About the Report
Base Year 2024Indonesia Hypodermic Syringes Needles Market Overview
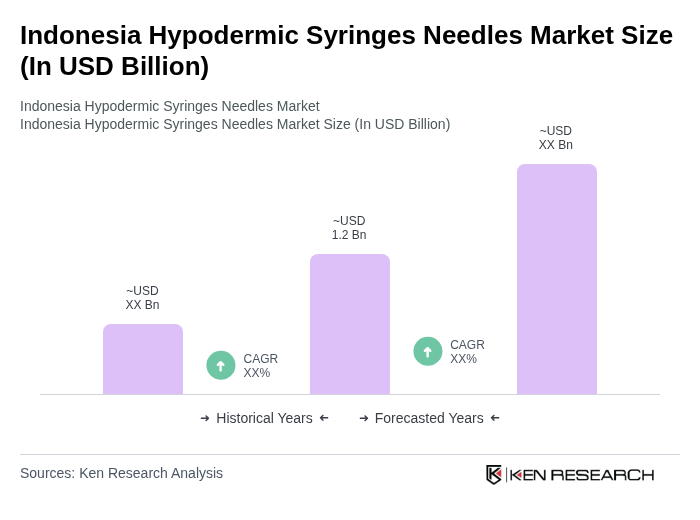
- The Indonesia Hypodermic Syringes Needles Market is valued at USD 1.2 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic diseases, the rise in vaccination programs, and the expansion of healthcare facilities across the country. The demand for hypodermic syringes is further fueled by advancements in medical technology and the growing emphasis on patient safety.
- Key cities such as Jakarta, Surabaya, and Bandung dominate the market due to their robust healthcare infrastructure and high population density. Jakarta, being the capital, serves as a hub for medical supplies and innovations, while Surabaya and Bandung have seen significant investments in healthcare facilities, contributing to the increased demand for hypodermic syringes and needles.
- In 2023, the Indonesian government implemented a regulation mandating the use of safety-engineered syringes in all public healthcare facilities. This regulation aims to reduce the risk of needle-stick injuries and enhance patient safety, thereby driving the demand for safety syringes in the market.
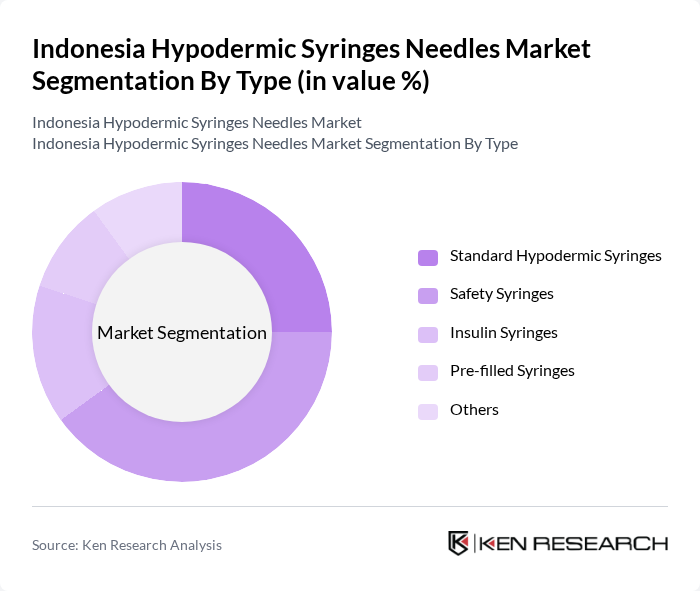
Indonesia Hypodermic Syringes Needles Market Segmentation
By Type:The market can be segmented into various types of hypodermic syringes, including Standard Hypodermic Syringes, Safety Syringes, Insulin Syringes, Pre-filled Syringes, and Others. Among these, Safety Syringes are gaining traction due to their ability to prevent needle-stick injuries, which is a significant concern in healthcare settings. The increasing focus on patient safety and infection control is driving the demand for these syringes, making them a leading sub-segment in the market.
