Region:Asia
Author(s):Geetanshi
Product Code:KRAD4827
Pages:96
Published On:December 2025
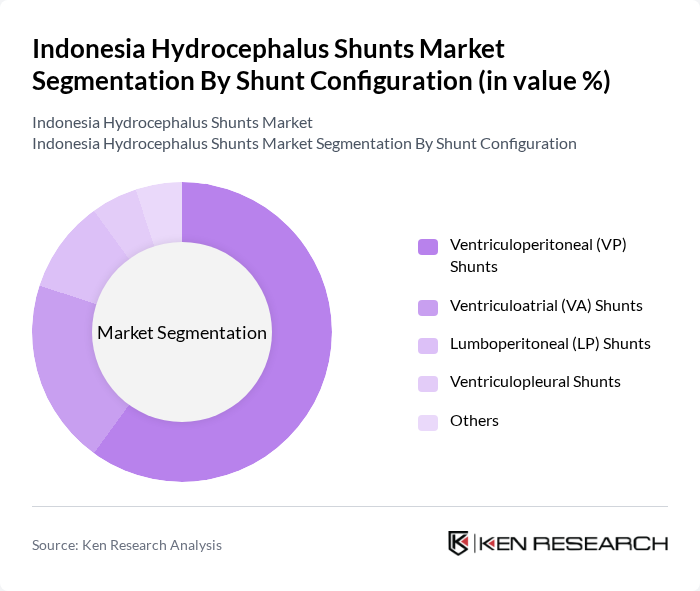
By Shunt Configuration:The market is segmented into various shunt configurations, each serving specific medical needs. The primary configurations include Ventriculoperitoneal (VP) Shunts, Ventriculoatrial (VA) Shunts, Lumboperitoneal (LP) Shunts, Ventriculopleural Shunts, and Others. Among these, VP shunts are the most commonly used worldwide and in Asia-Pacific due to their effectiveness in redirecting cerebrospinal fluid (CSF) from the ventricles to the peritoneal cavity and their broad clinical familiarity, making them the preferred choice for treating hydrocephalus.
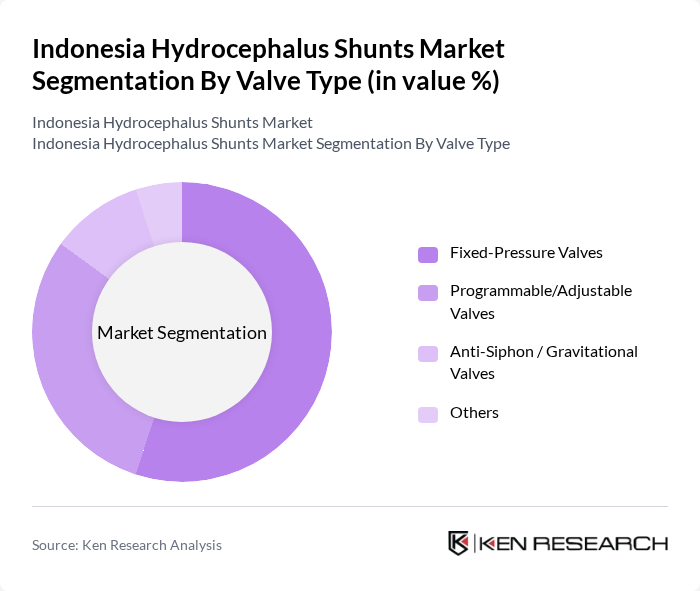
By Valve Type:The valve type segmentation includes Fixed-Pressure Valves, Programmable/Adjustable Valves, Anti-Siphon / Gravitational Valves, and Others. Fixed-Pressure Valves continue to hold a significant share in many emerging markets because of their relative simplicity, ease of use, and lower acquisition cost, which supports their widespread adoption in routine shunt procedures. However, the demand for programmable valves is increasing globally as they offer customizable pressure settings, reduce the need for revision surgeries, and can improve patient outcomes, particularly in complex or evolving hydrocephalus cases.
The Indonesia Hydrocephalus Shunts Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Integra LifeSciences Holdings Corporation, B. Braun Melsungen AG (Aesculap Division), Sophysa SA, Natus Medical Incorporated, Spiegelberg GmbH & Co. KG, Christoph Miethke GmbH & Co. KG, Tokibo Co., Ltd., Fuji Systems Corporation, Johnson & Johnson (DePuy Synthes), Stryker Corporation, R.M.S. Surgical (India), Neurona Medical, Local Indonesian Distributors (e.g., PT Enseval Putera Megatrading Tbk – Medical Devices Division), Other Regional Hydrocephalus Shunt Suppliers Active in Indonesia contribute to innovation, geographic expansion, and service delivery in this space.
The Indonesia hydrocephalus shunts market is poised for significant growth, driven by technological advancements and increased healthcare investments. As the government focuses on enhancing healthcare access, particularly in underserved regions, the demand for effective treatment options is expected to rise. Additionally, the integration of digital health technologies will facilitate better patient management and follow-up care, further supporting market expansion. The emphasis on patient-centered care will also play a crucial role in shaping future market dynamics.
| Segment | Sub-Segments |
|---|---|
| By Shunt Configuration | Ventriculoperitoneal (VP) Shunts Ventriculoatrial (VA) Shunts Lumboperitoneal (LP) Shunts Ventriculopleural Shunts Others |
| By Valve Type | Fixed-Pressure Valves Programmable/Adjustable Valves Anti-Siphon / Gravitational Valves Others |
| By Procedure Type | Primary Shunt Implantation Shunt Revision / Replacement Endoscopic Third Ventriculostomy (ETV) with Shunt Others |
| By Patient Group | Pediatric Patients (0–18 Years) Adult Patients (19–64 Years) Geriatric Patients (65+ Years) Others |
| By Material | Silicone-Based Shunts Polyurethane-Based Shunts Antibiotic-Impregnated Shunts Others |
| By End-User | Public / Government Hospitals Private Hospitals Specialized Neurosurgery & Tertiary Care Centers Others |
| By Distribution Channel | Direct Tender Sales to Hospitals Local Medical Device Distributors / Importers E-Procurement Platforms Others |
| By Region | Java (Including Jakarta) Sumatra Kalimantan Sulawesi Bali & Nusa Tenggara Papua & Maluku |
| By Policy & Reimbursement Environment | Coverage under BPJS Kesehatan (National Health Insurance) Central Government Procurement & E-Catalogue (e-Katalog) Local Content (TKDN) Requirements Grants & Funding for Neurosurgical Capacity Building Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurosurgeons in Major Hospitals | 60 | Neurosurgeons, Pediatric Surgeons |
| Hospital Administrators | 60 | Healthcare Administrators, Procurement Managers |
| Patient Advocacy Groups | 40 | Patient Advocates, Caregivers |
| Medical Device Distributors | 50 | Sales Representatives, Distribution Managers |
| Healthcare Policy Experts | 40 | Health Economists, Policy Analysts |
The Indonesia Hydrocephalus Shunts Market is valued at approximately USD 8 million, reflecting a steady growth driven by increasing prevalence of hydrocephalus, particularly in pediatric cases, advancements in shunt technology, and rising healthcare expenditures.