par Pharmaceuticals, PT Sido Muncul Tbk, and PT Indofarma Tbk contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Erleada market in Indonesia appears promising, driven by increasing investments in healthcare infrastructure and ongoing government support for cancer treatment initiatives. As awareness of prostate cancer and its treatments continues to grow, more patients are likely to seek advanced therapies. Additionally, the integration of digital health technologies and telemedicine is expected to enhance patient access to care, further propelling the market forward. Overall, the landscape is evolving positively, with significant opportunities for growth.
| Segment | Sub-Segments |
|---|---|
| By Type | Branded (Erleada/Apalutamide) Generic Apalutamide (upon patent expiry) Combination Therapies (Erleada with ADT or other agents) Others (off-label or pipeline therapies) |
| By End-User | Hospitals (Public & Private) Oncology Clinics & Cancer Centers Retail & Hospital Pharmacies Home Healthcare Providers |
| By Patient Demographics | Age Group (50–64, 65–74, 75+) Gender (Male) Socioeconomic Status (Low, Middle, High Income) Urban vs Rural Patients |
| By Treatment Stage | Non-metastatic Castration-Resistant Prostate Cancer (nmCRPC) Metastatic Castration-Resistant Prostate Cancer (mCRPC) Metastatic Hormone-Sensitive Prostate Cancer (mHSPC) Others (recurrence, off-label use) |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Direct Sales/Institutional Procurement |
| By Geographic Distribution | Java Sumatra Kalimantan Sulawesi & Other Islands |
| By Policy Support | Inclusion in National Formulary (e-Catalogue) BPJS Kesehatan Coverage Tax Incentives for Oncology Drugs Research & Clinical Trial Grants |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncologist Insights | 60 | Medical Oncologists, Urologists |
| Pharmacy Distribution | 50 | Pharmacy Managers, Retail Pharmacists |
| Patient Experience | 45 | Prostate Cancer Patients, Caregivers |
| Healthcare Policy Analysis | 40 | Health Economists, Policy Makers |
| Clinical Trial Feedback | 40 | Clinical Researchers, Trial Coordinators |
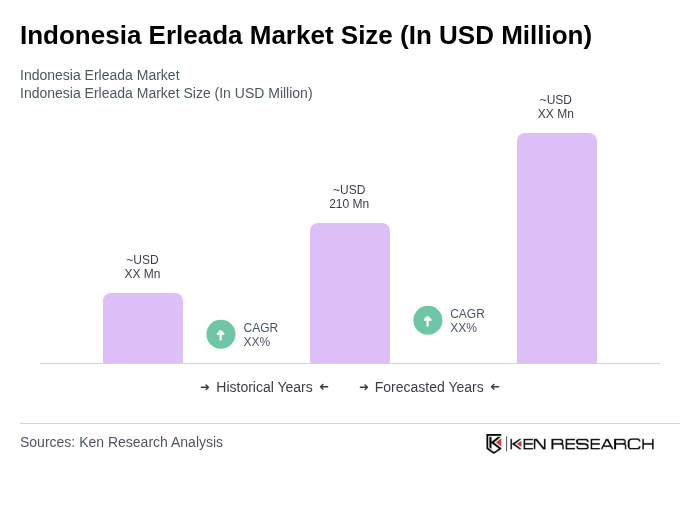
The Indonesia Erleada Market is valued at approximately USD 210 million, driven by the increasing prevalence of prostate cancer and advancements in targeted therapies. This valuation reflects a five-year historical analysis and normalization with regional Asia-Pacific market data.