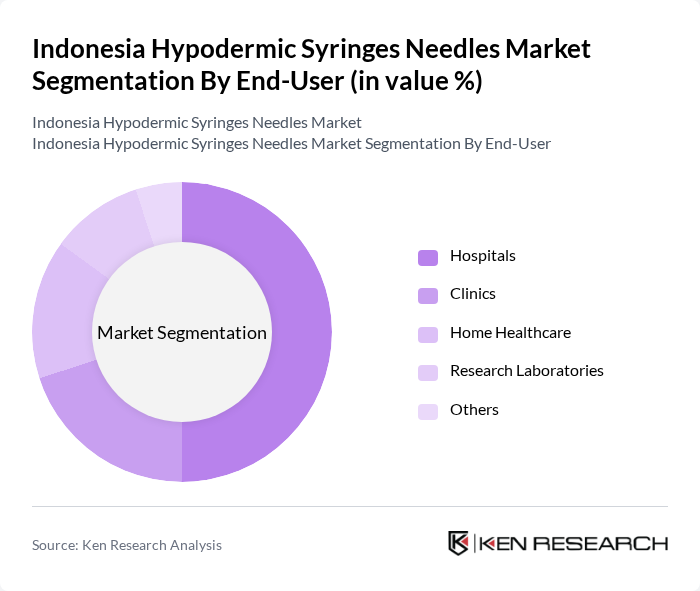
By End-User:The end-user segmentation includes Hospitals, Clinics, Home Healthcare, Research Laboratories, and Others. Hospitals are the dominant end-user segment, driven by the high volume of patients requiring injections and the need for safe and efficient drug delivery systems. The increasing number of hospitals and healthcare facilities in urban areas is further propelling the demand for hypodermic syringes.
Indonesia Hypodermic Syringes Needles Market Competitive Landscape
The Indonesia Hypodermic Syringes Needles Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT. Indofarma Tbk, PT. Kimia Farma Tbk, B. Braun Medical Indonesia, Terumo Indonesia, PT. Medisafe Technologies, PT. Sinar Medika, PT. Deltomed Laboratories, PT. Harsen Laboratories, PT. Surya Medika, PT. Anugerah Pharmindo Lestari, PT. Sido Muncul, PT. Karya Medika, PT. Medika Sarana Trijaya, PT. Citra Medika, PT. Sumber Daya Medika contribute to innovation, geographic expansion, and service delivery in this space.
Indonesia Hypodermic Syringes Needles Market Industry Analysis
Growth Drivers
- Increasing Healthcare Expenditure:Indonesia's healthcare expenditure is projected to reach IDR 1,200 trillion (approximately USD 84 billion) in future, reflecting a significant increase from IDR 1,000 trillion in the past. This rise is driven by government initiatives to enhance healthcare access and quality, which directly boosts the demand for hypodermic syringes and needles. As healthcare facilities expand, the need for medical supplies, including syringes, will continue to grow, supporting market development.
- Rising Prevalence of Chronic Diseases:The World Health Organization reported that chronic diseases account for 71% of all deaths in Indonesia, with diabetes and cardiovascular diseases being the most prevalent. In future, it is estimated that over 10 million Indonesians will be living with diabetes, necessitating regular insulin injections. This increasing prevalence drives demand for hypodermic syringes, as healthcare providers require reliable and safe injection methods to manage these conditions effectively.
- Expansion of Vaccination Programs:Indonesia's vaccination programs have expanded significantly, with the government aiming to vaccinate 70% of its population in future. This initiative includes routine immunizations and COVID-19 vaccinations, which require a substantial number of hypodermic syringes. The Ministry of Health has allocated IDR 50 trillion (approximately USD 3.5 billion) for vaccine procurement and distribution, further driving the demand for syringes and needles in the healthcare sector.
Market Challenges
- Stringent Regulatory Requirements:The Indonesian government enforces strict regulations on medical devices, including hypodermic syringes. Manufacturers must comply with the Ministry of Health's guidelines and obtain certifications, which can take up to 12 months. This lengthy approval process can hinder market entry for new players and slow down the introduction of innovative products, limiting competition and growth in the hypodermic syringe market.
- High Competition Among Local and International Players:The hypodermic syringes market in Indonesia is characterized by intense competition, with over 50 local and international manufacturers. This saturation leads to price wars, which can erode profit margins. For instance, local manufacturers often compete on price, making it challenging for international brands to maintain market share. This competitive landscape can stifle innovation and limit the overall growth potential of the market.
Indonesia Hypodermic Syringes Needles Market Future Outlook
The Indonesia hypodermic syringes needles market is poised for significant growth, driven by increasing healthcare investments and a rising focus on chronic disease management. As vaccination programs expand, the demand for safe and efficient injection methods will continue to rise. Additionally, technological advancements in syringe design and the growing emphasis on safety-engineered devices will shape the market landscape. Collaboration between manufacturers and healthcare providers will further enhance product offerings, ensuring that the market remains responsive to evolving healthcare needs.
Market Opportunities
- Technological Advancements in Syringe Design:Innovations such as smart syringes that track dosage and usage are gaining traction. In future, the demand for these advanced devices is expected to increase, driven by the need for improved patient safety and compliance. Manufacturers investing in R&D can capitalize on this trend, enhancing their product portfolios and market presence.
- Expansion of E-commerce Platforms for Medical Supplies:The rise of e-commerce in Indonesia presents a significant opportunity for hypodermic syringe manufacturers. With online sales projected to grow by 30% annually, companies can reach a broader customer base, including healthcare facilities in remote areas. This shift towards digital sales channels can enhance distribution efficiency and market penetration.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Standard Hypodermic Syringes Safety Syringes Insulin Syringes Pre-filled Syringes Others |
| By End-User | Hospitals Clinics Home Healthcare Research Laboratories Others |
| By Distribution Channel | Direct Sales Online Retail Medical Supply Distributors Pharmacies Others |
| By Material | Plastic Glass Metal Others |
| By Application | Vaccination Blood Sampling Drug Delivery Others |
| By Region | Java Sumatra Bali Kalimantan Others |
| By Regulatory Compliance | ISO Certification CE Marking FDA Approval Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, National Agency of Drug and Food Control)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Pharmaceutical Companies
Industry Associations
Financial Institutions
Players Mentioned in the Report:
PT. Indofarma Tbk
PT. Kimia Farma Tbk
B. Braun Medical Indonesia
Terumo Indonesia
PT. Medisafe Technologies
PT. Sinar Medika
PT. Deltomed Laboratories
PT. Harsen Laboratories
PT. Surya Medika
PT. Anugerah Pharmindo Lestari
PT. Sido Muncul
PT. Karya Medika
PT. Medika Sarana Trijaya
PT. Citra Medika
PT. Sumber Daya Medika
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Indonesia Hypodermic Syringes Needles Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Indonesia Hypodermic Syringes Needles Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Indonesia Hypodermic Syringes Needles Market Analysis
3.1 Growth Drivers
3.1.1 Increasing healthcare expenditure
3.1.2 Rising prevalence of chronic diseases
3.1.3 Expansion of vaccination programs
3.1.4 Growing awareness of safe injection practices
3.2 Market Challenges
3.2.1 Stringent regulatory requirements
3.2.2 High competition among local and international players
3.2.3 Fluctuating raw material prices
3.2.4 Limited access to healthcare in rural areas
3.3 Market Opportunities
3.3.1 Technological advancements in syringe design
3.3.2 Increasing demand for safety-engineered devices
3.3.3 Expansion of e-commerce platforms for medical supplies
3.3.4 Government initiatives to improve healthcare infrastructure
3.4 Market Trends
3.4.1 Shift towards disposable syringes
3.4.2 Growing adoption of smart syringes
3.4.3 Rising focus on sustainability in manufacturing
3.4.4 Increased collaboration between manufacturers and healthcare providers
3.5 Government Regulation
3.5.1 Compliance with WHO standards
3.5.2 Registration requirements for medical devices
3.5.3 Import regulations for syringes and needles
3.5.4 Guidelines for safe disposal of medical waste
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Indonesia Hypodermic Syringes Needles Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Indonesia Hypodermic Syringes Needles Market Segmentation
8.1 By Type
8.1.1 Standard Hypodermic Syringes
8.1.2 Safety Syringes
8.1.3 Insulin Syringes
8.1.4 Pre-filled Syringes
8.1.5 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Clinics
8.2.3 Home Healthcare
8.2.4 Research Laboratories
8.2.5 Others
8.3 By Distribution Channel
8.3.1 Direct Sales
8.3.2 Online Retail
8.3.3 Medical Supply Distributors
8.3.4 Pharmacies
8.3.5 Others
8.4 By Material
8.4.1 Plastic
8.4.2 Glass
8.4.3 Metal
8.4.4 Others
8.5 By Application
8.5.1 Vaccination
8.5.2 Blood Sampling
8.5.3 Drug Delivery
8.5.4 Others
8.6 By Region
8.6.1 Java
8.6.2 Sumatra
8.6.3 Bali
8.6.4 Kalimantan
8.6.5 Others
8.7 By Regulatory Compliance
8.7.1 ISO Certification
8.7.2 CE Marking
8.7.3 FDA Approval
8.7.4 Others
9. Indonesia Hypodermic Syringes Needles Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Product Innovation Rate
9.2.7 Distribution Efficiency
9.2.8 Pricing Strategy
9.2.9 Brand Recognition
9.2.10 Customer Satisfaction Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 PT. Indofarma Tbk
9.5.2 PT. Kimia Farma Tbk
9.5.3 B. Braun Medical Indonesia
9.5.4 Terumo Indonesia
9.5.5 PT. Medisafe Technologies
9.5.6 PT. Sinar Medika
9.5.7 PT. Deltomed Laboratories
9.5.8 PT. Harsen Laboratories
9.5.9 PT. Surya Medika
9.5.10 PT. Anugerah Pharmindo Lestari
9.5.11 PT. Sido Muncul
9.5.12 PT. Karya Medika
9.5.13 PT. Medika Sarana Trijaya
9.5.14 PT. Citra Medika
9.5.15 PT. Sumber Daya Medika
10. Indonesia Hypodermic Syringes Needles Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Education
10.1.3 Ministry of Defense
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Medical Equipment Procurement
10.2.3 Research and Development Funding
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Home Healthcare Providers
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training and Education Needs
10.4.2 Infrastructure Readiness
10.4.3 Financial Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings Analysis
10.5.2 Efficiency Gains
10.5.3 User Feedback and Adaptation
10.5.4 Others
11. Indonesia Hypodermic Syringes Needles Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from health organizations and government health departments in Indonesia
- Review of published market studies and white papers on hypodermic syringe usage and trends
- Examination of import/export data and trade statistics related to medical supplies
Primary Research
- Interviews with healthcare professionals, including doctors and nurses, to understand usage patterns
- Surveys with hospital procurement managers to gather insights on purchasing decisions
- Field interviews with manufacturers and distributors of hypodermic syringes in Indonesia
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including government health statistics and industry reports
- Triangulation of qualitative insights from interviews with quantitative data from surveys
- Sanity checks through expert panel reviews comprising industry veterans and market analysts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total market size based on national healthcare expenditure and hypodermic syringe allocation
- Segmentation by end-user categories such as hospitals, clinics, and home care settings
- Incorporation of government health initiatives promoting vaccination and diabetes management
Bottom-up Modeling
- Volume estimates derived from production data of leading syringe manufacturers in Indonesia
- Cost analysis based on pricing strategies of various syringe types and their distribution channels
- Calculation of market size using unit sales multiplied by average selling prices
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering population growth, healthcare access, and disease prevalence
- Scenario modeling based on potential regulatory changes and shifts in healthcare policies
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 100 | Procurement Managers, Supply Chain Coordinators |
| Healthcare Professionals | 150 | Doctors, Nurses, Medical Technicians |
| Pharmaceutical Distributors | 80 | Sales Managers, Distribution Coordinators |
| Public Health Officials | 60 | Health Policy Makers, Program Managers |
| Home Care Providers | 70 | Care Coordinators, Home Health Aides |
Frequently Asked Questions
What is the current value of the Indonesia Hypodermic Syringes Needles Market?
The Indonesia Hypodermic Syringes Needles Market is valued at approximately USD 1.2 billion, reflecting significant growth driven by factors such as chronic disease prevalence, vaccination programs, and advancements in medical technology.